Abstract

Introduction
Non-tuberculous mycobacteria (NTM) are environmental organisms found in soil and water throughout the world that can cause chronic lung infection, usually in the context of structural lung disease or immunodeficiency 1 There is considerable evidence that rates of NTM infection are increasing in patients with Cystic Fibrosis (CF). 2 In contrast to Mycobacterium tuberculosis, NTM do not seem to be transmissible from person to person nor re-activate following latent infection. 3 Observed increases in NTM infection in CF patients are therefore likely to reflect increased exposure and/or increased susceptibility to infection.
DECLARATIONS
None declared
Cystic Fibrosis Trust
Not applicable
UH
All authors contributed equally to the manuscript
This review aims to summarize the epidemiology diagnosis and treatment of NTM infection in CF patients focusing on the two most common mycobacterial groups causing disease: the M. avium-intracellulare complex (MAC) and the M. abscessus complex (MABSC).
Epidemiology
Prevalence of NTM in CF patients
Overall rates of recovery of NTM from the respiratory tract of CF patients vary from 3.7% to 24% depending on the geographical region and patient population. In the USA, a prospective cross-sectional study showed an overall prevalence rate of 13% (ranging from 7-24%), 4 whereas in France the prevalence rate was 6.6% (ranging from 3.7-9.6%). 2 The differences may reflect regional variation in the density of NTM in the environment, different laboratory sampling techniques and different patient populations studied at different time periods.
While, no human-to-human transmission has been proven to date, outbreaks in non-CF patients have been reported. For example soft tissue, bone and line infections have occurred following the use of contaminated surgical equipment. 3 Furthermore, disseminated NTM infection can occur in patients with genetic defects in the synthesis or response to interleukin -12 (IL-12) and interferon-γ (IFN-γ). 5
MAC and MABSC are the most frequently isolated NTM species in patients with CF2,4 Other NTM species found in the respiratory tract of CF patients include: M. gordonae, M. kansasii, M. lentiflavum, M. malmoense, M. terrae and M. simiae.2,6 However, CF patients are most likely to experience a decline in lung function with NTM pulmonary disease related to MABSC. 7
Risk factors for NTM infection
A large multicentre prevalence study in the US showed that CF patients with NTM positive sputum cultures were older, had a higher FEV1 and lower frequency of Pseudomonas aeruginosa pulmonary infection than patients without NTM. 4 In contrast, an Israeli study reported a significant association between the growth of Aspergillus species and NTM in sputum cultures of CF patients. 6 They also noted that markers of severe disease, such as lower FEV1, haemoptysis and higher frequency of Pseudomonas aeruginosa were correlated with NTM infection. 6 This discrepancy might be related to the mycobacterial species involved; MAC being the predominant mycobacterial pathogen in the USA, whereas MABSC is more frequent in Israel and Europe.
Interestingly, the long term use of azithromycin in patients with CF was recently shown to be associated with increased rates of NTM infection in a large UK CF centre. 8 Azithromycin at concentrations achievable during therapeutic dosing blocked intracellular killing of mycobacteria within macrophages and led to chronic NTM infection in a mouse model. 8
Finally the increased use of steroids and allergic bronchopulmonary aspergillosis (ABPA) are two other factors that have been linked to NTM infection in CF patients. 9 The altered immune response associated with ABPA, particularly the T-helper 2 mediated up-regulation of IL-4 and down-regulation of INF-γ, is likely to represent a favourable milieu for mycobacterial growth.
Diagnosis
Criteria for the diagnosis of NTM lung disease
As NTM are widely distributed in the environment, it may be difficult to determine whether an NTM isolate from a sputum culture is indicative of pulmonary disease. Guidelines for the diagnosis and management of NTM disease were published by the American Thoracic Society (ATS) in 2007. 1 However, these are not specific to patients with CF. In order for patients to be diagnosed with NTM pulmonary disease, they should meet certain clinical, radiological and microbiological criteria (Table 1).
ATS criteria for the diagnosis of NTM pulmonary disease (based on Ref 1)
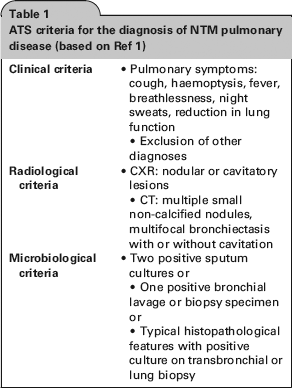
Microbiological investigations
In order to successfully isolate mycobacteria from sputum samples, it is necessary to include a decontamination step with N-acetyl-L-cysteine-sodium hydroxide (NALC/NaOH) which inhibits growth of other bacteria. 1 As respiratory tract specimens from CF patients often contain large numbers of aerobic gram-negative rods, an additional decontamination step with oxalic acid reduces overgrowth and increases the yield of NTM on culture. 10
Current techniques for the speciation of NTM include commercial polymerase chain reaction-based assays, high-performance liquid chromatography which identifies mycobacteria on the basis of the long chain fatty acids in the cell wall and 16 S ribosomal RNA sequencing. Recently, multilocus sequencing of the rpoB, hsp65 and secA genes has been used to further speciate the MABSC into M. abscessus sensu stricto, M. massiliense and M. bolletti. 11 All three species are thought to be pathogenic since the proportion of cases satisfying ATS criteria for NTM pulmonary disease was similar. 2
In order to determine appropriate antibiotic treatment, susceptibility testing should be performed, but it is clear that in vitro sensitivity does not always correlate with the clinical response to therapy in NTM lung disease. 1 For MAC, it is recommended that isolates are tested for susceptibility to macrolides, as there appears to be a correlation with clinical response. MABSC species are often resistant to multiple antibiotics in vitro and it is recommended that MABSC isolates are tested for susceptibility to amikacin, doxycycline, fluoroquinolones, sulfonamides, cefoxitin, clarithromycin and linezolid. 1
Management
Treatment for NTM pulmonary disease should be for at least 12 months and involves multiple antibiotics with frequent side effects. However, mycobacterial eradication may not be possible, particularly in the case of MABSC infection. It is therefore crucial that all potential outcomes are discussed with patients before starting treatment.
Some patients with NTM isolates may not meet all of the ATS criteria for disease. In these circumstances, patients require close monitoring of their clinical status with serial CT scans/sputum surveillance and if necessary, bronchoscopy with bronchoalveolar lavage. Some antibiotics which are used to treat traditional CF pathogens such as Pseudomonas aeruginosa have some activity against NTM (such as macrolides and fluoroquinolones). These should be withheld for an adequate time period prior to assessment to facilitate NTM culture.
Azithromycin is increasingly used as an immunomodulatory agent in CF patients. Prior to initiating azithromycin, it is important to screen patients for NTM, as resistance can develop rapidly in the context of macrolide monotherapy 1
The ATS treatment guidelines for NTM pulmonary disease are outlined below. 1 CF-specific management guidelines are being developed jointly by the European Cystic Fibrosis Society and the North American Cystic Fibrosis Foundation. As the treatment of NTM pulmonary disease (particularly that caused by MABSC) can be complex and associated with many drug-related side effects, it is recommended that treatment is conducted in liaison with a physician with experience in NTM pulmonary disease. Prior to the initiation of therapy, a CT scan of the chest should be performed as a baseline to assess radiological response to treatment.
Treatment of MAC pulmonary disease
The main anti-mycobacterial agents in the treatment of MAC are the macrolides (azithromycin or clarithromycin), ethambutol and rifampicin. In cases of severe cavitatory disease or previously treated infection, ATS guidelines suggest the addition of an injectable aminoglycoside, such as amikacin, during the initial two to three months of treatment.
Ideally, therapy should be continued for at least 12 months after sputum conversion. It is therefore imperative that sputum samples are sent for AFB culture regularly (perhaps monthly) while patients are on treatment.
To date, no studies examining the response to treatment of MAC lung disease in CF patients have been published. However, in non-CF patients with MAC-induced nodular or bronchiectatic pulmonary lesions, sputum culture conversion rates of over 90% were reported after 18 months of combination chemotherapy with clarithromycin, ethambutol and rifampicin. 12 Relapse rates are very low unless the treatment is stopped prematurely 13 Analysis of isolates from patients with recurrent MAC infection following successful initial treatment revealed infection with new strains rather than relapse. 13
MAC can acquire resistance to macrolides via a point mutation in the 23S r-RNA gene. 14 Risk factors for the development of resistance include macrolide monotherapy or combination chemotherapy with a macrolide and quinolone only 14 Aggressive combination treatment involving an injectable aminoglycoside is necessary when treating macrolide-resistant MAC pulmonary disease. 1
Treatment of MABSC pulmonary disease
Treatment of MABSC pulmonary disease is difficult as MABSC species are uniformly resistant to standard anti-tuberculous drugs, in addition to exhibiting variable in vitro susceptibilities to other anti-mycobacterial agents. 1 Amikacin, cefoxitin, glycylcyclines (e.g. tigecycline) and the newer macrolides (azithromycin, clarithromycin) often have in vitro activity against MABSC. A recent study has shown inducible macrolide resistance of MABSC due to expression of the erm(4:l) gene, 15 which may explain the failure of macrolide-based therapy despite infecting strains demonstrating in vitro clarithromycin susceptibility 15
The ATS guidelines recommend combination therapy involving parenteral treatment with amikacin and cefoxitin or imipenem for two to four months, in addition to prolonged oral treatment with susceptible drugs. Such therapy has obvious limitations, particularly the inconvenience of long-term intravenous therapy and the toxicity profile of the antibiotics (nephrotoxicity, ototoxicity and cytopenias).
Curative treatment for MABSC pulmonary disease in non-CF patients is most likely in the presence of limited lung involvement and a combination of surgical resection and chemotherapy 1 However, thoracic surgery is not usually an option for patients with CF because of multi-lobar involvement. Alternative goals of therapy such as clinical improvement and radiographic regression of infiltrates represent more realistic aims.
To date, several case reports have been published describing the management and outcome of MABSC lung disease in CF patients, but there have not been any large scale treatment trials.16-18 CuUen et al. described the successful eradication of MABSC in a patient with CF using intravenous treatment with amikacin and cefoxitin, alongside oral clarithromycin, initially for six months, but then repeated for a further five months. 16 This was followed by a maintenance regimen comprising alternate-month aerosolized amikacin and oral clarithromycin; and the patient has now had NTM-free sputum cultures for eight years. 17 Brown reported the case of a boy with CF who was first infected with and treated for MAC lung disease, but continued to deteriorate clinically and subsequent sputum cultures isolated MABSC species. 18 Sustained clinical improvement, but no eradication, was noted following aggressive therapy with intravenous cefoxitin, tigecycline and amikacin for three weeks, and subsequent long-term treatment with oral ciprofloxacin, clarithromycin, nebulised amikacin and nebulized meropenem.
Lung transplantation
NTM infection in patients with end-stage CF pulmonary disease can have important implications for lung transplantation. A recent study showed that 19.7% of CF patients referred for transplantation had previously cultured NTM from respiratory cultures. 19 However, the overall prevalence of invasive NTM disease after lung transplantation was 3.4% and this was associated with pre-transplant NTM infection, in particular MABSC pulmonary disease. 19 Gilljam et al. recently reported three patients with CF who developed skin infections and pulmonary disease secondary to MABSC post-transplant and required adjustment of their immunosuppressive therapy as well as prolonged anti-mycobacterial treatment. 20 Several years later, all three patients have stable lung function and no evidence of MABSC infection.
While it is possible to treat NTM disease successfully post-transplant, MABSC species can be associated with life threatening disseminated infection in lung transplant recipients.20,21 It is for this reason that many transplant centres consider active MABSC pulmonary disease to be a contraindication to lung transplant listing in CF patients.
Conclusion
Non-tuberculous mycobacteria are emerging pathogens in CF and there is a paucity of evidence to direct management. Further research is required to (a) improve the identification and speciation of NTM in CF respiratory samples; (b) to understand the pathophysiology of NTM infection within the CF lung; and (c) to develop more effective drug regimen for patients with CF and NTM pulmonary disease.
Footnotes
Acknowledgements
None
