Abstract

Chronic mesenteric ischaemia (CMI) typically causes post-prandial epigastric pain and weight loss, but can present with exclusively exercise-induced pain.
Case history
A 78-year-old man was referred for investigation of longstanding abdominal pain. He presented with a three-year history of severe dull pain in his epigastrium which only manifested on exertion. Originally, he would experience painful symptoms after 300 yards of walking, but this distance had progressively reduced to less than 100 yards. His symptoms would subside once he stopped walking. He had normal bowel habit with no rectal bleeding He had not experienced any weight loss over this time period, nor had he ever suffered any pain on eating. Extensive investigations had failed to reveal a cause for this pain.
His past medical history included ischaemic heart disease, treated with coronary stents, non-insulin-dependent diabetes, hypercholesterolaemia and hypertension. Clinical examination was unremarkable excepting a prominent antalgic gait and a raised BMI. The clinical history, in particular the diminishing ‘claudication’ distance, prompted us to arrange a CT angiogram. This demonstrated a high-grade stenosis of the coeliac artery (CA), in keeping with the reported foregut distribution of pain (Figure 1).
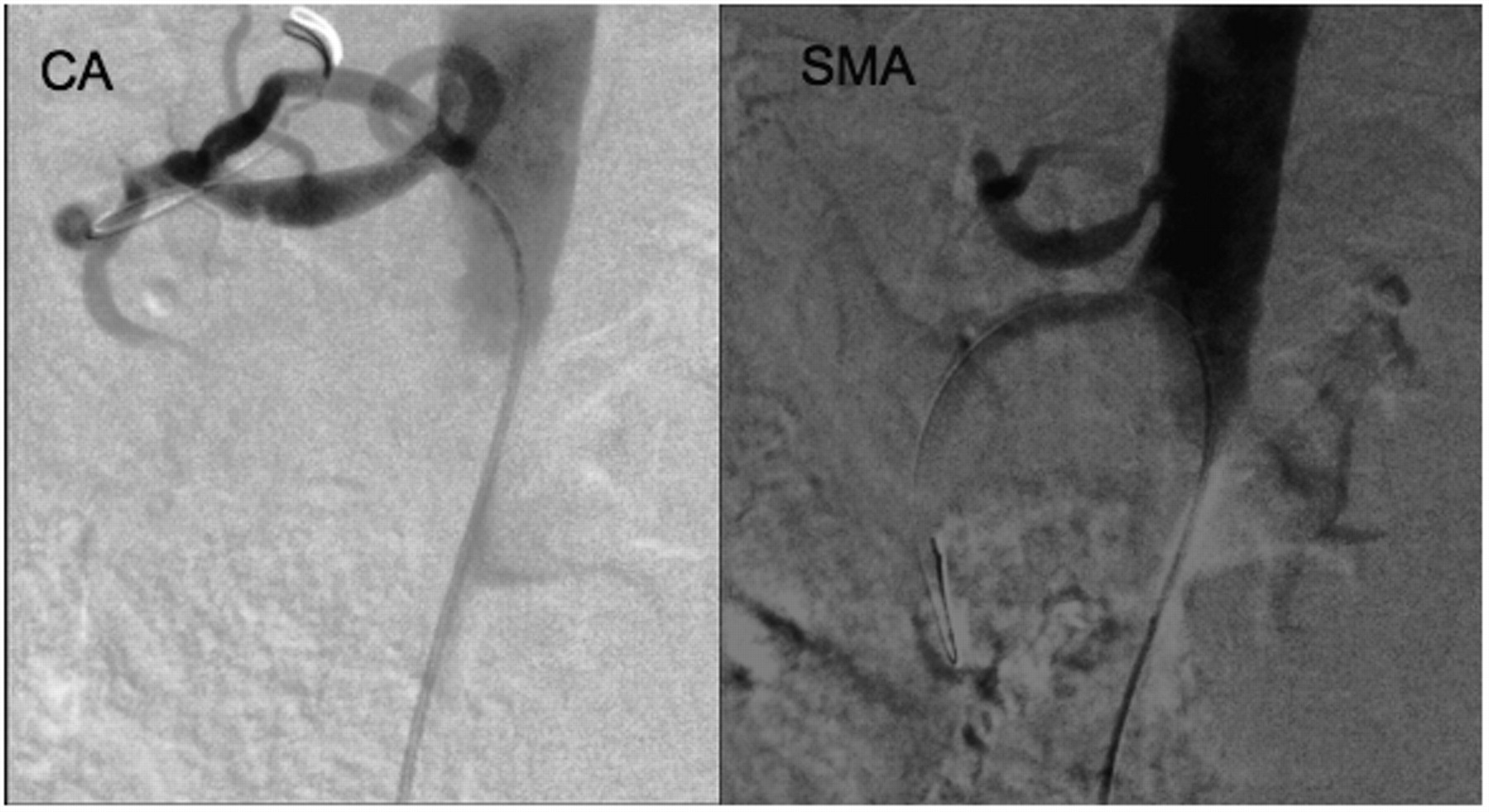
We proceeded to perform an angiogram via right common femoral artery access under ultrasound guidance. This confirmed high-grade stenosis of both the celiac artery and the superior mesenteric artery (SMA). Angioplasty and stenting of both vessels were performed during the same session using one 7 mm × 18 mm Genesis (Cordis) balloon-mounted steel stent for each vessel, with good angiographic results (Figure 2).
At two-week follow-up, the patient reported immediate relief of symptoms following the angioplasty and an uncompromised exercise tolerance. We are awaiting the result of long-term follow-up. The profound symptom relief experienced post-intervention leads us to the conclusion that CMI was indeed responsible for his symptoms.
Discussion
The incidence of chronic splanchnic stenosis in western populations is relatively high, with 0.5–15% of people having >50% narrowing of more than one splanchnic artery. 1 One study showed that out of a cohort of 870 patients over the age of 65 years, 62% could be shown to exhibit >70% stenosis of a mesenteric vessel. 2 Nonetheless, the diagnosis of symptomatic stenosis, or chronic mesenteric ischaemia, is infrequent. This is thought to be due to the abundant collateral circulation that characterizes the splanchnic tree, 3 but may also underline a low index of suspicion among clinicians. It has been traditionally held that stenotic disease of the splanchnic tree only manifests symptomatically when at least two of the mesenteric vessels are affected, however, there is mounting evidence that symptomatic single vessel disease is a genuine entity. 4
Patients with CMI have been known to experience abdominal pain on exertion in addition
to their more classical symptoms of post-prandial epigastric pain and weight loss. In a
cohort of107 patients with CMI, Mensink et al. report an 86% prevalence
of post-prandial pain and a 78% prevalence of weight loss, while 43% of the cohort also
reported exercise-induced pain.
5
CT image showing stenotic lesions of the coeliac artery (CA) and superior
mesenteric artery (SMA) Images showing increased patency of the coeliac artery (CA) and superior
mesenteric artery (SMA) following successful stent deployment across the
stenotic lesions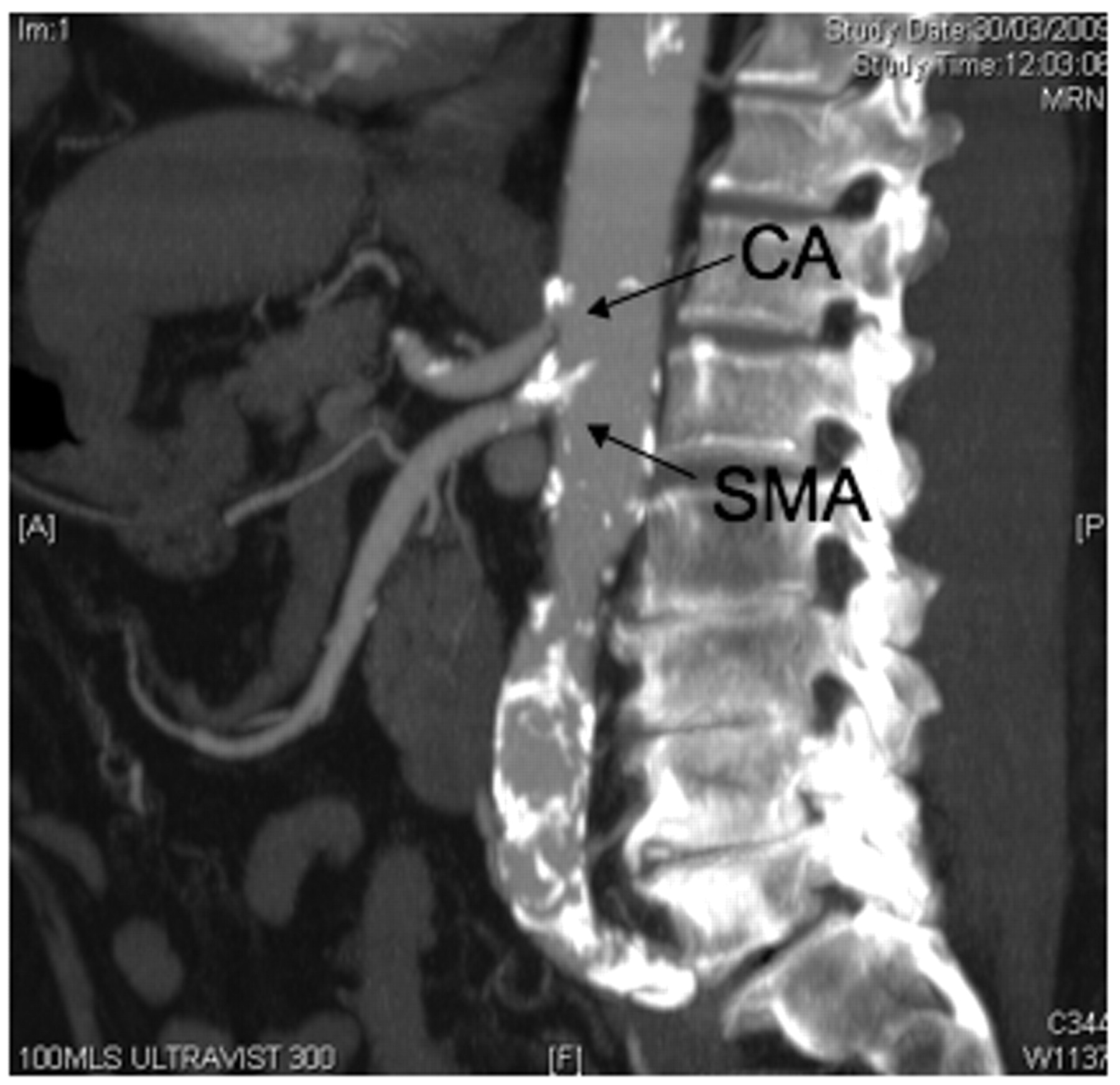
Gastric ischaemia during exercise is a well-established pathophysiological phenomenon which is utilized in gastric exercise tonometry (GET). This method measures gastric mucosal PCO2 in response to exercise as a marker of ischaemia and has been validated for use in the diagnosis of mesenteric ischaemia. 6 GET has been used to show that gastric ischaemia can even occur in healthy subjects during heavy exercise. Furthermore, there is anecdotal evidence of young athletes experiencing ischaemic symptoms while running, although this has frequently been attributed to compression of the coeliac axis by an aberrant median arcuate ligament – median arcuate ligament syndrome.
Various techniques are employed for open repair of mesenteric vessel stenosis, usually involving either a form of bypass procedure or endarterectomy. 1, 7 The last three decades have also seen the introduction and development of endovascular techniques to treat mesenteric artery stenosis. The optimum management of CMI is a matter of much debate. Currently at our centre, patients with significant co-morbidities that would render open repair hazardous are being recommended angioplasty and stenting. A review of the literature published by Aburhama estimates that endovascular intervention offers an 84% chance of early pain relief, with a 71% chance of long-term pain relief. Vessel patency averaged 63% at 25.9 months. 8 Similarly, Atkins et al. report a two-year clinical patency of 83% in patients undergoing open repair and 61% in patients undergoing endovascular repair. 9 A more recent study shows that both treatment options effectively improve symptoms but symptomatic re-stenosis and need for re-intervention were more common in patients treated with endovascular means. 10 It should be noted, however, that the vast majority of these studies review results going as far back as the 1980s, and make no real distinction between older endovascular techniques and modern techniques and stents.
Conclusion
We report a case of CMI presenting as epigastric pain on physical exertion without any commonly associated symptoms. To our knowledge this is the first such case to be discussed in the literature. While mesenteric artery stenosis is common in the population, CMI is rare. This may be partly attributable to a low index of clinical suspicion. We invite clinicians to be vigilant for isolated exercise-induced abdominal pain as a symptom of significant mesenteric artery stenosis.
Footnotes
DECLARATIONS
Footnotes
Acknowledgements
None
