Abstract

An evidential role for mechanisms
Systematic reviews of high quality randomized trials generally count as the ‘best evidence’. 1 However, well-conducted randomized trials are sometimes unavailable, 2, 3, unfeasible, 4 unethical 5 or unnecessary. 6, 7 In such cases other forms of evidence must be considered. Many EBM proponents accept mechanistic reasoning (‘pathophysiologic rationale’) for generalizability, 1, 8 hypothesis generation, 9 ruling out implausible hypotheses, 10,11 11 and for supporting efficacy in the absence of other ‘stronger’ forms of evidence. Yet because mechanistic reasoning has often led us astray, 12, 13 most EBM proponents are justifiably sceptical about using mechanistic reasoning as evidence for efficacy.
We suggest that the scepticism about the value of mechanistic reasoning should not extend to high quality mechanistic reasoning. Just as poor quality randomized trials (that are unblinded, 14, 15, 16 underpowered or biased, 17 that employ unconcealed allocation, 15, 16 or otherwise biased) will not provide high quality evidence for efficacy, so poor quality mechanistic reasoning will be unreliable. In this theoretical exploration we suggest that mechanistic reasoning involving a not incomplete inferential chain and that takes potential complexity into account can and should be used as evidence of efficacy. We support our rules for mechanistic evidence with three examples.
Comparative clinical studies and mechanistic reasoning
Comparative clinical studies (either randomized trials or observational studies)
contrast an experimental therapy with a comparator, and provide direct evidence of a
relationship between the intervention and the clinically relevant outcome. For example,
a randomized trial of antiarrhythmic drugs versus placebo suggested that the drugs
unexpectedly increased mortality by 3.3%.
19
This conclusion did not rely on an explanation of how they did so – that
remained a ‘black box’ ( The ‘black box’ in a comparative clinical study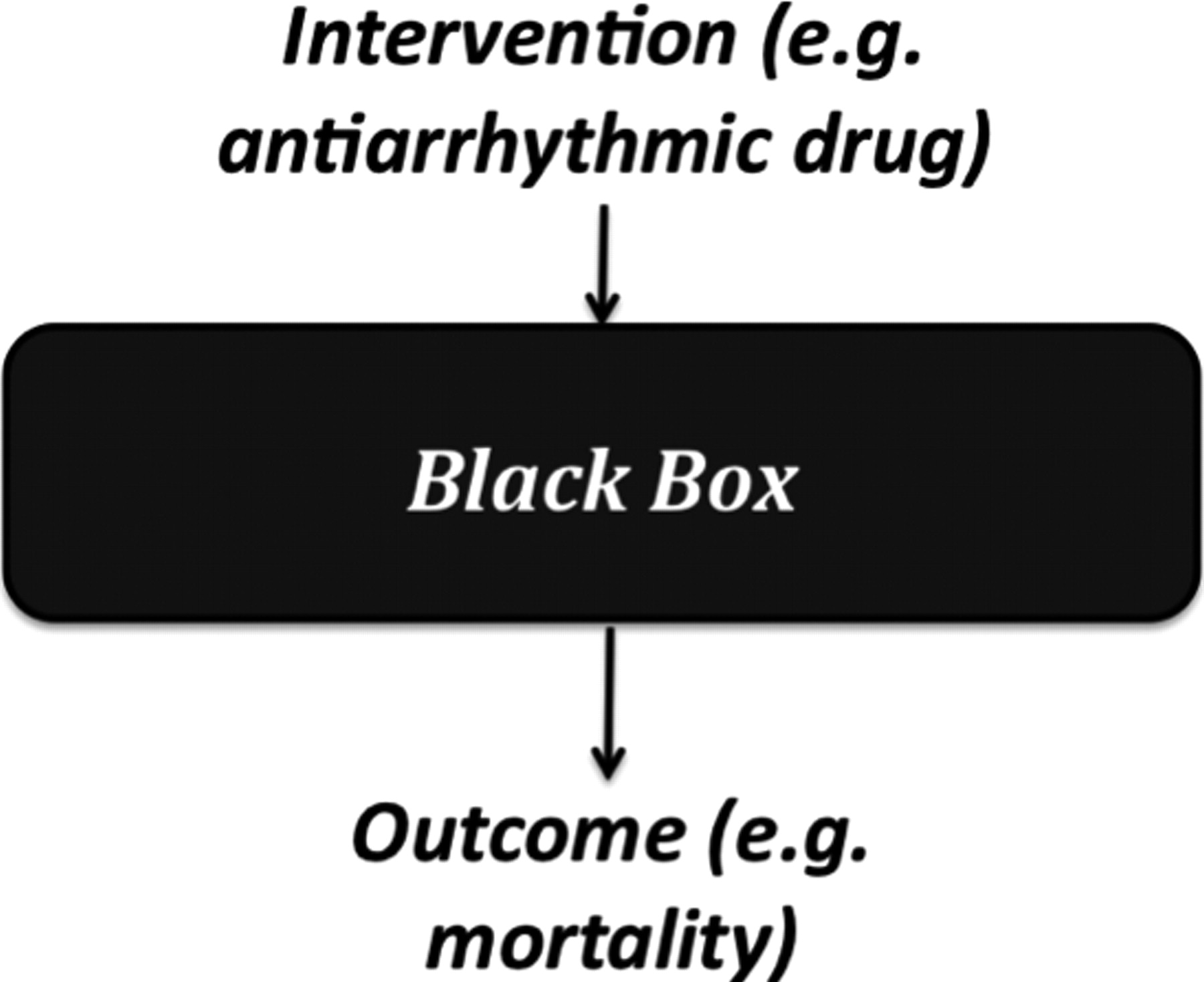
Mechanistic reasoning involves looking inside the ‘black box’, and relies on knowledge of the underlying mechanisms to predict what the relevant effect of a therapy will be. For example, it was known that myocardial infarction damages cardiac muscle and conducting tissues, leaving the heart susceptible to arrhythmias. One type of arrhythmia, ventricular extra beats (VEBs) can degenerate into ventricular tachycardia or fibrillation, followed by death in the absence of electric shock. Large-scale epidemiological studies suggested that 25–50% of sudden cardiac deaths were associated with arrhythmias. 20 Based on this knowledge of the underlying mechanisms, it seemed rational to assume that reducing VEBs would reduce mortality. As a result, many drugs were designed and subsequently prescribed to regulate VEBs.
Mechanisms and mechanistic reasoning
Before describing the problems with mechanisms as evidence, it is useful to distinguish between mechanisms and mechanistic reasoning.
Mechanisms are arrangements of parts/features that (allegedly) ensure a stable relationship between ‘inputs’ and ‘outputs’.
The heart (as a pump), the brain (as a ‘control centre’), and the liver (as a detoxifying agent, among other things) are all mechanisms in this sense. Previous terms have included ‘apriority’ 21 and ‘theory’, 22 but the term ‘mechanisms’ has gained purchase in the philosophical literature, 23 and is commonly used in scientific discussions.
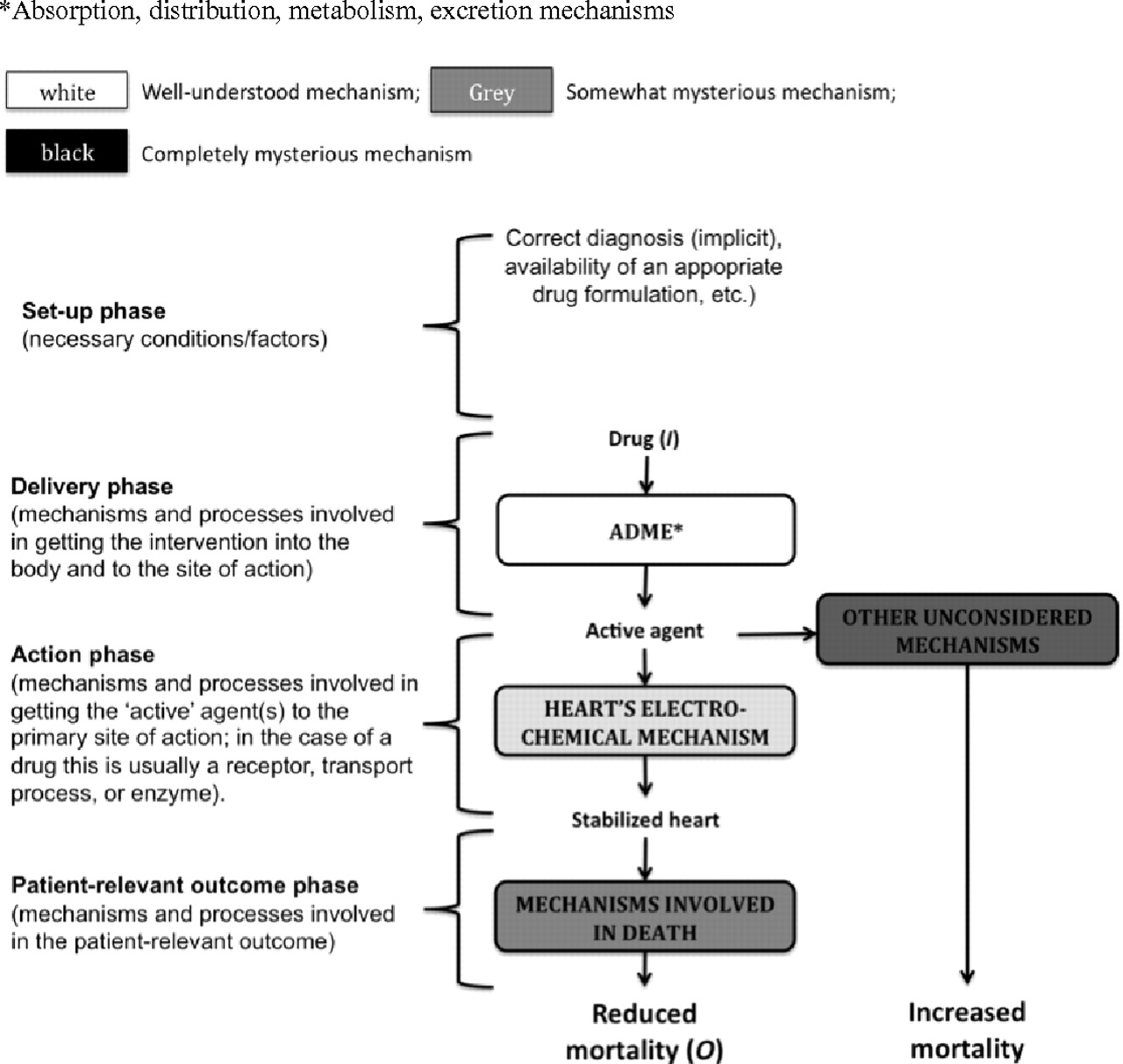
However, described mechanisms do not amount to evidence. Our knowledge of pathology and physiology is rarely sufficiently complete to infer precisely how an intervention will affect mortality or morbidity. For example, the proposed mechanism predicting that antiarrhythmic drugs would reduce mortality in patients with asymptomatic cardiac arrhythmias after myocardial infarction was mistaken (Figure 2). Although the drugs reduced VEBs they also had a proarrhythmic effect in some patients; moreover, the drugs might have affected other mechanisms that, in turn, increased mortality. For mechanisms to be evidential, we must make an inference from (alleged) knowledge of the relevant mechanisms to claims about treatment effects.
Mechanistic reasoning is the inference from mechanisms to claims that an intervention produced a patient-relevant outcome. Such reasoning will involve an inferential chain linking the intervention (such as antiarrhythmic drugs) with the outcome (such as mortality).
For example, antiarrhythmic drugs are orally administered, then delivered to the site of absorption in the gut by a combination of pharmaceutical mechanisms (related to the drug formulation) and physiological mechanisms (such as swallowing and gastric emptying). Next, the drugs are absorbed into the bloodstream, and they (or their metabolites) eventually reach their pharmacological targets in the heart via metabolizing, circulatory and binding mechanisms. Then antiarrhythmic drugs reduce the frequency of VEBs by modifying the heart's conducting mechanism. Finally, a reduction in VEBs (it was supposed) reduces the risk of sudden death (Figure 2).
It is almost always possible to describe a mechanism at a more detailed level. For instance, we might have included cardiac molecular mechanisms (e.g. actions on potassium channels) in our description of antiarrhythmic drug action. The essential feature of mechanistic reasoning as evidence is that it involves a coherent inferential chain linking the intervention with the patient-relevant outcome.
A framework for analysing mechanistic claims
We propose to analyse mechanistic reasoning in a spectrum involving four often
overlapping phases
24
that characterize levels of mechanistic functioning (Table 1):
Phase I – Set-up: In order for any mechanism to be
‘activated’, various background, or ‘set-up’ factors and conditions must
obtain. These include correct diagnosis, the availability of a suitable
formulation (of a drug), the successful administration of anaesthesia (for
surgery), the use of comfortable clothing (for physiotherapy), or the
establishment of a quiet, calm environment (for psychological therapy);
Phase II – Delivery: Various mechanisms are involved in
delivering the intervention. For the ‘pharmaceutical’ and ‘pharmacokinetic’
phases of orally-administered drugs these mechanisms include swallowing,
gastric emptying, and absorptive and distributory mechanisms. For a surgical
intervention such as a hip replacement, the relevant mechanisms might include
cutting tissues and tying blood vessels, in order to access the hip-bone. For a
psychological intervention such as cognitive behavioural therapy, they might
include introductions, history-taking, and other preliminaries, and all the
mechanisms required for speaking, listening and thinking;
Phase III – Action: Most therapies have a particular site of
action. For antiarrhythmic drug therapy this (the ‘pharmacodynamic’ phase)
would be the pharmacological effect on cardiac mechanisms. In surgery it would
be repair or re-building of the relevant body part (such as replacing a hip).
In psychological therapy it might involve providing insight (psychoanalysis) or
suggesting a new behavioural pattern (Cognitive Behavioural Therapy);
Phase IV – Outcome: Whether rapid or delayed, the action of
the intervention should produce a change in patient-relevant outcomes. Whenever
the intended therapeutic effect is to reduce mortality, the relevant mechanism
would be established by whatever definition of death is current (such as
irreversible cessation of circulatory and respiratory functions, or
irreversible cessation of all functions of the entire brain).
25,
26
An example of poor mechanistic reasoning; the original hypothesis
proposed that antiarrhythmic drugs would reduce mortality (middle
column); in fact they increased it (right hand column) Different phases of mechanisms ADME = absorption, distribution, metabolism and excretion
mechanisms
First problem with mechanistic reasoning: ‘empty’ and ‘partial’ mechanisms
In the spirit of Fisher's hypothesis tests 27 and Popper's falsification, 28 we propose that mechanistic reasoning should be ranked according to the extent to which it overcomes obvious flaws (Table 2).
Some mechanistic reasoning is flawed because the mechanisms on which it relies are ‘empty’ (they have little evidential basis). Many medical therapies, including leeching, blood-letting, cocaine as a non-addictive panacea, 29 and psychosurgery, 30 were based on assumptions about mechanisms that lacked an empirical basis. Dr Spock's advice to put babies to sleep prone in order to reduce the risk of Sudden Infant Death Syndrome (SIDS) was also empty. His reasoning, ‘if [an infant] vomits, he's more likely to choke on the vomitus’, 31 was seductive but unsupported by evidence. Even if choking on vomitus caused SIDS, Dr Spock's advice was not evidence-based. Healthy babies, unlike drunk or drugged adults, are skilled at swallowing and spitting, and well-conducted comparative clinical studies 32, 33 showed that putting babies to sleep on their stomachs increased the risk of SIDS. It should hardly be surprising that inferences from ‘empty’ mechanisms will not provide reliable evicence for efficacy.
The problem with mechanistic reasoning of this kind extends to ‘partial mechanisms’ that have some obvious gaps in the inferential chain linking the intervention to the clinically relevant outcome. Ironically, mechanistic reasoning based on partial mechanisms often causes more harm than reasoning based on empty mechanisms. A partial empirical basis for a mechanism can lend an air of authority, which in turn leads to greater use of a harmful treatment. For example, the mechanisms linking antiarrhythmic drugs with a reduced risk of arrhythmias was partially supported by strong evidence, but the link between VEBs and mortality was merely an association. Worldwide it has been estimated that antiarrhythmic drugs killed more people than were killed in action during the whole of the Vietnam War. 34
Surrogates (such as a reduction in VEBs) for the desired outcome (such as mortality) are common examples of mechanistic reasoning based on ‘partial’ mechanisms. The evidence linking the surrogate and the clinically relevant outcome is often lacking. 35, 36, 37
Problems with mechanistic reasoning
Second problem with mechanistic reasoning: the probabilistic and complex nature of mechanisms
While it is generally accepted that most associations are probabilistic (nobody believes
that all smokers develop lung cancer), the implication of the probabilistic and complex
nature of mechanisms often goes unacknowledged. For example, antiarrhythmic drugs did
not always suppress VEBs – they worked in about 90% of patients The probabilistic nature of mechanistic reasoning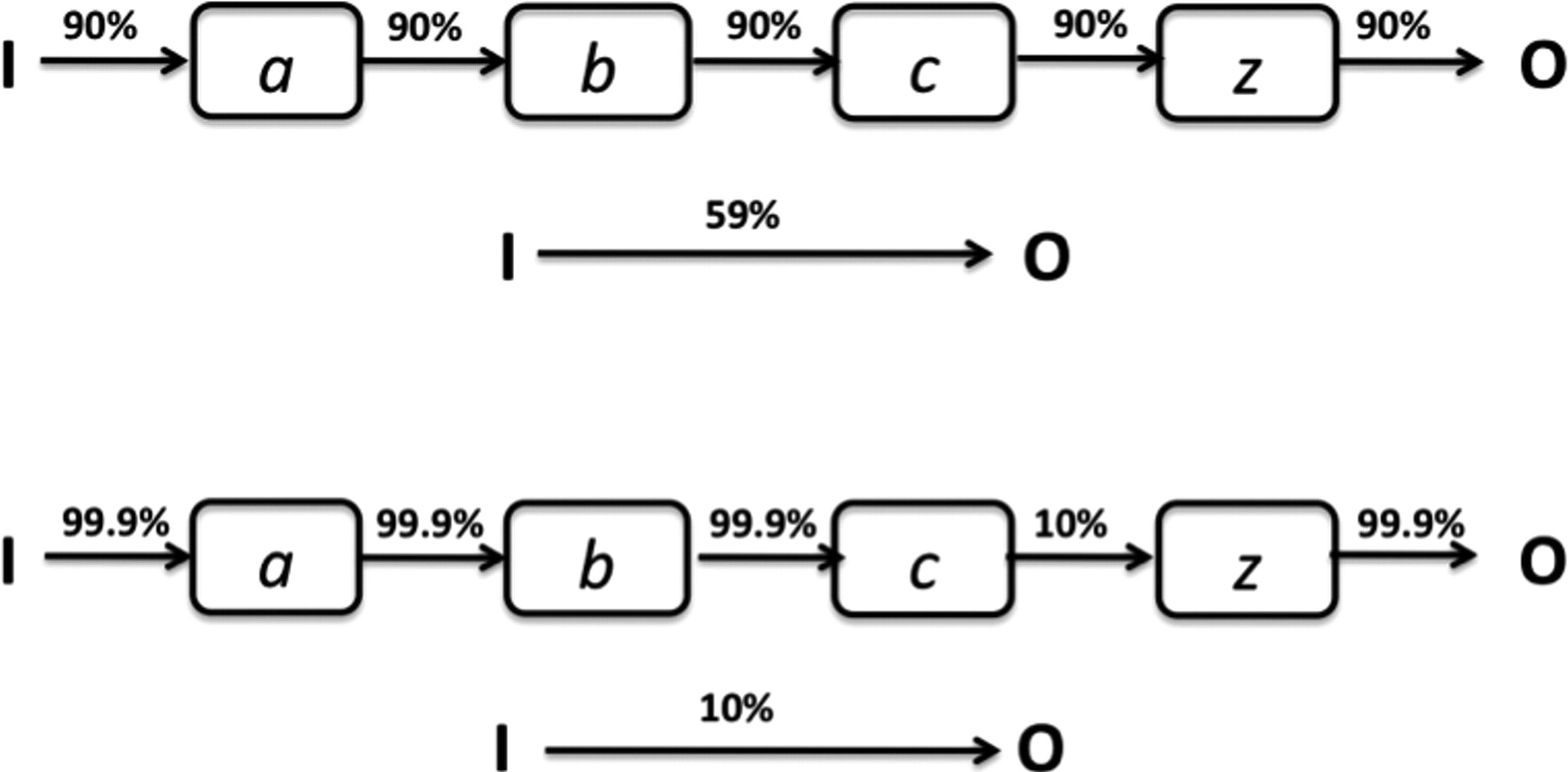
But even if the failure to assume independence makes it difficult to estimate the precise effect size, it might seem reasonable to infer some association, given complete mechanisms. In fact, this assumption is unwarranted. Even if one causal pathway between the intervention and a particular outcome (via the correctly identified mechanisms) is identified, it remains possible that the intervention (or some other component of the mechanisms) instigates another series of events that negates the impact of the outcome of the initial causal pathway (Figure 2). A rare but illustrative type of complexity involves a ‘paradoxical’ response, whereby one outcome occurs in some and the opposite outcome in others. 38, 39 For example, there was a proarrhythmic effect of antiarrhythmic drugs in about 7% of patients. 34
Paradoxical responses and unexpected adverse effects are not the only type of complexity. The same phenomenon can be produced by many different causes. Hypertension, depression, cancers, and many other ailments have more than one cause, and most (if not all) medical interventions are far more complex than is generally assumed. Viagra therapy, for example, involves not only the pharmacological action of sildenafil, but also, among other things, the potentially relevant effects of tablet excipients (e.g. bulking agents), the liquid with which the tablet is swallowed, and the patient's beliefs associated with Viagra. 14, 40, 41 These further treatments can, and sometimes do, affect further relevant mechanisms.
The complexity of both individual mechanisms and of the interactions between the various mechanisms involved in any treatment makes reasoning from knowledge of what happens via (some of the) mechanisms under intervention to a prediction of a clinically relevant outcome highly uncertain.
To recap, mechanisms are never completely understood, and they are all potentially
complex in unsuspected ways. But the problem with mechanistic reasoning must be
contextualized. For one, incomplete knowledge is the norm in science: even the best
randomized trial, for example, could have some potentially confounding difference
between comparison groups. Moreover, it is unreasonable to assume that all mechanistic
reasoning suffers from the problems described above to the same degree Evidence-based mechanistic reasoning, without obvious missing links, supporting
the effectiveness of an accelerated hepatitis B immunization regimen*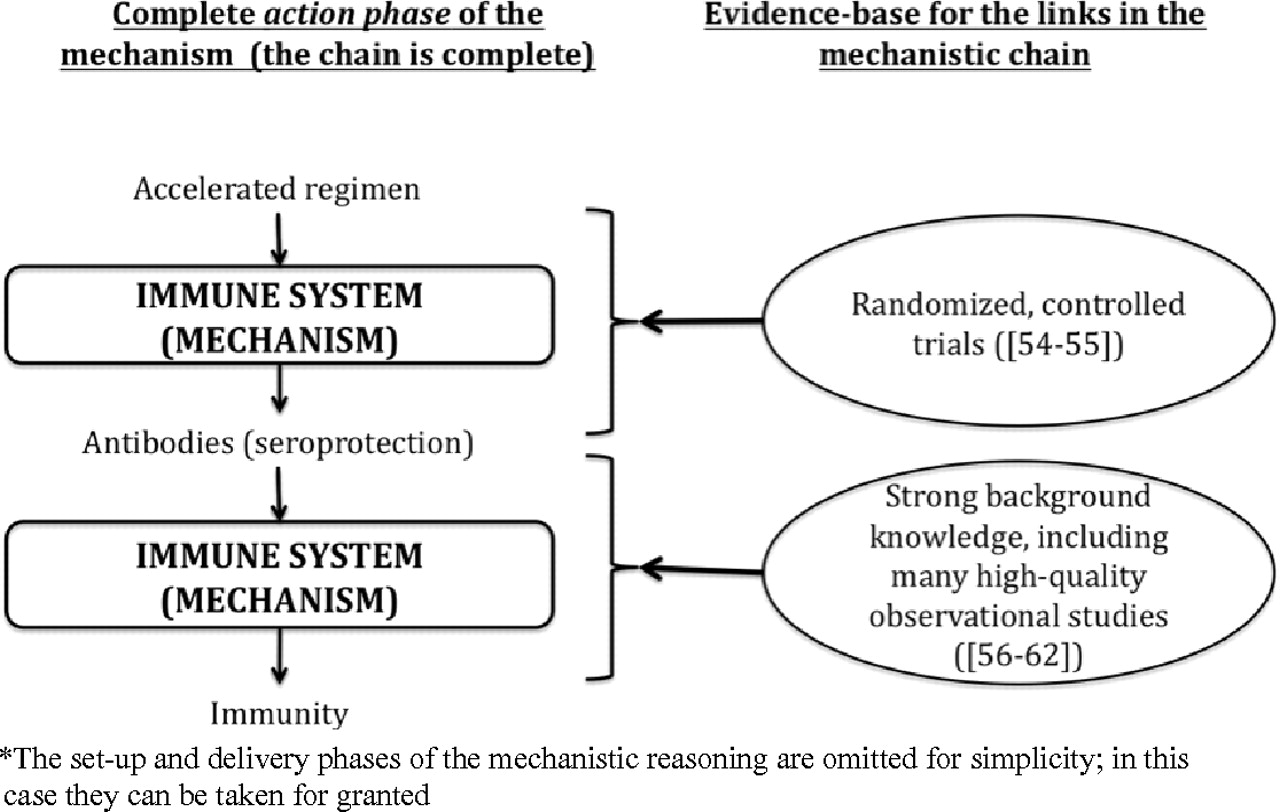
A proposal for redemption: mechanistic reasoning based on mechanisms without obvious gaps is useful evidence
We propose that whenever mechanistic reasoning is used, (a) the links in the mechanistic
chain should be made explicit; and (b) the evidence for the links should be provided
(Figure 4). The quality of mechanistic
reasoning can be evaluated according to the extent to which it overcomes the problems
listed above, and can, therefore, be rated according to the extent to which it satisfies
the following two factors: The knowledge of mechanisms upon which the mechanistic reasoning is based, is
not incomplete. The input–output relationship for each ‘link’ in the
mechanistic chain is evidence-based (for example, based on randomized
trials); The probabilistic and complex nature of the mechanisms are explicitly taken
into account when inferring from the mechanism to any claims that an
intervention has a patient-relevant benefit.
Examples of ‘high quality’ mechanistic reasoning
The following are examples of what we believe to be ‘high-quality’ mechanistic reasoning.
Example 1: Accelerated hepatitis B immunization
Conventional hepatitis B immunization schedules involve injections at 0, 1, and 6 months. This is inconvenient for travellers who have to go to a hepatitis B virus endemic area at short notice. An accelerated regimen of injections at 0, 10, and 21 days, has been studied in randomized trials and shown to result in the same high seroproduction rates as the regular regimen. 42, 43 Similar seroproduction rates indicated that the accelerated regimen produced the same immunity as the longer regimen. The simplified mechanism here is: accelerated regimen → seroprotection → immunity (Figure 4). The trial provided strong evidence for immunization → seroprotection (with no short-term paradoxical or serious adverse events), while strong background evidence links seroprotection with immunity: it has been known for several decades that hepatitis B is caused by the hepatitis B virus (HBV), 44 that the antibodies in the vaccine neutralize the virus, 45, 46, 47 and that seroprotection is related to immunity. 48, 49, 50 In this example our knowledge of the action of vaccines permits us to infer that even if the therapy is delivered differently, the effect is likely to remain the same. In this case there are no missing links in the mechanistic chain, and no studies have revealed unexpected paradoxical or harmful effects (Figure 4).
Concerns might still arise, of course, about the duration of the seroprotection achieved with an accelerated regimen, and even about whether seroprotection is in fact the cause of immunity. However, in the absence of randomized trials linking the accelerated immunization regimen with immunity, it is reasonable to use the accelerated regimen for people who are travelling to a hepatitis B endemic area at short notice, based on the mechanistic evidence.
Example 2: Obstruction by a goitre
Large nodular goitres obstruct the airway and impair respiratory function. At the same time, there is strong evidence that radiotherapy shrinks a goitre (and does not cause any serious adverse effects), 51 although radiotherapy can also cause short-term thyroid swelling. 52 Mechanistic reasoning allows us to conclude that radiotherapy will improve respiratory function in the longer term. One might even question whether the trial that tested the hypothesis was justified, given the quality of the available mechanistic reasoning.
Example 3: Reminder packaging
There is evidence for a link between blood glucose concentration, blood pressure control, and the prevention of diabetic complications. 53, 54 However, controlling the glucose concentration and blood pressure often results in complex therapy involving 10–15 tablets per day. Adherence to such therapy is inversely proportional to the complexity of the administration regimen 55 and special packaging can improve patient adherence. 56 If we assume that the medication itself causes the reduction in blood pressure, we can appeal to mechanistic reasoning to infer that special packaging will help prevent diabetic complications by increasing adherence. Indeed a randomized trial showed that calendar blister packs significantly improved glucose concentrations and blood pressure. 57 Although the direction of the effect was unsurprising, the size of the effect of calendar packaging could not have been predicted from mechanistic reasoning – that is not its purpose.
Using mechanistic reasoning to deny that a treatment has an effect
Low quality mechanistic reasoning is equally unreliable as evidence that an intervention is ineffective. The introduction of many useful treatments, such as antisepsis 58 and antibiotics for peptic ulcers 59 was delayed because of failure to consider mechanisms properly. 4 Warren and Marshall, for example, were ridiculed for presenting empirical evidence that Helicobacter pylori caused peptic ulceration. Sceptics wrongly inferred (from low quality mechanistic reasoning) that bacteria could not live in the hostile environment of the stomach. 59
Conclusion
Our modest aims in this paper were to point out that not all mechanistic reasoning was created equal, and set out a preliminary set of standards that mechanistic reasoning can and should be held up to when used to support claims about treatment effects. Whereas comparative clinical studies have been held up to explicit standards, 60 according to the strength of evidential support they provide, all forms of mechanistic reasoning as evidence for efficacy have hitherto been lumped together and generally denigrated. Whenever mechanistic reasoning is used to justify a therapeutic intervention, the stages and chain of reasoning should be shown, accompanied by the evidence that supports each link in the chain (Figure 4). Mechanistic reasoning based on empty or partial mechanisms should be disregarded. Yet, our analysis and examples suggest that high quality mechanistic reasoning involving an evidentially justified chain without any obvious missing links and that takes potential complexities into account can and should be used to support hypotheses of therapeutic efficacy. Further research will determine how high quality mechanistic reasoning fits into current evidence ranking schemes. 60, 61
Footnotes
DECLARATIONS
Footnotes
Acknowledgements
The authors are grateful to NancyCartwright, Sir Iain Chalmers, Lindley Darden, Alex Broadbent, the GRADE Working Group, Stuart Glennan, Sian Harrison, and Rogery Kerry for comments on earlier drafts of this paper and/or discussions of the role of mechanisms. JH also attended the Workshop on Biological Mechanisms in Evidence-Based Medicine at Johns Hopkins Bloomberg School of Public Health in Baltimore, MD (30 November 2009). This paper was revised while JH was a recipient of an MRC/ESRC Interdisciplinary Postdoctoral Fellowship (G0800055)
