Abstract

Respiratory exacerbations of cystic fibrosis (CF) lung disease have been associated with increased mortality, hospitalization, antibiotic administration, health costs and diminished quality of life. Preventive strategies such as vaccination against common respiratory pathogens have been developed to reduce the frequency of pulmonary exacerbations. 1
DECLARATIONS
None declared
None
Not applicable
MJ
Both authors contributed equally
Children with CF are vaccinated as per the normal childhood vaccination schedule. They are equally at risk from the common childhood diseases but are particularly susceptible to respiratory complications. Young children with CF may miss out on the normal immunizations schedule through illness and general practitioners need to be aware that cough is not a reason to withhold or delay vaccination. Adults may be more susceptible to infections despite being vaccinated in infancy as immunity reduces over time. The extent of damage caused by vaccine preventable diseases in CF is not known. 2 The potential benefit of supplemental vaccination strategies is discussed below.
Pneumococcal vaccination
The incidence of invasive pneumococcal disease is highest in young children and the introduction of universal infant vaccination with the seven–valent conjugate vaccine in 2007 protects children with CF as others. The polysaccharide-protein conjugate vaccine triggers a T-cell dependent immune response with production of high level of antibodies and generation of memory cells. It is highly protective against invasive disease. However more than 90 serotypes of Streptococcus pneumoniae (S. pneumoniae) have been identified and there is evidence that invasive disease from non-vaccine sero-types has increased since universal vaccination started (Figure 1) although in part this may be related to increased surveillance.
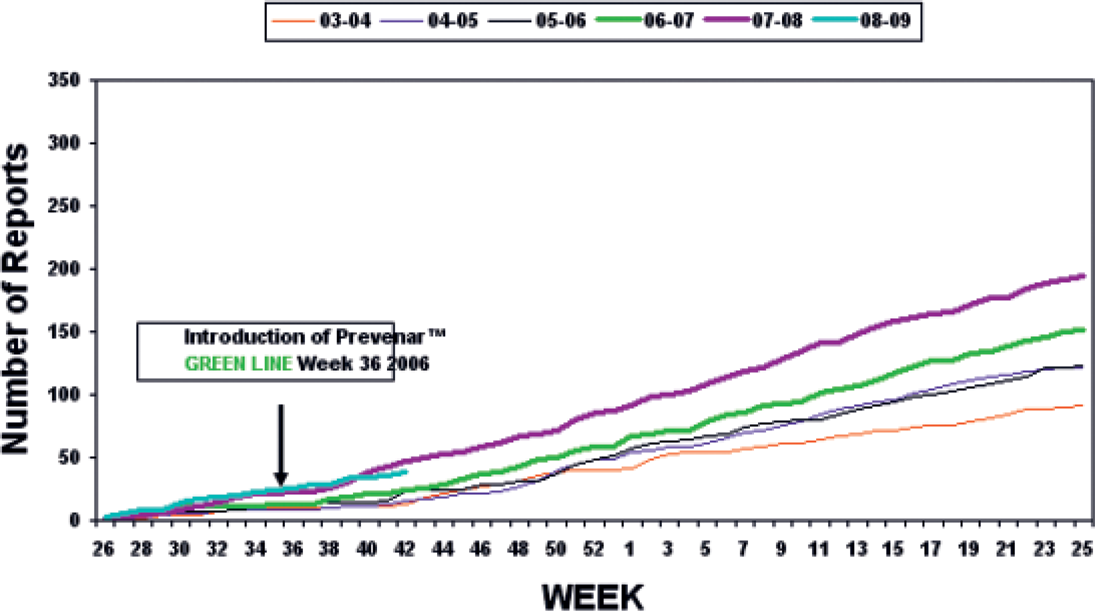
Cumulative weekly reports of invasive pneumococcal disease due to serotypes not in Prevenar July-June 2003-October 2008
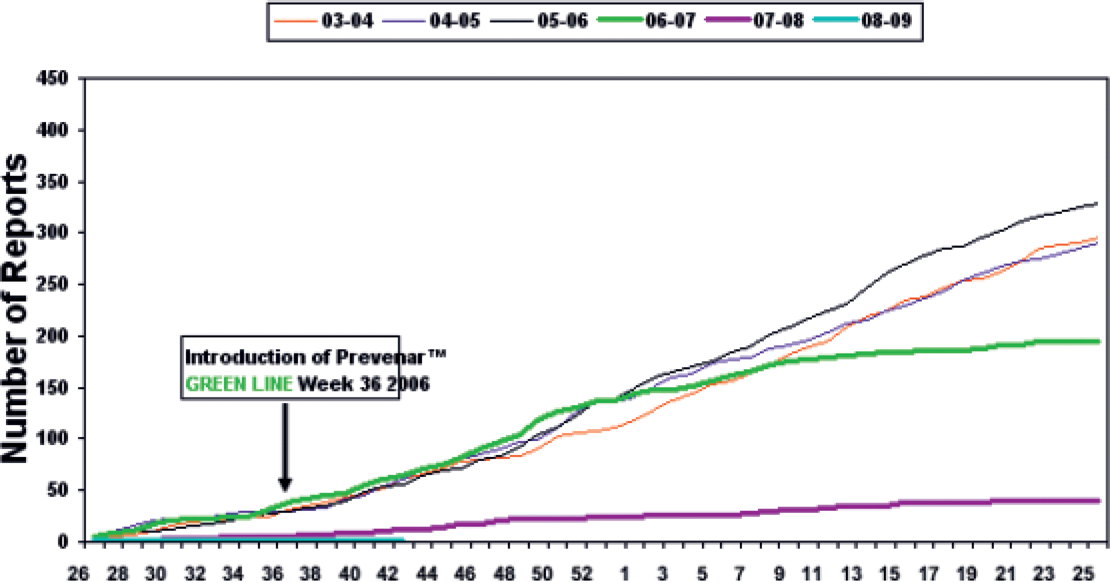
Cumulative weekly reports of invasive pneumococcal disease due to serotypes in Prevenar July-June 2003-October 2008
A study of a 13–valent pneumococcal conjugate vaccine given as part of the UK infant vaccine schedule has just been completed. 3 It was shown to be immunogenic and well tolerated. Vaccination at 2 and 4 months resulted in 78–96% of vaccinees being protected for the six additional serotypes with 91% of recipients achieving protection against serotype 19A; a predominant invasive serotype in the USA and 96% protection against serotype 1; incriminated in complicated pneumonia and empyema in the UK.
S. pneumoniae is commonly isolated from the sputum of patients with CF and its frequency may be underestimated as it can be difficult to isolate where there is heavy growth of other organisms. However nasopharyngeal carriage of S. pneumonia is commonplace in normal children and adults. A recent study showed that 63% of children < 5 years; 38% of 5–17-year-olds and 14% of adults carried pneumococcus in the nasopharynx. 4 As always in young children where sputum is not available it is difficult to assess the significance of finding S. pneumoniae on a cough swab.
Before infant immunization was available Lahiri and Waltz 5 reported that a majority of CF patients aged 1–19 years had protective levels of pre-immunization pneumococcal antibody. However, a significant proportion (between 17% to 39%, depending on the serotype) did not exhibit adequate levels.
The 23–valent polysaccharide vaccine (Pneumovax) covers approximately 90% of all the serotypes which cause invasive disease and vaccination results in antibody formation against around three-quarters of the antigens present in the vaccine. 1 However the evidence of efficacy of this vaccine is insecure and being reconsidered. 6
As polysaccharide vaccines do not lead to the generation of memory cells it was anticipated that re-vaccination would be needed to maintain protective immunity. However there is only a weak booster effect after re-immunization and some preliminary evidence that previous immunization with the polysaccharide vaccine prevents a protective response to the conjugate vaccine.
The immune response to polysaccharide vaccines is T lymphocyte independent and the immature immune system is not able to mount a response. It has previously been recommended therefore that all CF patients older than 2 years receive the 23–valent polysaccharide immunization against pneumococcus. 7 The emerging evidence on poor efficacy of the polysaccharide vaccine and potential availability of newer conjugate vaccines with increased serotype coverage means that this advice is likely to change in the near future.
Influenza vaccination
There is evidence that viral infections in CF contribute to pulmonary exacerbation and disease progression with worsening lung function, increased hospitalization rates and that they may predispose to bacterial infections.8–11 Newer viral identification techniques permit further evaluation of the role of viral disease. Wat et al. 12 recently reported a prospective study of 71 patients over 17 months where nasal swabs for virus detection using a nucleic acid sequence based amplification process were obtained whenever there were respiratory symptoms and also at routine review. There were 272 routine samples and 165 taken during times of respiratory symptoms. Viruses were detected in 46% of samples during respiratory exacerbation and 16.9% of routine samples. Only influenza A (8% vs 1.5%), influenza B (7.2% vs 0%) and rhinoviruses (15.9% vs 7.4%) were more common during exacerbations.
A study of frequency and impact of viral infections in adults with CF receiving intravenous antibiotics identified non-bacterial infection in only 5.1% of 3156 courses of treatment in 305 adults. 13 Influenza virus A (2.0%) and B (0.9%) were the most common. Patients with evidence of viral infection did not have a worse short-term clinical outcome.
Pribble et al. 14 showed that infection with influenza A virus led to a higher proportion of CF patients having a significant fall in lung function compared with patients with other non-bacterial infection and the group without non-bacterial infection.
There is some evidence that CF epithelial cells may behave differently when encountering the influenza virus. An in vitro examination of the mucosal host responses to influenza revealed less antiviral and greater inflammatory gene expression responses in CF airway epithelial cells. 15
Influenza viruses have the unique feature of exhibiting antigenic shift and drift allowing them to escape host defence mechanism. The virulence of influenza infection thus changes from year to year and there is evidence of morbidity in all children. Poehling et al. 16 conducted a population-based surveillance in children < 5 years and found that the average annual rate of hospitalization associated with influenza was 0.9 per 1000 children. The estimated burden of outpatient visits was 50 primary care visits and six emergency department visits per 1000 children during the 2002–2003 season and 95 clinic visits and 27 emergency department visits per 1000 children during the 2003–2004 season.
Annual vaccination is considered the best means of protection against influenza infection. The US 2008 recommendations are for universal annual influenza vaccination for children aged 6 months to 18 years but children 6 months– 4 years and those with a chronic pulmonary illness are considered to be at higher risk. The UK advice is for annual vaccination of anyone aged older than 6 months who is considered to be at risk of complications. It is not recommended in those younger than 6 months due to poor immunogenicity. The recommended schedule for children aged 6–35 months is two half doses with a one-month interval. Children aged 3–8 years receive two doses for the first year at least four weeks apart. Subsequently single dose is given annually as in adults. Immunization against influenza A virus has been shown to provoke an adequate antibody response in patients with CF.
A Cochrane review of four randomized trials comparing any influenza vaccine with placebo or with another type of influenza vaccine in CF did not show any clinical benefits in vaccination. 17 However a more recent study compared influenza infection rates between 41 vaccinated and 22 non-vaccinated patients where nasal swabs had been obtained during respiratory exacerbations and at routine visits over a winter season. 18 There was only one influenza virus isolation in the vaccinated group compared to four in the non-vaccinated (p=0.046) confirming the anticipated protective effect.
There have been a number of studies looking at influenza vaccine uptake rates in CF. Data from 518 French CF patients revealed a coverage rate of 85.6% in children and 69.4% in adults but only 44.4% in transplanted patients. 19 In the US a telephone reminder to families where the CF centre did not have documentation of them receiving flu vaccine increased uptake from 83.3% to 95.8%. 20 Oxford data show 82% uptake in children in 2007.
Respiratory syncytial virus (RSV)
There have been a number of small prospective studies of RSV acquisition in CF. In an early study Abman et al. 21 prospectively followed 48 children diagnosed on newborn screening. At a mean follow-up age of 28 months (R5–59) 18 infants had been admitted to hospital a total of 30 times for respiratory distress and RSV identified on seven admissions. They concluded that RSV lower respiratory tract infection was a frequent cause of hospitalization for acute respiratory illness in CF patients during infancy, and was associated with significant morbidity and prolonged hospitalization. In contrast Armstrong 8 followed 80 infants diagnosed by newborn screening. Thirty-one children (39%) were admitted to hospital with respiratory symptoms in the first year of life and a respiratory virus detected in 16 cases (four with concomitant bacteria) with RSV in seven. In the absence of co-infection with bacteria patients with viral infection had a self-limiting and uncomplicated course. Overall 35% of infants admitted to hospital in infancy acquired infection with P. aeruginosa by 50 months of age compared with only 6% of non-admitted patients (RR 5.8; CI 1.9–24) but six out of the seven patients with RSV remained free of P. aeruginosa.
Hiatt 22 studied 22 infants with CF aged <2 years (through 30 winter seasons) and 27 age-matched controls (28 winter seasons). Both controls and CF infants had a mean of 5.3 acute respiratory illnesses per season but CF infants were more likely to develop a lower respiratory tract infection (LRTI) than controls as assessed by a visiting study nurse using a modified respiratory illness score. (OR 4.6; CI 1.3–16.5). Control infants had twice as many viral identifications including RSV 19/28 vs 7/30 p<0.01 but none of the control infants with RSV were admitted compared to three of the CF RSV positive infants. Of interest, three times as many control infants attended day care (31% vs 10%). This group performed infant lung function tests (Vmax FRC) pre and post season and lung function increased in the control infants with no association with either viral infection or LRTI. In contrast there was a fall in lung function in the CF patients who had experienced a LRTI when compared with both the control group and the CF infants who had URTI only (p<0.05). In regression analysis both RSV infection with LRTI and being male with LRTI were associated with a reduction in Vmax FRC.
RSV in older children and adults
Wang 23 examined the effect of respiratory viral infections on pulmonary function in 49 older children and young adults with CF (mean age 13.7 years) in a prospective study over 2 years using 19 siblings as controls. Viral infection was identified by serology. The frequency of proven viral infections was identical 1.67 infections per annum in CF patients vs 1.7 in controls, but respiratory symptoms were reported more often in CF patients 3.7 times per annum vs 1.7 (p <0.001). RSV was responsible for 24/105 infections in CF patients, 12 of which were asymptomatic. The authors found significant correlation between the annual incidence of viral infections and the rate of disease progression over the 2-year period with the 15 patients who had >1.67 infections per annum doing less well in all parameters.
In a more recent study 44 children with CF aged 7–18 years and FEV1 > 45%P were followed prospectively for LRTI through an RSV season. 24 Samples of nasopharyngeal wash secretions and from a throat swab were sent for viral identification with each LRTI and in addition blood for serology was taken at start of study; halfway through the RSV season and at the end of the season. A total of 56 episodes of LRTI and 18 hospitalizations were identified in the 44 children. RSV was found in two NPA samples; Flu Ain one; Paraflu 3 in one and picornavirus in one. Sixteen children were shown to have been infected with RSV by serology; 9/24 aged 7–12 years and 7/20 aged 13–18 years. There were no hospital admissions in the RSV positive patients. When the period in which serology became positive was compared to a non-infected period there appeared to be more LRTI in the RSV positive periods but this was not statistically significant.
LRTI with RSV is accompanied by a neutrophil predominated inflammatory response in the lungs in CF as in other children. BAL results on inflammatory markers with viral infections are similar to those seen with bacterial LRTI in CF. 8 In the laboratory RSV infection has been shown to increase the adherence of P. aeruginosa to epithelial cell mono-layers suggesting that there may exist specific viral bacterial interactions important in CF. 11
A recent study examined the prevalence and clinical impact of serologically defined infection with respiratory viruses in 305 adults receiving 3156 courses of intravenous antibiotics over a 10-year period. 13 RSV was identified on 15 cases (0.5% of antibiotic courses). Evidence of viral infection did not affect clinical course and in particular there was no evidence of increased frequency of P. aeruginosa isolation after a viral infection.
There is considerable interest in prevention of RSV infection and in any drug which may ameliorate disease.
Palivizumab is a humanized monoclonal antibody to RSV. It provides passive immunization and has been shown to decrease hospitalization with RSV disease in certain high-risk populations but does not prevent infection. It is administered in a dose of 15 mg/kg body weight, once a month during the RSV season (maximum of five injections/season).
Giebels 25 retrospectively studied the effect of palivizumab prophylaxis on hospitalization for acute respiratory illness in 75 young children with CF during the first RSV season after diagnosis and used historical controls. During the RSV season 3/35 palivizumab recipients vs 7/40 controls were admitted with a respiratory illness. Virology was performed rarely and RSV identified in 0/3 palivizumab recipients tested and 3/7 controls tested. No other statistically significant differences were found.
McCormick 26 conducted a national questionnaire study of 42 CF centres in the UK to determine the number of infants with CF who would be potential candidates for RSV prophylaxis and to quantify hospitalization rates and mortality from RSV in this cohort. A total of 143 infants with CF aged <1year were identified. Sixteen (11.2%) of the 143 were hospitalized owing to RSV infection but none required PICU admission and none died. Fourteen (9.8%) infants received palivizumab between three CF centres, one was subsequently hospitalized with RSV infection. Two of the three centres used palivizumab annually as they had separate local funding. From this data 22 CF patients would need to be treated to prevent one hospital admission.
A report on data from the Palivizumab Outcomes Registry identified 91 CF children who had received palivizumab off-label between 2000 and 2004 and when followed prospectively none had been admitted with an RSV-positive illness. 27
The next generation of RSV preventative drugs may be more useful in CF. Motavizumab is derived from palivizumab differing by only 13 amino acid residues and is a highly potent humanized monoclonal antibody directed to the RSV Fusion protein. Compared with palivizumab it has 70-fold greater affinity to the F protein and is 20-fold more potent at neutralizing RSV in tissue culture. Experiments in the cotton rat model showed 100-fold decrease in RSV titres in the lung compared with palivizumab and in addition motavizumab prevented RSV replication in the upper respiratory tract. 28
Aphase 3 study of motavizumab vs placebo has been completed in 1410 high risk infants and showed a significantly greater reduction in hospitalizations due to RSV (1.4% vs 8.3%; p<0.001). In addition significantly fewer treated infants had RSV related respiratory infections which required outpatient management (2.8% vs 9.5%; p<0.001). There was no difference in adverse events between groups. In a previous non-inferiority study of motavizumab and palivizumab, in 240 patients motavizumab demonstrated a 26% reduction in RSV-associated hospitalization and a 50% reduction in RSV infection needing outpatient management. A phase 1/2 study demonstrated safety in 136 children who received either motavizumab or palivizumab in a second winter season after having motavizumab in the first year. 29 A New Drug Application for Motavizumab has been filed in the US and a Marketing Authorisation Application for Europe in expected in the first half of 2009. A phase 2 study of Motazivumab for early and late treatment of RSV in children >1 year is underway.
The biggest avoidable risk factor for RSV hospitalization in children is household smoking. Simple measures for the prevention of transmission of viral illnesses in early life should not be forgotten. These include limiting contact with other infants, avoiding shared toys, avoiding older children and adults with URTI and ensuring carers’ hands are washed before contact.
Conclusion
Viral infection is common in CF with rhinovirus predominating. Influenza virus is implicated in respiratory exacerbations and annual influenza immunisation is recommended. RSV infection is universal in young children and CF infants are more likely to be admitted to hospital with RSV infections than the normal population. Palivizumab may be useful in CF but it is costly and over 20 infants would have to be treated to avoid one hospital admission. Newer drugs likely to be on the market in 2–3 years may be more effective.
All children and adults should be protected from invasive pneumococcal infection with a conjugate vaccine. The policy on the use of polysaccharide vaccines is likely to change.
