Abstract

Introduction
A 69-year‐old woman with postoperative complications required prolonged nasogastric feeding. This unfortunately caused gastrointestinal haemorrhage and stricturing oesophagitis with perforation.
Case report
A 69-year‐old woman presented to A&E with a two‐day history of malaise, lower back pain and vomiting. Her abdomen was soft and non‐distended, with mild tenderness in the epigastrium and normal bowel sounds. Her past medical history included previous open cholecystectomy for acute cholecystitis, and a subsequent laparotomy for severe adhesional bowel obstruction with creation of a permanent ileostomy. Following this procedure she developed a massive incisional hernia. She was not known to have oesophagitis or peptic ulcer disease, and was not receiving any form of gastric acid suppression.
She was initially managed conservatively with omeprazole while awaiting upper gastrointestinal endoscopy. Two days into the admission the patient became more unwell, with derangement of her renal function. There was clinical evidence of peritonitis and CT scan revealed extensive intraperitoneal free fluid and air consistent with intestinal perforation. A laparotomy was performed at which a large perforated anterior duodenal ulcer was discovered and oversewn. The procedure was complicated by both the incisional hernia and extensive adhesions. During adhesionolysis, an enterotomy occurred necessitating limited resection and end‐to‐end anastomosis. Her initial postoperative recovery was satisfactory but 14 days after the surgery she deteriorated suddenly. A further laparotomy was performed at which gross faecal contamination of the peritoneal cavity was evident and a small bowel perforation identified, which was resected.
Her subsequent recovery followed a very stormy course. The patient became very depressed, and consequently her oral intake dropped and she required nutritional support. It was felt that feeding via the enteral route would be preferable, and that the risks of bloodstream infection and severe sepsis in a malnourished patient with a heavily contaminated open wound outweighed the benefits of parenteral nutrition. Nasogastric feeding was therefore instituted using a fine‐bore feeding tube. Once this was established her physical condition slowly improved, as evidenced by slow but progressive wound healing. She remained on proton pump inhibitors throughout her hospital admission. Her depression, however, took longer to resolve and nasogastric feeding was continued for approximately three months.
When oral nutrition was finally recommenced, the patient complained of dysphagia and was
able to tolerate only small and infrequent boluses of solid food. Three days later,
melaena was observed from the ileostomy, and her haemoglobin fell from 12.1 g/dL to 8.2
g/dL, associated with haemodynamic instability. An emergency oesophagoduodenoscopy was
performed. This revealed the nasogastric tube in situ ( Views at oesophagoduodenoscopy showing Chest radiograph demonstrating a large right‐sided pneumothorax with associated
intrapleural fluid and pneumonitis, secondary to oesophageal rupture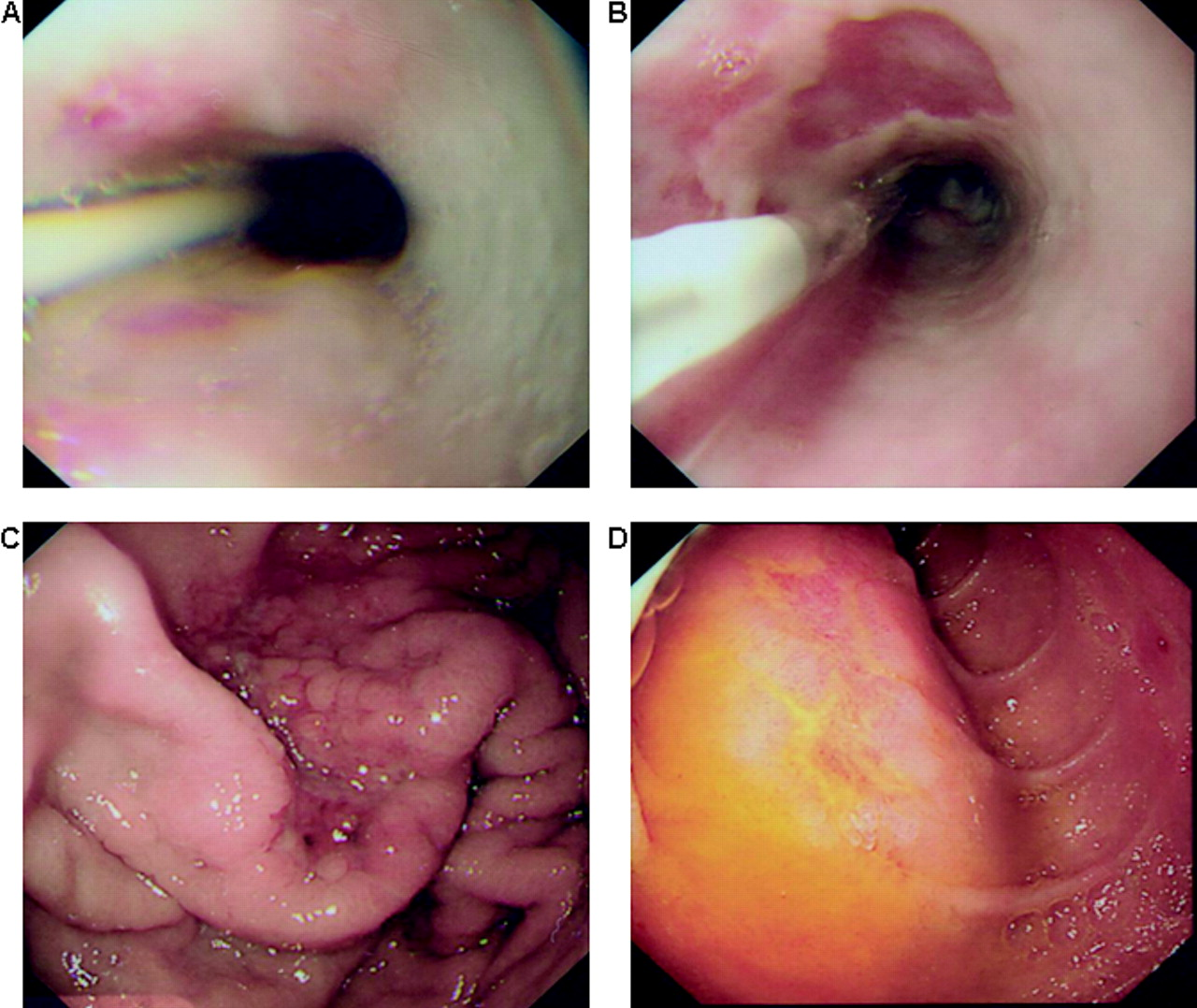
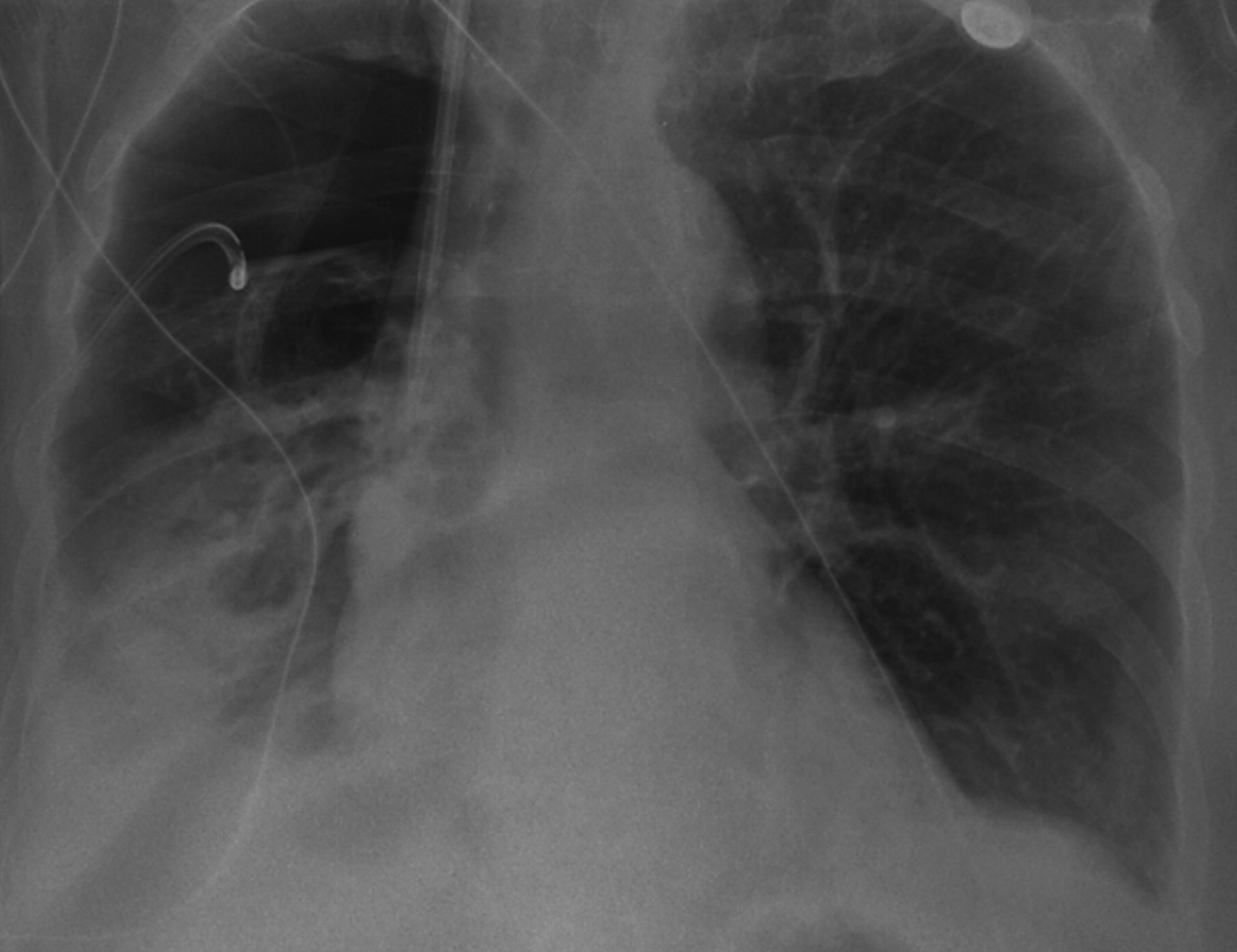
The patient was readmitted three days later following a collapse. On this occurrence, her haemoglobin was stable at 11.5 g/dL and there was no evidence of further haemorrhage. Chest X‐ray revealed a right‐sided pneumothorax in conjunction with obliteration of the right costophrenic angle by fluid (Figure 2). A chest drain was inserted, and the aspirate demonstrated to have pH<5, consistent with gastric contents. On microscopy, the fluid contained pus, no bacteria and scanty candida. A clinical diagnosis of oesophageal perforation was made. She was initiated on conservative treatment, but progressively deteriorated and passed away two days after readmission.
Discussion
Nasogastric tubes for nutritional support have been used for over a century, although their long‐term use has now been largely superseded by percutaneous gastrostomy. It is generally well tolerated but does possess recognized complications, occurring in 0.3–8% of patients. 1 These predominantly occur acutely, usually related to difficulties with tube insertion. Complications can be divided into enteric problems (tube impaction, double‐backing, kinking, obstruction and perforation) and non‐enteric issues caused by tube malplacement. 2 The latter most commonly affect the respiratory tract (in particular bronchial placement), 3 but intravascular 4 and intracranial 5 penetration have also been reported.
Very few studies have examined the effects of long‐term placement of feeding tubes. This is partly because nasogastric feeding is generally a short‐term intervention and only a few studies report use beyond 28 days. 6, 7 It is, however, well recognized that tubes can become rigid over time with exposure to acidic stomach contents and that this is likely to increase the risks of irritation and erosion to the upper gastrointestinal tract. Such inflammatory changes in the lower oesophagus may result in stricturing and haemorrhage, and if severe may produce penetrating ulceration and oesophageal perforation. 8 The latter has been most frequently reported in neonates and infants. 9 Current guidelines therefore recommend that tubes remain in situ for a maximum of seven days for wide‐bore tubes or two months for fine‐bore tubes. 2
Conclusion
This patient presented a particular challenge in view of her protracted need for nutritional support. Her recent upper abdominal surgery and her extensive intra‐abdominal adhesions made her unsuitable for a percutaneous gastrostomy. Furthermore, in the presence of an open and heavily contaminated wound, total parenteral nutrition would have been associated with significant risk. Although many patients will tolerate long‐term fine‐bore nasogastric intubation without incident, this case serves as a reminder that this commonly used nutritional intervention, which so clearly has great clinical benefit, is not without risk.
Footnotes
DECLARATIONS
Footnotes
Acknowledgements
None
