Abstract
Although surgery is the usual management strategy for acquired benign tracheoesophageal fistula, sometimes this approach is contraindicated or the patient declines surgical management. In this report, we describe a case of a patient with tracheoesophageal fistula at the level of the carina due to bronchial arterial infusion chemotherapy. Closure could not be achieved in response to multiple treatment strategies, including airway stenting, esophageal stenting, occlusion with microcoils, or cyanoacrylate glue. We subsequently achieved closure of this fistula through the combination of a modified silicon stent and metallic stents.
A tracheoesophageal fistula (TEF) can occur after surgery, radiation, or intra-arterial chemotherapy, and can have a profound effect on quality of life by causing chronic aspiration pneumonia and dyspnea. Although several different management strategies have been employed for TEF, there is no established, definitive treatment. In this report, we describe a case of a patient with intractable TEF who was successfully treated with a combination of a modified silicon stent and metallic stents.
Case report
A 70-year-old man with hepatocellular carcinoma (HCC) underwent surgical resection, transarterial chemoembolization (TACE), percutaneous ethanol injection therapy (PEIT), and radiofrequency ablation (RFA). Although the primary tumor responded well, follow-up computed tomography (CT) scan demonstrated new pulmonary nodules.
CT-guided biopsy confirmed pulmonary metastases from the HCC. Surgical treatment and radiotherapy was precluded by the multiple metastases within the right lung. Thus, local treatment was performed with bronchial arterial infusion (BAI) of epirubicin (30 mg), mitomycin (6 mg), and cisplatin (25 mg) via the right bronchial artery. One month later, a second BAI was given. Measurement of “protein induced by vitamin K absence or antagonist-II” (PIVKA-II) levels showed a decrease in the normal range.
One week after the second BAI, the patient developed back pain, chest pain, sputum production, and paroxysms of coughing after eating or drinking. Chest CT scan revealed a TEF between the mid-esophagus and the tracheal carina, and bronchoscopy demonstrated a fistula 1 cm in diameter located at the membranous portion of the carina (Fig. 1).
Chest CT scan (a) and bronchoscopy (b) shows a TEF (size = 1 cm) at the membranous portion of the carina (arrow)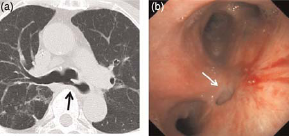
The patient was established on total parenteral nutrition (TPN) and treated conservatively. Repeat examination revealed that the wall of the fistula had completely epithelialized. The patient declined surgical management, and thus, a silicon tracheobronchial tube stent (Dumon Y-stent, Novatech, La Ciotat, France; 110 mm in length and 16 mm in outer diameter [OD] for trachea, 50 mm in length and 13 mm in OD for each bronchus, cut to 30 mm for trachea and 20 mm for right bronchus and 30 mm for left bronchus in length) was placed in the carina using a rigid scope under general anesthesia on four months after the final diagnosis. However, the stent did not fit the carina adequately, and the fistula remained patent.
After retrieving the tracheobronchial stent, further treatment was attempted through the use of metallic coils and cyanoacrylate glue (n-butyl cyanoacrylate [NBCA], Histoacryl-Blue, Braun, Melsungen, Germany), but these measures were also unsuccessful because the esophagus had dilated focally with a large diverticulum communicating directly with the carina without having a defined tract (Fig. 2). Thus, application of any occlusive or adhesive would subsequently fall into the bronchial or esophageal lumen.
The esophagus dilated focally with a large diverticulum communicating directly with the carina without having a defined tract
Fifteen months after the final diagnosis, the next attempt at treatment consisted of a retrievable and fully covered esophageal stent (16 × 140 mm Niti-S Esophageal Covered Stent; Taewoong Medical, Seoul, Korea) which was fixed externally to prevent migration. This was achieved by a transcervical approach in which the stent was fixed into place by tying the distal end of the stent to the dermis of the neck. Again, the fistula remained patent because of a gap between the stent wall and the dilated diverticulum-like esophageal wall. During these failures to close the fistula, the patient experienced recurrent aspiration pneumonia.
Two years after the final diagnosis, the esophageal stent was retrieved, and airway stenting was attempted using a combination of a modified silicon stent (TM Y-stent, Fuji Systems, Tokyo, Japan; 100 mm in length and 16 mm in OD for the trachea; 50 mm in length and 13 mm in OD for each bronchus, cut to 30 mm for trachea and 20 mm for right bronchus and 30 mm for left bronchus in length) and two bare metallic stents (Gianturco Z-stent, Cook Inc., Bloomington, IN, USA; 20 mm in diameter). A modified Y-shaped silicon stent was constructed by creating a longitudinal center cut to fit the tracheal and the bronchial lumens. The tube stent was introduced into the trachea by a rigid scope under general anesthesia and was placed at the carina so that the center cut faced forward. Next, two bare metallic stents were placed within the tube stent to expand its lumen (Fig. 3). This technique resulted in close approximation between the silicone tube stent wall and the tracheobronchial wall, successfully covering the fistula. No immediate or short-term complications were noted.
Modified Y-shaped silicon stent with a longitudinal center cut (a) and bare metallic stent (b). Bare metallic stents are placed in the silicone stent to expand its lumen (c). Bronchoscopy shows the modified Y-shaped silicon stent over an expanded bare metallic stent, which covers the fistula at the level of the carina (d)
Following this procedure, aspiration remitted, and there was no leakage through the fistula by esophagography. Following swallowing-training by a qualified therapist, the patient began to eat a soft diet and was discharged. However, he died of HCC four months after the final procedure. During the follow-up period, there were no findings of aspiration pneumonia.
Discussion
Acquired TEF can be caused by malignancy or by a host of non-malignant entities. Causes of acquired non-malignant TEF include blunt and penetrating trauma, granulomatous mediastinal infections, and iatrogenic injuries (1). Bronchial arterial infusion (BAI) chemotherapy is one iatrogenic cause, although there are relatively few reports of this phenomenon in the literature (2,3). BAI-induced TEF may result from flow of anti-cancer drugs into the esophageal branch of the bronchial artery in BAI and causes intractable aspiration pneumonia, which profoundly affects quality of life and can be life-threatening. Closure of the TEF can reduce the risk of aspiration pneumonia, allow recovery for an existing pneumonia, and relieve intractable coughing.
Operative closure is usually needed for TEF of benign etiology, but resection of the fistula and reconstruction of the airway and alimentary tract carry a high risk of complications (4). Some centers have utilized stenting, adhesives, or occlusives when surgical treatment is contraindicated or when a patient declines surgical management. Adhesives and occlusives may consist of fibrin glue, cyanoacrylate glue, bovine collagen, gelfoam, metallic coils, or metallic clips (5–7), and can produce good outcomes in patients with small fistulae without local infection but is less successful in patients with large diameter fistulae. In the present case, metallic coils and cyanoacrylate glue were unsuccessful due to the absence of a defined tract between the carina and the dilated diverticulum-like esophageal wall.
Other groups have utilized esophageal stenting, airway stenting, or both in combination for the treatment of TEF, and each approach has its benefits and drawbacks. The combination of esophageal and tracheal stenting may be more effective than either procedure alone, but mechanical friction between the two devices may cause a breakdown of the interposed portion of the tracheobronchial wall. Hence, the indication for combined placement has been limited (8).
There is no commercially available Y-shaped covered metallic stent that is suitable for use for a TEF at the level of the carina. Further, when placing a covered esophageal stent, external fixation is needed to prevent stent migration when distal stricture is absent. Eleftheriadis et al. described a method of stent fixation in which they tied threads to the proximal end of the stent and brought them out through the nostril and secured them with tape on the patient's auricle (9). However, this technique is associated with significant patient discomfort. Thus, in the present case, we achieved stent fixation using a technique of percutaneous transesophageal gastrotubing (PTEG) to tie the stent to the dermis of the neck. In the original PTEG method (10), a rupture-free balloon (RFB) catheter was inserted through the nose into the esophagus and inflated at first. The RFB was punctured percutaneously aided by ultrasonography and conventional fluoroscopy. Then, a guide wire followed by a dilator with a sheath was inserted. Finally an indwelling catheter was inserted into the digestive tract through the sheath. Using this technique prevented stent migration but still could not achieve closure of the fistula, due to gap between the stent wall and the dilated diverticulum-like esophageal wall.
Although self-expandable covered metallic stents can fit well within the tracheal or bronchial lumen, there is no commercially available Y-shaped stent for use within a bifurcating lumen. Silicon Y-stents, such as a Dumon stent or a TM stent, have the advantage of good flexibility and easy removal, but cannot always fit within the carina due to variable anatomy between patients and because the stent lacks expansile force. Indeed, this was the reason for failure when we employed an unmodified Dumon stent in the present case.
To solve this problem, we modified a Y-shaped silicon stent by creating a longitudinal center cut to fit the tracheal and the bronchial lumens. After placement of the modified stent at the carina with the center cut facing forward, we placed bare metallic stents within the tube stent to expand the modified stent lumen, resulting in close fit of the stent within the tracheobronchial wall. To our knowledge, this is the first time this procedure has been described in the published literature.
Spontaneous healing of fistulae has been reported in patients with acquired benign TEFs (11); thus, retrievable stents should be used whenever possible. In this case, the bare metallic stents placed within the silicone stent were not retrievable. We felt this was an acceptable option in this patient with a poor prognosis due to metastatic HCC. In patients with a more favorable prognosis, placement of irretrievable stents may be contraindicated.
In conclusion, acquired benign TEF at the level of the carina may not respond well to conventional management. The novel technique described in this report provides another effective option for selected patients.
