Abstract
Entrenched in current laboratory protocols for the measurement of plasma glucose is the false belief that sodium fluoride (NaF) is an effective inhibitor of glycolysis. The failure of NaF to properly control glycolysis decreases plasma glucose concentrations. The amount of the decrease can be clinically significant. The most recent (2011) laboratory guidelines for diagnosis and management of diabetes mellitus, approved by the American Diabetes Association, no longer recommend the use of NaF alone to control glycolysis. If plasma cannot be iced and separated from cells within 30 min of collection, the new guidelines recommend the use of a tube that contains a low pH buffer such as citrate. Proper control of glycolysis will affect current glucose cut-off points, but the exact effect is yet to be defined.
Historical background
Otto Folin and Hsien Wu 1 did not have problems with glycolysis in 1919, when they developed the first reliable and practical method for measuring blood glucose as a reducing substance. Folin and Wu prepared a protein-free filtrate from 2 mL of whole blood immediately after collection, thereby eliminating uncontrolled glycolysis.
Sodium fluoride (NaF) fails to prevent glycolysis for the first 3–4 h after blood is collected. 2 For the first two hours after blood is mixed with NaF, the rate of glycolysis is essentially identical to that occurring in a paired blood sample collected without the addition of NaF and stored under identical conditions. 3
This is not a new discovery. Seventy years ago, Ernest Bueding and Walter Goldfarb 4 reported that NaF did not prevent a decrease in glucose concentrations when blood was assayed four hours after collection, having been kept at 208C. Glucose concentrations fell by 2–12% in samples preserved with 1% NaF. Only samples containing iodoacetate, in addition to NaF, were found to have minimal loss of glucose (<4%).
These early findings, however, had no impact on the subsequent development of laboratory protocols for the collection of blood for glucose studies. NaF alone, in the absence of supporting data, became the preservative of choice for stabilizing blood glucose. For four decades, no one seriously questioned the validity of using NaF to inhibit glycolysis until Kazuo Uchida and associates at Kyoto Medical Science Laboratory applied for a US patent for a new method (acidification) to inhibit glycolysis in blood samples. 5 The patent application described why NaF alone failed to inhibit glycolysis for the first few hours. NaF inhibits enolase, an enzyme acting late in the glycolytic pathway, and has no effect on enzymes that act early in the glycolytic pathway. In contrast, acidification of blood below a pH of 6.0 stops glycolysis immediately.
In 1988, Uchida et al. 6 published a full description of the experimental studies upon which the patent was based. Acidification inhibits hexokinase and phosphofructokinase, enzymes that act early in the Embden–Meyerhof pathway. Glycolysis is instantly inhibited in erythrocytes, leukocytes and platelets when the blood pH is maintained between 5.3 and 5.9 with a citrate buffer.
In 1989, Chan et al. 7 confirmed the 1941 findings of Bueding and Goldfarb and recommended that physicians not depend on NaF to inhibit glycolysis. Chan's well-designed study had no impact, at the time, on manufacturing practices or the recommendations of organizations such as the American Diabetes Association (ADA).
Rediscovery of NaF failure to control glycolysis
My associates and I rediscovered the failure of NaF to properly inhibit glycolysis when, as part of a study to demonstrate that glucose concentrations in plasma and serum were essentially identical, we compared glucose concentrations in routinely collected paired samples of serum and plasma. The pairing of serum and plasma glucose concentrations was a random and infrequent event. It occurred only when a physician ordered a fasting glucose test that specified using plasma from a NaF tube, and at the same time the physician ordered a group of tests that included glucose and required the submission of serum from a centrifuged barrier tube. The barrier tubes were centrifuged in the physicians’ offices, whereas the NaF tubes were always centrifuged later in the laboratory.
We collected data from 1828 paired samples and found the glucose concentration in 61% (1118/1828) of the serum samples from barrier tubes to be greater than the glucose concentration in the paired plasma samples from NaF tubes. This was a surprise because we did not expect the serum glucose concentration to ever be higher than the glucose concentration in paired NaF plasma. The difference could be quite large. For example, the serum minus plasma glucose delta was >0.55 mmol/L in 206 samples, and the delta was >0.825 mmol/L in 23 samples. Figure 1 shows the pattern of the differences we observed.
A total of 1828 samples for fasting glucose were taken into both serum barrier tubes and tubes containing sodium fluoride (NaF) as an inhibitor of glycolysis. Results are reported as a percentage of the cohort that exceeds the differences indicated on the abscissa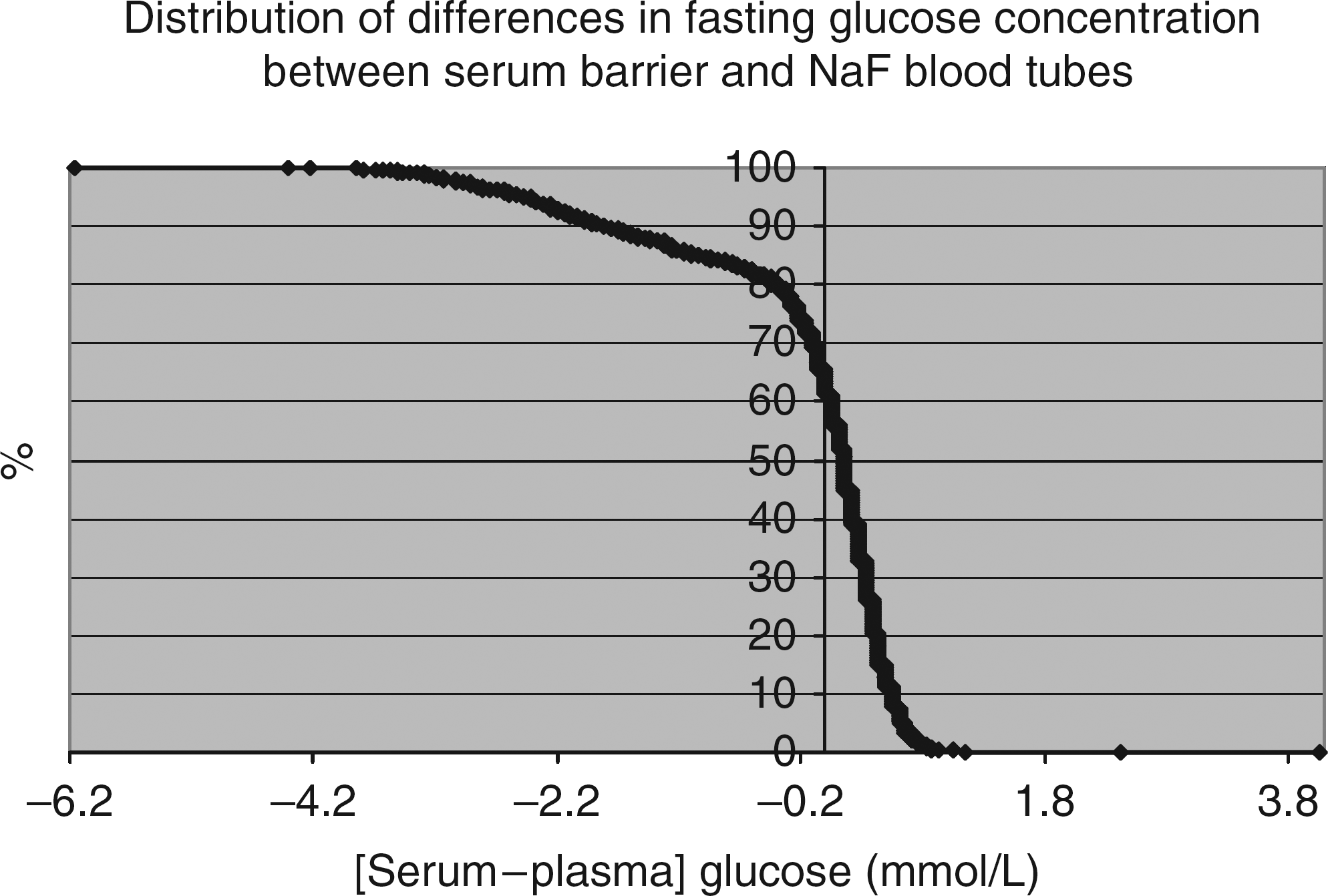
Serum glucose should never be higher than plasma glucose if glycolysis in plasma is inhibited properly. We found no difference in the glucose concentration of plasma versus serum in 91 paired plasma (heparin) and serum samples stored for 30 min at the same temperature and separated from cells at the same time. 3 Because of the time required for whole blood to clot, serum glucose concentrations will always be lower than plasma glucose concentrations if glycolysis in a plasma sample is inhibited immediately. The amount of the difference will vary with the glycolysis rate in the individual specimen and the time elapsed between collection and centrifugation. 2
New guidelines no longer recommend the use of NaF alone
For accurate results, glycolysis must be stopped immediately after blood is collected. To do this, follow the most recent (2011) guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus, 8 which state:
To minimize glycolysis, one should place the sample tube immediately in an ice-water slurry, and plasma should be separated from the cells within 30 min. If that cannot be achieved, a tube containing a rapidly effective glycolysis inhibitor, such as citrate buffer, should be used for collecting the sample. Tubes with only enolase inhibitors, such as sodium fluoride, should not be relied on to prevent glycolysis.
What to do
Stop relying on NaF to control glycolysis. Follow the new guidelines approved by the ADA. If a collection tube containing citrate buffer is not yet widely available in your country, ensure that plasma is separated from cells within 30 min of collection. Manufacturers are now aware that tubes containing NaF alone are no longer acceptable as a means of controlling glycolysis. Heparinized barrier tubes are available. Such a tube, if centrifuged soon after blood is collected, will stabilize plasma glucose for at least 24 h as it does for serum glucose. 3
Impact of the change and why it matters
The diagnosis of diabetes, the treatment of diabetes and the assessment of the risk of developing diabetes all require accurate measurement of glucose. Fixed cut-off values of plasma glucose are used to classify patients and to manage the disease. Almost all of the currently defined concentration cut-off values were derived from plasma glucose data using NaF tubes. 2 These values must be redefined using new data from samples that follow the new guidelines.
The impact on individual patients will be even greater than the impact on glucose concentration cut-off values. Small increments in blood glucose substantially increase the risk of developing diabetes mellitus. The continuous and graded quantitative relationship of fasting plasma glucose (FPG) to the risk of developing diabetes was well documented by Tirosh et al.9–11 For example, a person with an FPG between 4.83 and 5.00 mmol/L has an age-adjusted risk of developing diabetes that is 1.81 times that of a person with an FPG <4.55 mmol/L. Thus, a difference as small as 0.28 mmol/L nearly doubles the risk. Higher concentrations of FPG were correlated with higher risk ratios, for example, 3.05 times higher when FPG was between 5.27 and 5.49 mmol/L. The failure of NaF to properly control glycolysis falsely increases the within-person biological variability of plasma glucose and hinders our ability to accurately quantify the risk of developing diabetes for an individual patient.
Declarations
Footnotes
Acknowledgements
None.
