Abstract
Background
Because the causes of stones are uncertain, interventions to prevent recurrence have an insecure foundation. Progress depends on careful evaluation of stone formers.
Methods
A descriptive retrospective database study of 1983 men and 816 women from the Southampton stones clinic from 1990 to March 2007. Anonymized data from the first attendance were analysed using non-parametric statistical tests.
Results
Sex ratio (2.43:1), age (median 49 y, 2.5th-97.5th percentiles, 23-77 y men, 20-79 y women), recurrent stone formers (30%) and type of stone were similar to other centres. Women more often had a positive family history (24% versus 19% men), previous urinary infection (31% versus 5%) and structural urinary tract abnormality (14% versus 7%); more men had gout (5% versus 1%) and bladder outlet obstruction (3% versus <1%). Calcium, oxalate and uric acid excretion were increased in 43%, 17% and 22% respectively of men and 31%, 7% and 10% of women. Urinary calcium, oxalate and uric acid correlated significantly, r ranging from 0.149 to 0.311 for 24 h excretion and 0.510 to 0.695 for concentrations per litre. Twenty-two percent of men and 8% of women with normal parathyroid hormone had phosphaturia (excretion of phosphate corrected for glomerular filtration rate (TmPO4/GFR) < 0.70 mmol/L); 6% men and 1.6% women also had low plasma phosphate. Many variables correlated significantly but often weakly with age. Creatinine clearance, pH and (men) TmPO4/GFR decreased from 50 y, urine creatinine, calcium and citrate from 60 y.
Conclusions
Risk factors for stones differ between men and women, change with ageing and in some may have a genetic basis. The role of phosphaturia merits further exploration.
Introduction
Kidney stones are common. In America, the life-time risk for men is 12% and for women 5 1 with estimated annual incidences for men ranging from 100 to 300 per 100,000. 2 They are 2-4 times commoner in men than in women 2 and their prevalence has risen over the last 40 y.3,4 There is a high recurrence rate, estimated at around 25% in 10 y for a first time stone former and 75% for recurrent stone formers. 5 Passage of a kidney stone is extremely painful and may require surgical intervention. The unpredictability of recurrent episodes may affect the quality of life and career choice. The costs of medical and surgical procedures plus costs from loss of work are considerable. 6 There are therefore, strong incentives to prevent recurrence.
The majority of stones (around 75-80%) contain calcium salts, generally calcium oxalate often with calcium phosphate. In total, 5-10% of stones are uric acid and there are rare inherited stones.1,3,5 Many factors contribute to stone formation and often more than one is involved in individual patients. These include abnormally high excretion of stone constituents, poor fluid intake, alteration in urine pH and decreases in protective urinary components.1,7 Around 20% of stone formers have a family history of stones, probably indicative of a genetic predisposition.1,4,8 Medical disorders including urinary infections, diabetes and inflammatory bowel disease increase the risk for stones.2,5
A focused history and selective biochemical investigation of stone formers in outpatient clinics will often reveal abnormalities of life-style or metabolism which increase the risk for more stones. Management to prevent recurrence can then be tailored to the individual and has been shown to be clinically effective 1 and cost-saving. 9 Because of the uncertainty of the causes of stones, however, preventive measures have an insecure evidence base, are controversial and sometimes inappropriate.6,10 Low calcium diets, formerly advised widely, are associated with increased stone production. 11 Progress can only stem from careful evaluation of stone formers, including relevant biochemistry. There are published reports of observations on large cohorts of stone formers investigated in research centres in America and Europe12–15 and the UK. 3 These have often focused on subsets of patients. There have been no recent reports from a large British clinic.
The Stones Clinic in Southampton was established in the 1970s to advise about preventing stone recurrence. There are very few similar clinics in the UK. This report presents an overview of demographic and biochemical data of 2800 patients from their first clinic attendance between 1990 and 2007. Data for subsets of stone formers will be reported separately.
Methods
Database
The clinic database, held on Microsoft Access (97-2003), was created in 1996 and includes data for most patients investigated for stones risk at their initial presentation to the Clinic from June 1990 to March 2007. A minority of patients may have been missed at times of staff changes. In addition to biochemistry test results and composition of stones analysed, the records included age, sex, age at first clinic attendance, age of first stone episode, numbers of first-degree relatives with stones, whether patients had recurrent and/or bilateral stones, structural renal tract abnormalities, another recognized risk factor for stones, took mineral or vitamin D supplements or medications relevant to stones formation. Data were input by DG, two medical assistants and two departmental secretaries. For the research study, the data were anonymized by removing all items that could identify the patients, before any processing or statistical analyses were undertaken, as directed by the Regional Ethics Committee which approved this study of historic data (REC reference 07/H0502/172).
Patients
Patients investigated in the Renal Stones Clinic of the Department of Clinical Biochemistry, SUHT from June 1990 to March 2007 without exclusions.
Samples
At the first clinic attendance, 8 mL of non-fasting blood was collected routinely into both a lithium heparin tube and plain tube and a paired fresh random urine sample into a sterile container. The blood was centrifuged within 30 min of collection. Patients were instructed carefully about collecting a 24 h sample of urine directly, or via a sterile jug, into a new polythene container containing thymol crystals as preservative. On receipt in the laboratory, the 24 h urinary volume was determined from weight, an aliquot was removed for uric acid analysis and the urine remaining in the container was then acidified with 6 mol/L hydrochloric acid to pH 2.00 for other analyses. The standard tests for the clinic were: plasma: electrolytes, bicarbonate, creatinine, calcium adjusted for albumin, phosphate, alanine transaminase, alkaline phosphatase and parathyroid hormone (PTH); fresh random urine: pH, phosphate and creatinine concentrations; 24 h urine sample: volume, calcium and oxalate. Urine uric acid, citrate and magnesium were added variably, often for patients with more severe stone problems or with associated medical disorders.
Analyses
Samples were analysed by the Clinical Chemistry Department at Southampton General Hospital with assays used routinely. With the exception of PTH, all the plasma measurements and urine phosphate, creatinine, calcium, magnesium and uric acid analyses employed one of three main automated systems in use during the study: DAX (Bayer Diagnostics, Tarrytown, NY, USA), Advia 1650 (Bayer Diagnostics) and from the end of 2006 Beckman-Coulter DXC800 (Beckman Coulter Instruments Inc, Fullerton, CA, USA). The methods correlated well on all three instruments without need to alter reference limits. Calcium was measured with o-cresol phthalein complexone until introduction of an ion-selective electrode method late in 2006, magnesium with xylidyl blue or calmagite and uric acid with uricase procedure. Inter-assay imprecision (coefficient of variation) was <2% for plasma assays except creatinine (<5%), <3% for urinary phosphate and <5% for urinary creatinine, magnesium and uric acid. Citrate was measured with a citrate lyase procedure 16 using a Boehringer Mannheim Diagnostics Kit (Roche, Welwyn Garden City, UK). After removing interferences with activated charcoal, oxalate was measured by an oxalate oxidase method with a Sigma Diagnostics Kit (Sigma-Aldrich Company Ltd, Poole, UK) or Thermo Scientific Oxalate Kit (Alphalaboratories, Hampshire, UK). Cobas Mira (Roche, Welwyn Garden City, UK) and later Beckman-Coulter DXC800 analysers were used for both oxalate and citrate analyses and inter-assay variation was <5% and <10%, respectively. Intact PTH was measured using Immulite 2000 and 2500 Analysers (DPC, Gwynedd, UK) with a chemilumi-nescent immunometric method using Immulite kits. Three kits with small differences in sensitivity were used during the study period. Most samples were analysed with the first (up to December 2001). The second was used in 2002 and 2003, and the third from 2004. Inter-assay variation was <10%, recovery 100 ± 10%. All the assays performed well in external quality assessment schemes (WEQAS, UK NEQAS [PTH]). The pH of fresh urine was measured using a glass electrode (Whatman PHA 400 pH meter; Whatman, Maidstone, Kent, UK or Checker 1 pH meter; Hanna Instruments, Leighton Buzzard, UK). Stones were analysed qualitatively, with chemical methods using a Merckognost urinary calculi analysis kit (Merck, Darmstadt, Germany), with good performance in a National quality assurance scheme, UCL renal calculi QA scheme (University College, London, UK).
TmPO4/GFR was calculated from phosphate and creatinine concentrations of paired plasma and random urine samples using a computer programme.17,18 For an approximation of fractional calcium excretion, filtered calcium was assumed to be 60% of total plasma calcium (50% ionized and approximately 10% complexed) and used to calculate the ratio of calcium clearance to creatinine clearance.
Data analysis
Microsoft Excel (97-2003) was used to process the data, calculate mean and median values and 2.5th and 97.5th percentiles.
Few of the raw and log-transformed data had a Gaussian distribution (D'Agostino-Pearson test) and non-parametric statistical analyses were applied universally: Spearman's rank test to look for significant correlation between biochemical indices, and the two-tailed Mann-Whitney U test and two-sided Fisher's exact test or Chi-square tests to compare differences between groups. Graph Pad Prism 5 version 5.04 and Graph Pad Instat 3 (GraphPad, La Jolla, CA, USA) were used for the statistical computations.
Results
Demographics
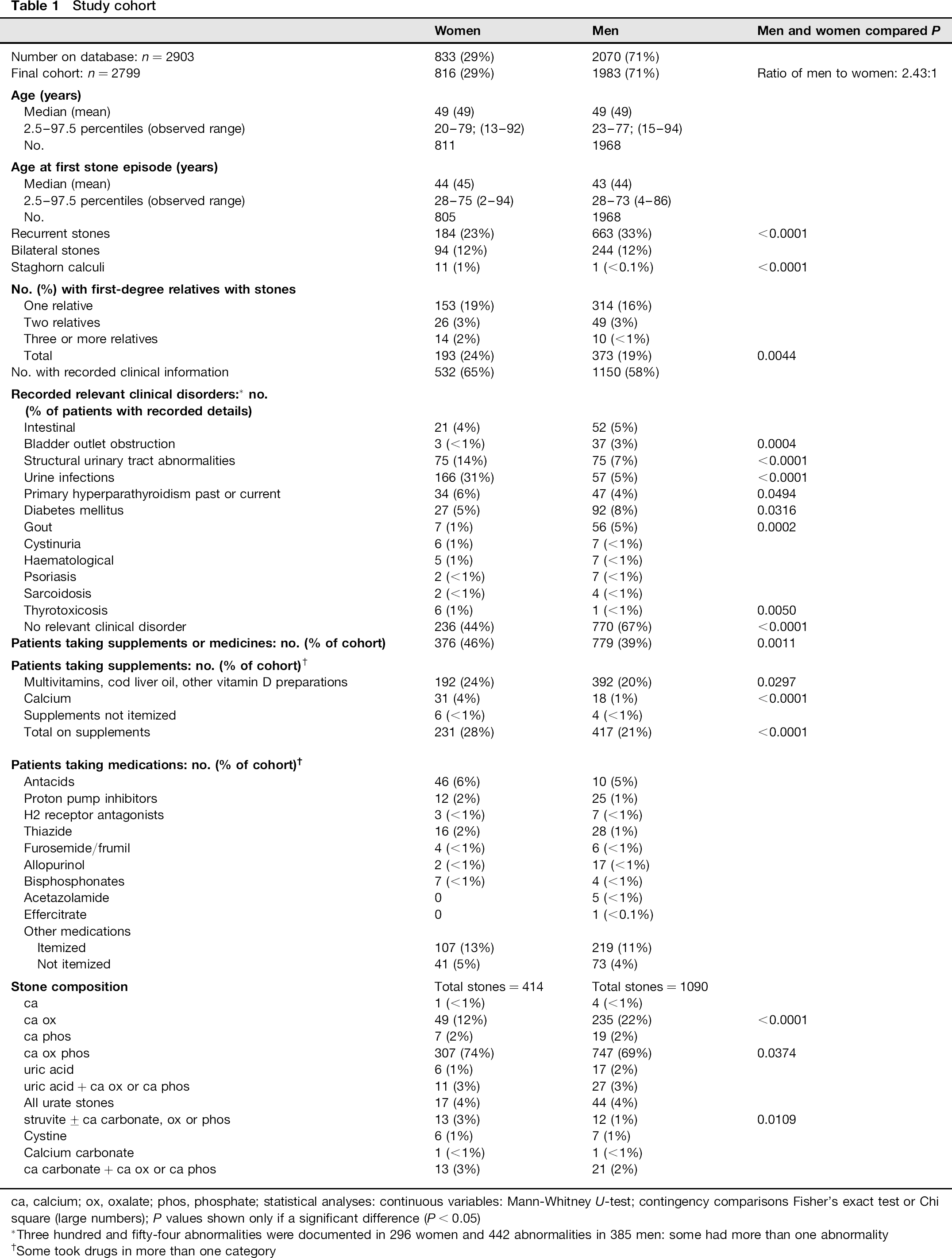
There were 2903 patients on the database. Of these, 104 were excluded because of duplicate entries, omission of gender or obvious errors in the date of birth. The final cohort comprised 2799 patients: 1983 men and 816 women (ratio of men to women 2.43:1), of whom 847 (30%) had produced stones on more than one occasion and were classed as recurrent stone formers. There were clinical entries for only 65% of women and 58% of men. Frequencies of clinical disorders are expressed as the percentage of the patients with recorded data and not of the full cohort. Structural urinary tract abnormalities included congenital abnormalities, single kidneys as a result of nephrectomy and multiple, but not single, renal cysts. Haematological abnormalities were uncommon and encompassed a miscellaneous group: leukaemia, thalassaemia, myeloma and lymphoma.
Table 1 summarizes the data. Findings of interest were:
Study cohort
ca, calcium; ox, oxalate; phos, phosphate; statistical analyses: continuous variables: Mann-Whitney U-test; contingency comparisons Fisher's exact test or Chi square (large numbers); P values shown only if a significant difference (P < 0.05)
Three hundred and fifty-four abnormalities were documented in 296 women and 442 abnormalities in 385 men: some had more than one abnormality
Some took drugs in more than one category
The surprisingly high percentage of patients (24% of women, 19% of men) with one or more first-degree relatives with stones. Fourteen women and 10 men had three or more affected relatives; The large percentage of women (31%) with a history of urinary infections compared with men (5%); The occurrence of large staghorn calculi in 11 women (seven required nephrectomy) but only one man. He and five women had recorded urinary infections; Higher frequencies of gout, bladder outlet obstruction (generally due to prostatic enlargement) and diabetes in men and structural urinary tract abnormalities in women; The high proportion of patients (20%) who took vitamin D supplements, generally over-the-counter preparations of cod liver oil or similar. Thirty-one women took a calcium preparation. Current or previous primary hyperparathyroidism in 5% of patients. The higher frequency of pure calcium oxalate stones in men and mixed calcium, oxalate, phosphate stones in women.
Plasma and urine biochemistry
Overview
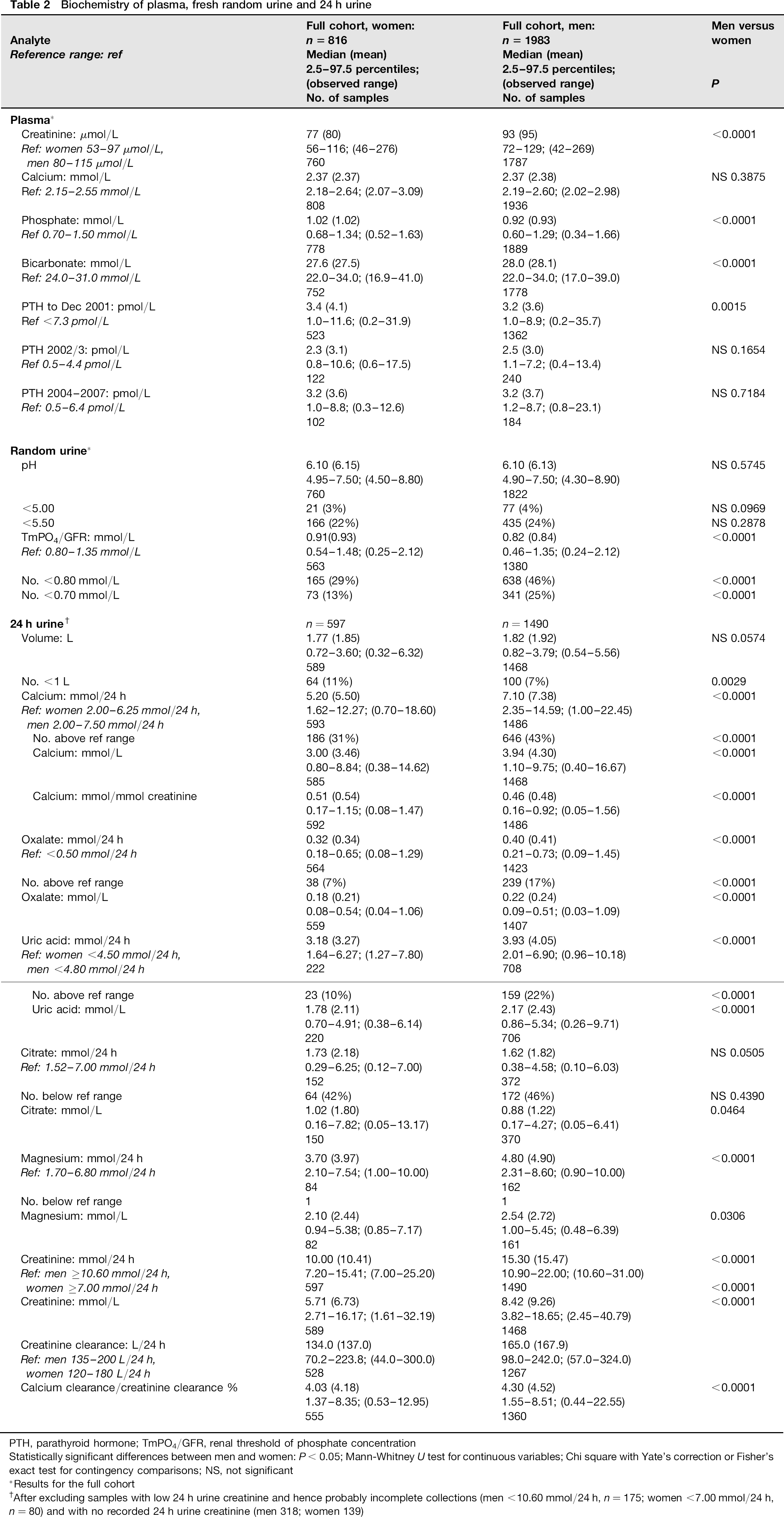
Table 2 presents data for plasma and random urine for the full cohort and for 24 h urine after excluding collections which were probably incomplete 19 (creatinine: men <10.6 mmol/24 h, n = 175; women <7.00 mmol/24 h, n = 80) and samples with no recorded creatinine (men 318, women 139): final cohorts 1490 men and 597 women. There were no other exclusions and the data were not trimmed for outliers. Most PTH measurements were with the first analytical kit (74%) and only 14% and 11% with kits used in 2002-2003 and from 2004, respectively. Data were frequently incomplete. After creatinine exclusions, 24 h urine uric acid, citrate and magnesium were measured in only 930, 524 and 246 samples, respectively. Height and weight were seldom recorded in the clinic hence creatinine clearance is not related to surface area.
Biochemistry of plasma, fresh random urine and 24 h urine
PTH, parathyroid hormone; TmPO4/GFR, renal threshold of phosphate concentration
Statistically significant differences between men and women: P < 0.05; Mann-Whitney U test for continuous variables; Chi square with Yate's correction or Fisher's exact test for contingency comparisons; NS, not significant
Results for the full cohort
After excluding samples with low 24 h urine creatinine and hence probably incomplete collections (men <10.60 mmol/24 h, n = 175; women <7.00 mmol/24 h, n = 80) and with no recorded 24 h urine creatinine (men 318; women 139)
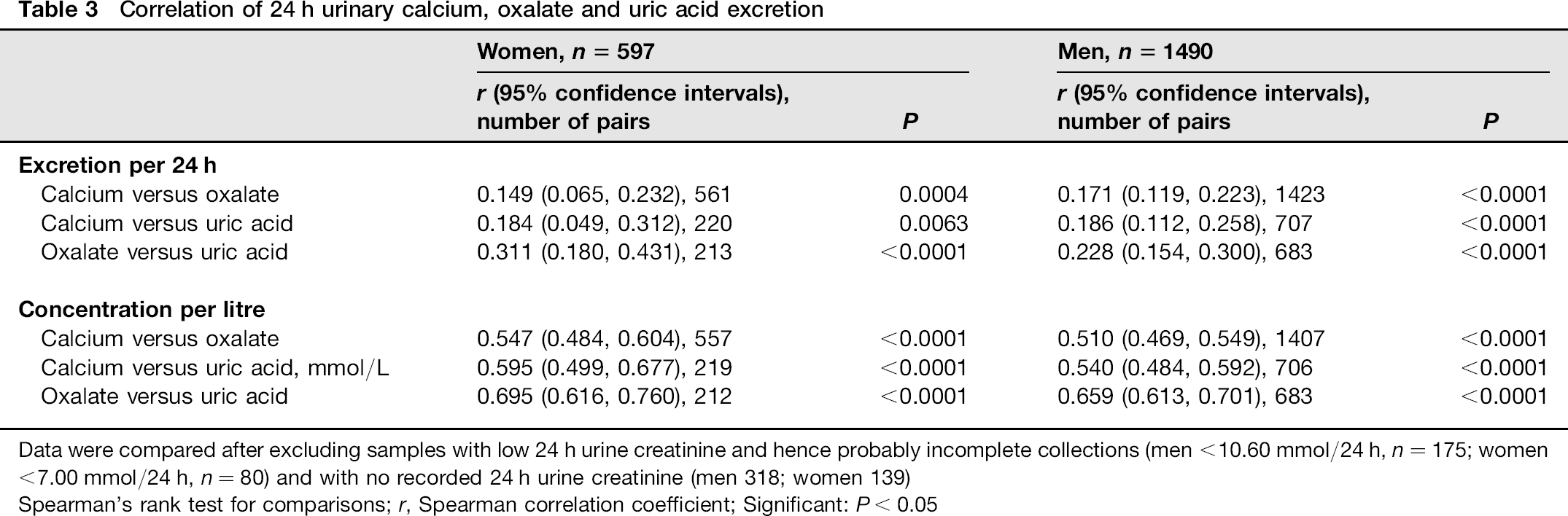
Calcium, oxalate and uric acid excretion were significantly correlated, with stronger association for concentration than for 24 h excretion (Table 3). Citrate excretion and urinary pH were not significantly correlated.
Correlation of 24 h urinary calcium, oxalate and uric acid excretion
Data were compared after excluding samples with low 24 h urine creatinine and hence probably incomplete collections (men <10.60 mmol/24 h, n = 175; women <7.00 mmol/24 h, n = 80) and with no recorded 24 h urine creatinine (men 318; women 139)
Spearman's rank test for comparisons; r, Spearman correlation coefficient; Significant: P < 0.05
Features of note were the significant sex differences observed for some plasma and urine constituents, the higher frequency of abnormalities associated with stones risk in men than in women, the high frequency of phospha-turia as defined by low TmPO4/GFR and of hypocitraturia, and the collective increases in abnormalities with low fluid intakes.
Sex differences
Plasma and urine creatinine and creatinine clearance were higher in men;
Plasma bicarbonate was higher in men but the difference between median concentrations was small;
Plasma phosphate and TmPO4/GFR were both significantly lower in men. The data were re-examined after excluding patients with presumptive primary hyperparathyroidism, others with raised PTH or with no PTH result. The median and 2.5th-97.5th percentiles for plasma phosphate for men were 0.93, 0.60-1.29 mmol/L, n = 1596; women 1.04, 0.70-1.34 mmol/L, n = 631 (P < 0.0001) and for TmPO4/GFR for men were 0.83, 0.48-1.36 mmol/L, n = 1163; women 0.93, 0.59-1.48 mmol/L, n = 445 (P< 0.0001). Plasma phosphate was strongly correlated with TmPO4/GFR (men r= 0.738, 95% confidence intervals CI 0.709, 0.763; women r = 0.701, CI 0.649, 0.747, P < 0.0001);
Men had higher 24 h urinary excretion and urine concentrations of calcium, oxalate, uric acid, magnesium but lower citrate concentration. The ratios of calcium to creatinine and of calcium clearance to creatinine clearance were lower;
Men more often had increased excretion of the major stone constituents: 24 h urine calcium (>7.50 mmol) was raised in 43%, oxalate in 17% and uric acid (>4.8 mmol) in 22%, compared with increased 24 h urine calcium (>6.25 mmol) in 31%, oxalate in 7% and uric acid (>4.5 mmol) in 10% of women. Only 17% of women excreted >7.5 mmol of calcium per 24 h.
Phosphaturia
A high percentage of patients had TmPO4/GFR below the reference limit of 0.80 mmol/L. 17 After excluding patients with absent or abnormal PTH results (see 3 above) TmPO4/GFR was <0.80 mmol/L for 44% of men and 24% of women (P < 0.0001), and applying a lower threshold of <0.70 mmol/L, 20 for 22% of men, 8% of women (P< 0.0001). Six percent of men and 1.6% of women had evidence of a renal phosphate leak as defined for fasting samples by plasma phosphate below the reference range, TmPO4/GFR <0.70 mmol/L and a normal plasma PTH concentration. 20 Compared with stone formers with a normal PTH, plasma phosphate and TmPO4/GFR >0.80 mmol/L, men with phosphate leak were older (49, 27-79 y [median 2.5fh-97.5fh percentiles], n = 72; controls 46, 22-74 y, n = 615; P= 0.0264), had higher plasma creatinine (94, 76-121 μmol/L, n = 69; controls 91, 70-120 μmol/L, n = 589; P = 0.0055) and lower plasma calcium (2.35, 2.20-2.50 mmol/L, n = 72; controls 2.38, 2.22-2.54 mmol/L, n = 618; P= 0.0008). Women with a phosphate leak had lower plasma bicarbonate (25.0, observed range 23.0-28.0 mmol/L, n = 7; controls 28.0, 22.8-34.0 mmol/L, n = 303; P = 0.0098) and more often had a first-degree affected relative (5 [71%], n = 7; controls 87 [28%], n = 311; P = 0.0078). No other significant differences were found.
Collective increases in urinary risk factors
Because uric acid was analysed on few samples, combined data for urinary calcium, oxalate and uric acid were available from only 683 men and 211 women. Collective increases of calcium, oxalate and uric acid excretion per 24 h were observed for 26 (4%) men and 4 (2%) of women. Applying arbitrary upper limits of 3.8 mmol/L for calcium, 0.25 mmol/L for oxalate and 2.40 mmol/L (the upper reference limits for 24 h excretion for men in 2 L of urine) however, the concentrations of all three constituents were raised in 144 (21%) samples from men and 31 (15%) of samples from women. Poor fluid intake was recorded in 66 of the men and 24 women, and inflammatory bowel disease ± ileostomy in 10 of the other patients. For 43 men with high concentrations and 18 women, 24 h excretion of all three constituents was normal; however, their 24 h urine volume was low: men 1.05, 0.62-1.48 L/24 h (median and 2.5th-97.5th percentiles), 20 <l L/24 h; women 0.77, 0.48-1.41 L/24 h, 13 <1 L/24 h.
Other observations
Twenty-nine (4%) of 747 women and 45 (2.5%) of 1787 men with paired plasma calcium and PTH results had biochemical abnormalities consistent with primary hyperparathyroidism (plasma calcium >2.55 mol/L and PTH>3.0 pmol/L; >2.0 pmol/L, 2002-2003 assay).
The median (2.5th-97.5th percentiles) urine calcium of 30 women taking a calcium supplement was 4.04 (0.81-12.44) mmol/24 h. Five had marginally raised excretion (6.60-7.10 mmol/24 h) and three had significant increases (9.57, 11.80 and 14.14 mmol/24 h).
Changes in biochemistry with age
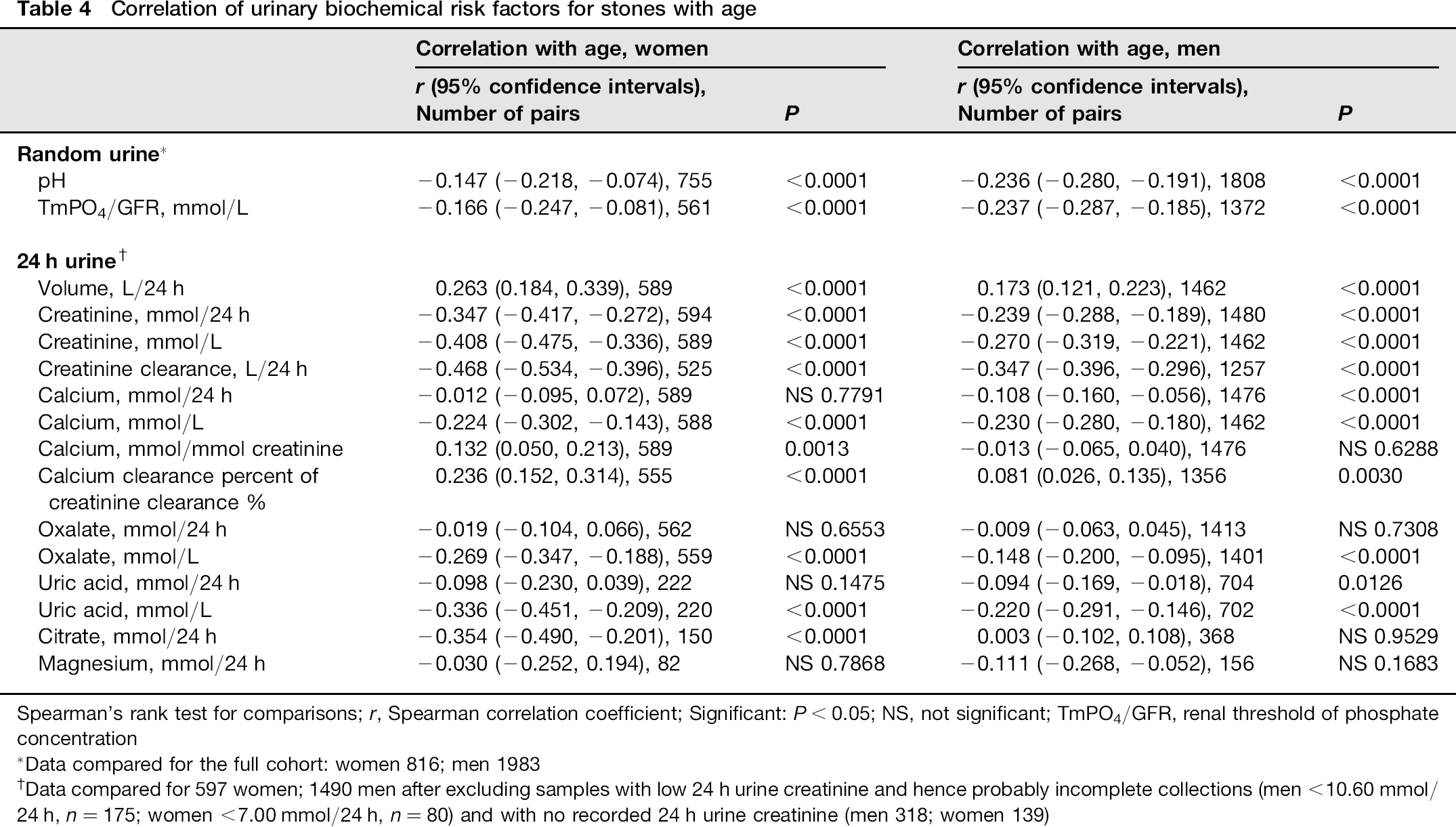
There were weak correlations between the ages of stone formers and their measured urine variables (Table 4). Of interest were the negative correlations for urine pH and TmPO4/GFR, particularly in men.
Correlation of urinary biochemical risk factors for stones with age
Spearman's rank test for comparisons; r, Spearman correlation coefficient; Significant: P < 0.05; NS, not significant; TmPO4/GFR, renal threshold of phosphate concentration
Data compared for the full cohort: women 816; men 1983
Data compared for 597 women; 1490 men after excluding samples with low 24 h urine creatinine and hence probably incomplete collections (men < 10.60 mmol/24 h, n = 175; women <7.00 mmol/24 h, n = 80) and with no recorded 24 h urine creatinine (men 318; women 139)
Data for each decade were examined after excluding patients with clinical disorders, medications and supplements which may alter mineral metabolism (Tables 5 and 6). Men were noted to have a progressive increase in urine volume with age, decreases in creatinine clearance, urine pH and TmPO4/GFR over 50 y and in 24 h urine calcium, citrate and creatinine over 60 y. There was little change in oxalate excretion. Urate stones increased from 1 -2% to 10% of stones after 60 y and mixed calcium, oxalate, phosphate stones decreased from around 70% to 60% of stones over 60 y. Similar trends were noted in women. Exceptions were TmPO4/GFR which showed little change until 70 y, and an increase in pure calcium oxalate stones from 10% to 24% after 60 y.
Urine and stones biochemistry of 1340 men grouped according to age decades
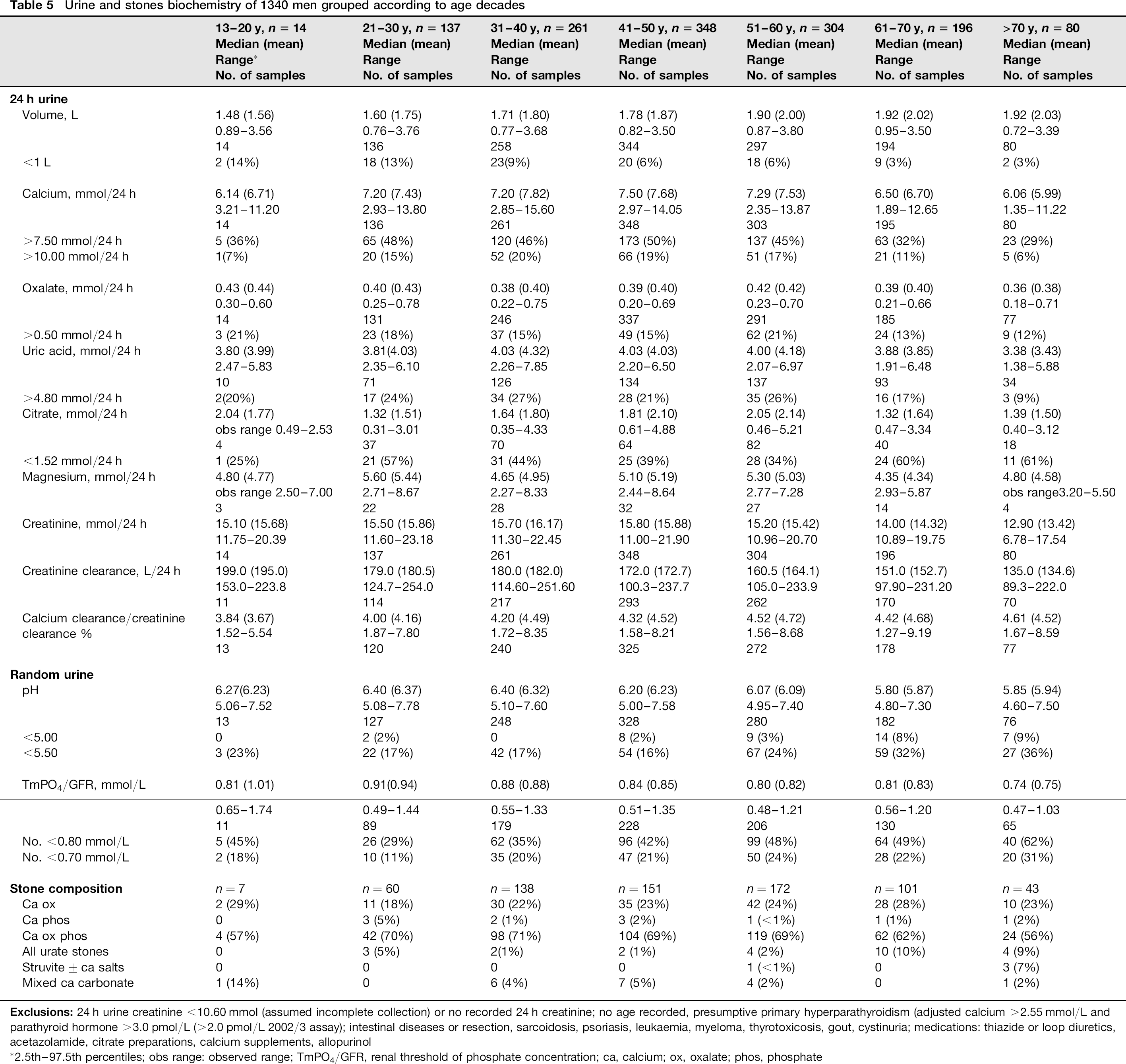
2.5th-97.5th percentiles; obs range: observed range; TmPO4/GFR, renal threshold of phosphate concentration; ca, calcium; ox, oxalate; phos, phosphate
Urine and stones biochemistry of 509 women according to age decades
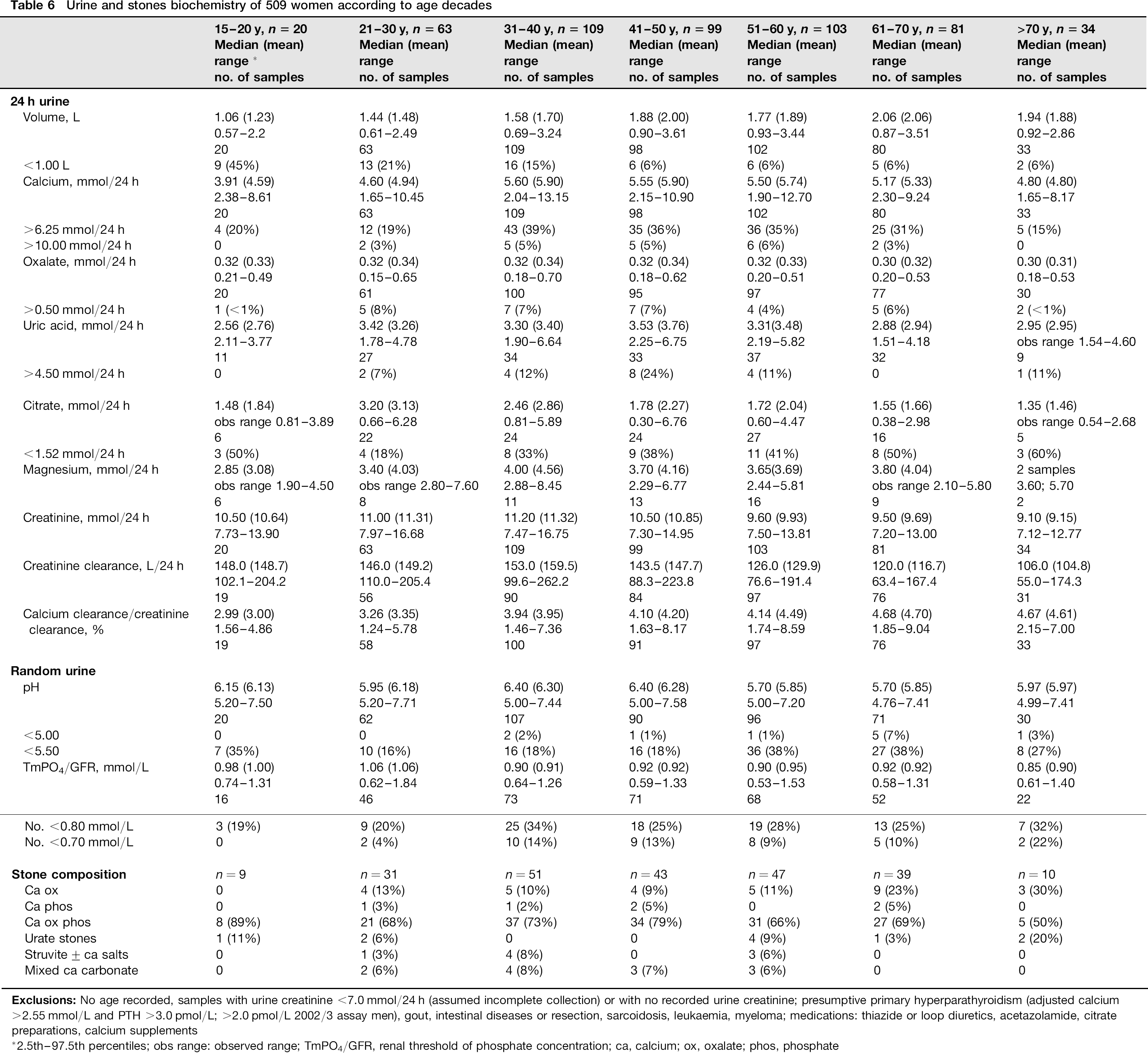
2.5th-97.5th percentiles; obs range: observed range; TmPO4/GFR, renal threshold of phosphate concentration; ca, calcium; ox, oxalate; phos, phosphate
Discussion
The first attendance at the Southampton stones clinic is a triage assessment to distinguish between patients with no, or only minor, biochemical risks for stones who merely require dietary and life-style advice, and those who require further investigation and preventive management. A relatively simple investigation protocol was implemented to cause minimal inconvenience for the patients. This is the largest cohort of stone formers from a UK clinic to be reported recently. Because it was a single centre study there was consistency in investigative protocols and laboratory analyses.
The study has certain limitations. The quality of the primary data from the clinics varied, with variation among the consultants in eliciting and recording clinical details. Height and weight were not recorded routinely. Non-fasting blood and urine samples were collected at the clinic. Although a central core of biochemistry tests was the norm, requests for 24 h urine uric acid, magnesium and citrate varied among consultants, were often selected for more complex patients and the results may not represent the full patient cohort. There were deficiencies in the database, particularly in entries of relevant clinical details and medications. These could not be rectified because the data were anonymized. However, the effects of incomplete data were largely over-ridden by the large number of patient results and probably had little impact on the overall conclusions.
Demography
The ratio of men to women of 2.43:1 and age at presentation agreed closely with other clinics.2,21,22 Thirty-three percent of men and 23% of women were recurrent stone formers, in line with other reports.2,5 Most stones produced were calcium oxalate (89%) with a 1:4 ratio of calcium to oxalate to mixed calcium oxalate phosphate stones. Pure calcium phosphate stones were rare (2%). These are similar to frequencies in the USA, Northern Europe and Australia.1,2,5 Uric acid stones (4%) were less common than 5-23% reported, as was the incidence of struvite stones (<2%) compared with 5-15%reported.1,2 The referral practices, population, dietary characteristics and period of study all influence the clinic stones profiles. Mixed calcium oxalate phosphate stones were commoner in women but phosphate was not quantified. A higher frequency of stones containing >50% of phosphate was reported in women. 23 As in other studies, pure calcium oxalate stones were commoner in men and struvite stones in women; however, uric acid stones were not increased in men.2,22 The incidences of past or current primary hyperparathyroidism and of presumptive hyperparathyroidism from tests at the clinic are in line with reported frequencies of 2-8% and 1.65-13.3% of stone formers.24,25
The surprisingly high percentage of patients with a first-degree relative with stones is similar to reported frequencies of 16.8% of men and 22.7% of women with stones compared with 6% of controls, 2 and indicate that genetic factors are implicated in stone formation in some kindreds.1,4,8 The complexity of mineral and phosphate absorption, turnover and excretion has become increasingly apparent and the long list of genes involved is extending. Inherited pathological gene mutations associated with hypercalciuria, phosphaturia and impaired hydrogen ion excretion are well-recognized. Common polymorphisms of these and other genes may explain the heritable risk.1,4,8,26
There were clear differences between men and women in the frequency of disorders which increase stones risk. The most striking was the large percentage of women who had previous urinary tract infections. This is perhaps a little surprising since stones are commoner in men and might provide a nidus for infection. Female anatomy is more likely to account for the increase in the majority of cases. Structural abnormalities of the urinary tract affecting urine flow were probably responsible in a minority. Infection stones are commoner in women. 22 Very few women had symptomatic infections at the clinic prompting bacteriological investigation, and the incidence of struvite stones in the cohort was low. Inflammatory processes associated with previous infections might be implicated in stone formation. 27 Eleven women but only one man had large damaging staghorn stones which are commonly attributable to infection (44% in a recent series 28 ).
Twenty percent of patients took vitamin D supplements, generally cod-liver oil or similar preparations with a low vitamin D content (400 IU in most standard formulations), some of which also contained small amounts of calcium. It is highly unlikely that such low doses had a significant impact on urine biochemistry or stones risk. However, three of 30 women taking a calcium supplement had significantly increased urinary calcium. Since calcium and vitamin D supplements were found to increase stones frequency in postmenopausal women, 29 it seems advisable to measure 24 h urine calcium of stone formers receiving calcium treatment.
Abnormalities of urine biochemistry associated with stones risk
Urinary abnormalities which increase stones risk were frequent and were commoner in men. Twenty-four hour calcium excretion is a continuous variable. There is no clear cut-off between normal and abnormal 30 and the widely used reference limits of 7.50 mmol/24 h for men and 6.25 mmol/24 h for women are arbitrary.1,7 Hypercalciuria, hyperoxaluria and hyperuricosuria were commoner in men than in women, with similar frequencies to other cohorts.2,4,7,31 An important observation was that excretion of calcium, oxalate and urate were significantly correlated, with stronger association between urine concentrations than 24 h excretion. In a small subset of patients with complete data, the proportion with collective increases in all three increased from 3% for 24 h excretion to 20% when concentrations were expressed per litre. Those patients with normal 24 h excretion but increased concentrations had low urinary volumes, often less than 1 L/24 h, demonstrating the risk of an inadequate fluid intake. Collective increases in concentration of all three stones constituents will increase urine supersaturation and increases stones risk significantly. 19 While measurement of 24 h urine excretion is essential for metabolic diagnosis, concentrations per litre should also be calculated as a more appropriate guide for stones prevention. 19 Achieving a safe urine volume in patients with an ileostomy may be difficult.
Hypocitraturia is listed as a major cause of calcium stones.1,7,15 With lower limits of 2.0 and 1.7 mmol/24 h, two studies reported hypocitraturia in 30% and 32.7% of recurrent stone formers.7,32 However, one large study found no difference in citrate excretion between stone formers and controls. 19 Almost 50% of Southampton patients had citrate excretion below the lower reference limit of 1.52 mmol/L, derived in-house from healthy laboratory staff and students and, contrary to expectation, there was no correlation with urine pH. 33 The assay performed well in the external quality assessment scheme, and thymol in the collection container protected against preana-lytical bacterial degradation. The explanation for low measured citrate in stone formers is unknown. Underestimation of complexed citrate in the enzyme-based assay might be a possibility. The high positive rate limits its value for decision-making in a triage clinic.
Magnesium may also have a protective role in calcium stone formation. 7 Urine magnesium was measured in only a small proportion of clinic patients and only three had low levels, in line with a reported incidence of only 4% in recurrent stone formers 31 and of no major differences between stone formers and controls. 7 Magnesium analysis could be restricted to those at risk of depletion from intestinal disorders or chronic diuretic use, or with a possible genetic defect.
Phosphaturia
The renal threshold of phosphate concentration (TmPO4/ GFR) is a practical means of assessing overall tubular capacity for tubular phosphate re-absorption which is independent of GFR. 17 Low values indicate phosphaturia. Considering only patients with biochemical evidence of normal parathyroid function, phosphaturia was common among stone formers in this study, even applying the low threshold of TmPO4/GFR <0.70 mmol/L. 20 Analysis of non-fasting samples may have contributed to the high frequency. Although 24 h urine phosphate is measured in clinics, this largely reflects dietary phosphate. Data on phosphaturia in stone formers are sparse. 7 Calcium stone formers with normal PTH have lower TmPO4/GFR than normal subjects. 34 In around 20%, both plasma phosphate and TmPO4/GFR are low, consistent with a renal phosphate leak, 20 found in 6% of men and 1.6% of women in this cohort using criteria for fasting samples. 20 When renal phosphate re-absorption is reduced a large intake of phosphate will increase phosphaturia, calcium salt supersaturation and the risk of forming calcium stones. Since calcium phosphate precipitation increases with increasing pH,1,7,35 the risk might be greatest postprandially, if an influx of phosphate is combined with increased urine pH (the alkaline tide) which some, but not all, 36 have observed after meals. There is evidence that calcium phosphate deposition initiates calcium oxalate stone formation.1,7,35 Phosphate supplements to correct low plasma phosphate levels may be inadvisable because of the increased risk for stones.
The explanations for the high frequency of phosphaturia among stone formers without hyperparathyroidism and its commoner occurrence in men are unknown. Around 80% of circulating phosphorus is filtered at the glomerulus and then 70-100% is actively re-absorbed in the proximal tubule under hormonal control and influenced by volume and acid-base status.37,38 Most phosphate re-absorption is via the sodium-phosphate co-transporter Npt2a which is inhibited by the principal phosphaturic hormone FGF23 in conjunction with a co-factor, klotho. 38 Genetic variation in any of the factors involved in this complex system might account for phosphaturia and increases in serum FGF23, observed in stone formers with phosphate leak. 20
Changes in biochemical risk factors with age
Comparable with this study, urinary calcium, uric acid, citrate and pH decreased in stone formers over 60 y. 15 Similar trends were observed in healthy non-stone formers for urine calcium, uric acid, pH and (women only) citrate; 21 however, their urine volume decreased. Stone formers may have deliberately increased their fluid intake or have decreased renal water re-absorption. Others have reported falling pH with age in normal individuals 39 and stone formers.14,15 The explanation is unknown. Decreased renal ammonia production has been proposed,39,40 possibly attributable to insulin resistance with decreased activity of glutaminase and of the NHE3 Na+/H+ exchanger in the proximal tubule. 40 A urinary pH persistently <5.5 significantly increases the risk of uric acid precipitation1,5 and probably underlies the higher incidence of uric acid stones in patients over 60 y as found in this cohort. 22 Dissolution of phosphate from mixed calcium oxalate phosphate stones at low pH 5 might explain the fall in the ratio of mixed to pure calcium oxalate stones observed in older women. To our knowledge, decreasing TmPO4/GFR with age has not been reported. It may be a consequence of declining renal function.
Summary
Many women did not have a demonstrable biochemical abnormality to explain their stones and other factors must be sought. The high incidence of previous urinary infections, structural abnormalities of the urinary tract and possibly genetic factors may be relevant. Significant correlation between calcium, oxalate and uric acid excretion, reported by few centres, is important. Phosphaturia similarly is under-reported but, particularly in men, would be consistent with emerging concepts of stone formation.
Declarations
Footnotes
Acknowledgements:
We are grateful to Dr Emmanuel Abu for allowing us to include his patients, Dr Mary Curran and Morag Sloan for their substantial help with data input, Dr Paul Cook for helpful discussion and Dr John Middleton who started the Clinic
