Abstract
Background
There are a number of inborn errors of dopamine biosynthesis for which prolactin measurement may be a useful screening tool. However, the interpretation of prolactin results is difficult as age-related paediatric reference intervals are scant especially in infancy.
Methods
Serum prolactin reference intervals were calculated from the existing laboratory data using an indirect method, based on patients (n = 2369) attending a tertiary care hospital over a 10-year period. The data were log transformed and partitioned by age and sex.
Results
The prolactin results for all age and sex stratifications show non-Gaussian distributions (P < 0.0001). There are apparent age-related differences for prolactin during the first year of life with prolactin declining sharply during the first year of life and continuing to decline until five years of age when the concentrations stabilize. The sex differences are small until 13-17 years of age when prolactin is higher in girls than in boys.
Conclusions
This is the first published study to report age-related prolactin reference intervals spanning all paediatric age groups where the data are age partitioned in infants under one year of age using an ‘indirect’ approach. It is envisaged that these reference intervals will be used to aid in the selection of patients presenting with features of central dopamine deficiency for lumbar puncture. Also, these reference intervals have the potential for treatment monitoring of patients with diagnoses of inborn errors of dopamine biosynthesis.
Introduction
A number of inborn errors of metabolism affecting the metabolism of the monoamine neurotransmitters, serotonin and dopamine, are documented. These include disorders of tetrahydrobiopterin (BH4) metabolism, tyrosine hydroxylase deficiency, aromatic amino acid decarboxylase (AADC) deficiency and mutations affecting the dopamine reuptake transporter. 1 Clinically, these disorders can have a wide presentation that can include a severe movement disorder associated with oculogyric crisis, athetosis, truncal hypotonia and limb hypertonia as well as hypersalivation, nasal congestion, excess sweating and sleep disturbances. This group of disorders is amenable to treatment. Furthermore, it appears that the earlier the commencement of treatment the better the clinical outcome. While many of these symptoms are observed in patients during the first few months of life, the median age of diagnosis, e.g. for AADC deficiency, is six years of age (range: 4 months to 24 years). 2
Currently, once clinical suspicion has arisen, a biochemical diagnosis is initially achieved by cerebrospinal fluid (CSF) analysis of the dopamine and serotonin metabolites, homo-vanillic acid (HVA) and 5-hydroxyindoleacetic acid, in conjunction with assessment of BH4 and related metabolites. 1 The decision to proceed to a diagnostic lumbar puncture (LP) is most often based upon clinical presentation. In view of the fact that a number of unrelated conditions could mimic the primary disorders of dopamine and serotonin metabolism, a peripheral biomarker that reflects central neurotransmitter status would be helpful. Ideally, this analyte would reflect responsiveness to treatment thereby reducing the frequency of ‘treatment monitoring LPs’.
There are case reports of high serum prolactin in association with dopamine deficiency and the use of dopamine receptor antagonists.3,4 Hypothalamic dopamine is the major inhibitor of prolactin release from the anterior pituitary gland.5,6 Therefore, in central dopamine-deficient states, serum prolactin can be elevated.7,8 Concerning the inherited disorders of monoamine neurotransmitter metabolism, Spada et al. 4 demonstrated elevated serum prolactin concentrations in patients with BH4 deficiency. Furthermore, these authors reported a decrease in concentrations in response to treatment with the dopamine precursor, L-dihydroxyphenylalanine, and an inverse relationship between CSF HVA and serum prolactin. Thus, assessment of peripheral prolactin status may be a useful adjunct with regards to the investigation/monitoring of patients with inherited disorders that affect central dopamine availability.
Measurement of prolactin is widely available. However, there is an apparent lack of paediatric reference intervals reported in the literature. Currently, the interpretation of serum prolactin results is difficult in the absence of age-and sex-appropriate reference intervals. Methods of establishing and verifying reference intervals, advocated by the International Federation of Clinical Chemistry, are described in the Clinical Laboratory Standards Institute guideline, C28 A3. 9 The measurement of the analyte concentrations in over 120 healthy individuals per reference interval is required to establish reference intervals but is very difficult and considered unacceptable in children, especially where there are significant age differences requiring multiple age partitions. Consequently, the aim of this study was to establish age-and sex-related paediatric reference intervals using an indirect method that could then be utilized for the investigation of patients suspected of having neurometabolic conditions affecting dopamine metabolism.
Methods
An indirect approach 10 of establishing reference intervals from existing laboratory data was used as an alternative method. All prolactin results reported between August 2002 and August 2010 were extracted from the Laboratory Information System into a Microsoft Excel file (n = 4040). All prolactin results were obtained from serum samples and were measured using the prolactin assay on the Siemens Immulite 2000/2500 analysers (Siemens Healthcare Diagnostics (UK), Camberley, Surrey, UK). The assay is an automated chemiluminescent sandwich immunoassay which is standardized to the third International Reference Preparation (IRP) World Health Organization reference standard 84/500. The coefficients of variance for the prolactin assay were 4.1% at 170 mU/L, 5.1% at 319 mU/L and 5.2% at 1272 mU/L over a one-year period using three levels of quality control material (product number CON6; Siemens Healthcare Diagnostics). The percentage bias from the all laboratory trimmed mean was -11.5%, based on six months external quality assurance data form February 2010 to July 2010. Clinical details were checked and data were excluded in patients with evidence of hypopituitarism, brain disease, epilepsy and other related endocrine disorders. Data were also excluded where there was more than one result on a patient. The data were then anonymized. The remaining prolactin results (n = 2369) were processed further. For children over one year of age, the data were initially partitioned by sex and by one-year increments. The data were combined if the reference intervals were not significantly different using Student's t-test. For children under one year of age, partitioning was limited to age alone (1-3-month increments) due to low numbers in these reference sample groups. Earlier studies have not demonstrated significant sex difference in infants. 11 Obvious outliers were removed using the Dixon test. The data were found to be non-Gaussian (skewed to the left with a long right hand tail) but on log transformation the data normalized (Gaussian). A parametric method was used as there were insufficient data to use non-parametric methods. The 2.5th and 97.5th percentiles were calculated and represent the lower and upper reference limits (Table 1). The software used for this study was Analyse-it statistical package for Microsoft Excel (Analyse-it Software Ltd, Leeds, UK).
Prolactin reference intervals (2.5th and 97.5th percentiles)
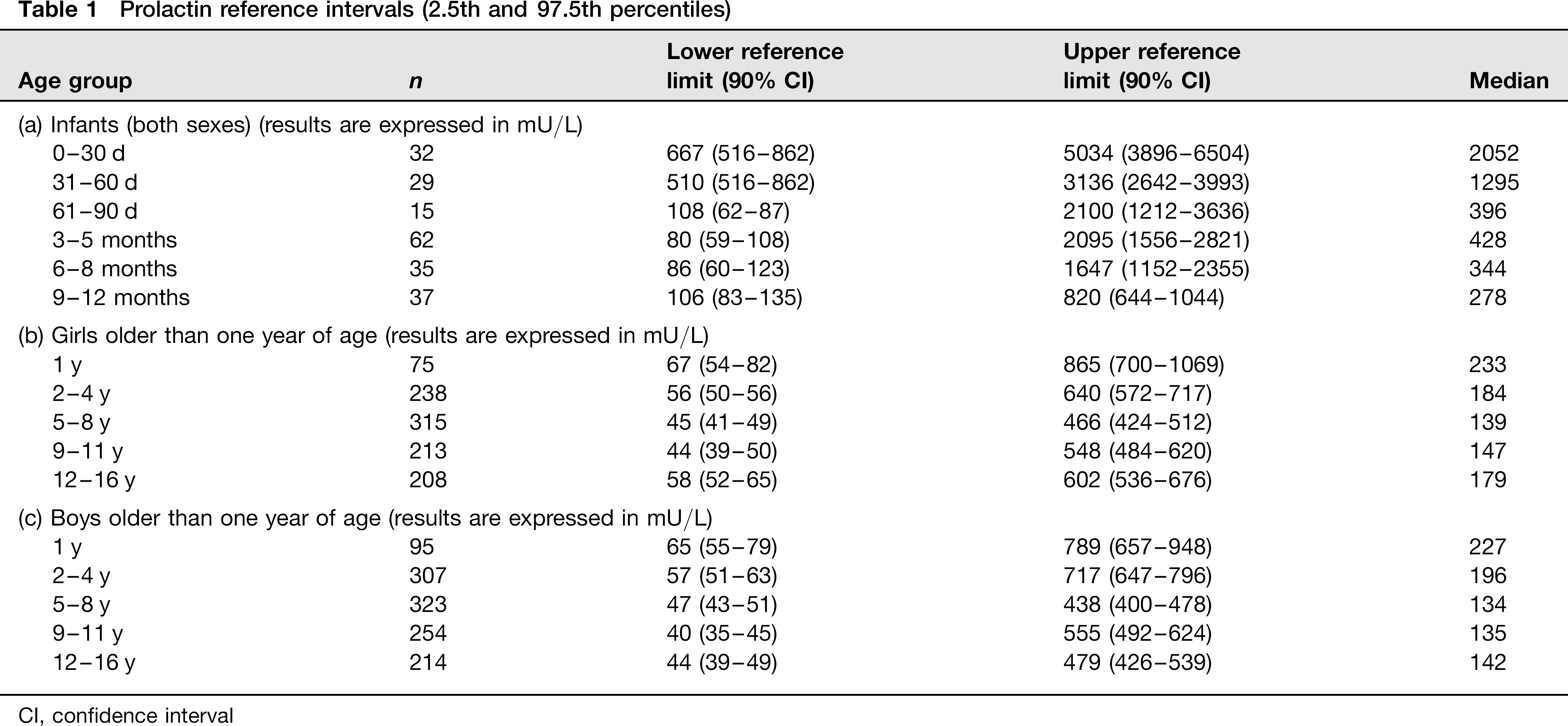
CI, confidence interval
Results
The prolactin results for all age and sex stratifications showed non-Gaussian distributions (P < 0.0001). There were significant age-related differences for prolactin during the first year of life with prolactin declining sharply during the first year of life and continuing to decline until aged five years when the levels stabilized. The sex differences were found to be small until aged 13-17 years when prolactin was demonstrated to be higher in girls than boys.
Discussion
This is the first published study to report age-related prolactin reference intervals spanning all paediatric age groups where the data are age partitioned in infants under one year using an ‘indirect’ approach’. The sample reference group represents a cohort of infants and children, which was under clinical investigation in a tertiary care setting. The results are generally similarly to those published by Cook using the Hybritech Tandem prolactin procedure, 11 who also showed that high concentrations after birth declined to adult concentrations by nine years of age but then increasing during puberty. Data provided by the prolactin kit manufacturer only provided a reference interval up to 15 days of age and then from one year to 18 years. 12 The prolactin reference intervals were higher than in our study during the first 15 days of life but were lower in the one year to 18 years age groups. The reference intervals produced by Cook were derived from hospitalized children in Washington DC, USA, and those quoted by the kit manufacturer were derived from healthy German children. Although most prolactin assays are now standardized to the third IRP 84/500, there are significant differences between methods. These variables may account for the differences between published intervals.
In this study, although prolactin results were excluded if the clinical information (including drug history) given about the patient increased the likelihood of a raised prolactin, it was not possible to completely exclude the possibility that a small number of patients may have had conditions or may have been taking medication known to cause hyper-prolactinaemia. Nor was it not possible to control the prea-nalytical variables such as stress, time of specimen collection and specimen handling.
It is vital that reference intervals used to interpret laboratory data are appropriate for the method used and population under investigation. This latter point is of particular importance for paediatric referral centres where patients with suspected neurometabolic disorders are regularly investigated. Consequently, we would recommend that such hospitals adopt a similar approach to that reported here with regards to assessment of serum prolactin.
Serum prolactin is a cheap and readily available test in most diagnostic clinical biochemistry laboratories with a same day turnaround time. It is envisaged that these age-and sex-related reference intervals will be used to aid in the selection of patients presenting with features of central dopamine deficiency for LP. These reference intervals also have the potential for treatment monitoring of patients with diagnoses of inborn errors of dopamine biosynthesis.
