Abstract
Background
Metabolites of ethylene glycol (EG) can cross-react in L-lactate assays, leading to falsely elevated serum lactate levels in case of EG intoxication. In this study, we evaluated the effects of EG and its metabolites on routinely used lactate measuring methods.
Methods
Serum aliquots were spiked with either L-lactate, EG or one of its metabolites (all 12.5 mmol/L): glyoxal, glycolate, glyoxylic acid or oxalate. An unspiked sample (L-lactate 2.6 mmol/L) served as a control. L-Lactate levels in these samples were measured in 31 national hospitals on 20 different analysers from nine manufacturers.
Results
The L-lactate concentrations in the control sample and in the samples spiked with L-lactate, EG, glyoxal and oxalate provided correct results by all routinely used methods. However, the glycolate and glyoxylic acid spiked samples resulted in falsely elevated L-lactate concentration with all blood gas methods and with the majority of general chemistry methods using L-lactate oxidase.
Conclusion
The EG metabolites glycolate and glyoxylic acid were shown to falsely elevate L-lactate results with most of the currently used methods due to cross-reactivity with the oxidase enzyme. Falsely elevated L-lactate results can lead to misdiagnosis and inappropriate management of patients with EG intoxication.
Introduction
Ethylene glycol (EG) intoxication, as occurs with ingestion of antifreeze, is a common cause of pseudo hyperlactataemia. 1 Although EG itself is rather non-toxic, EG poisoning is characterized by profound metabolic acidosis, cardiopulmonary events and acute renal failure, and is lethal if left untreated. 2 In the liver, EG is metabolized by alcohol dehydrogenase to glyoxal, which, in turn, is metabolized to glycolate, glyoxylic acid and oxalate. Glycolate induces severe metabolic acidosis, while oxalate induces tubular necrosis resulting in renal failure. 2
In addition, the molecular structure of glycolate shares similarities with that of L-lactate. Consequently, elevated glycolate levels may interfere with the enzymatic measurement of L-lactate, resulting in falsely elevated lactate concentrations. Although the interference of glycolate with L-lactate measurement has been reported previously,3,4 it is still unclear which analysers and methods used for L-lactate measurements show this interference. Therefore, in the present study, the effects of EG metabolites on L-lactate measurements were investigated in a multianalyser survey.
Materials and methods
The interference of EG metabolites on L-lactate measurements was assessed using a multicenter, multianalyser approach. A total of 31 national medical centres with 20 different analysers (chemistry analysers, blood gas analysers and point-of-care testing analysers [POCTs]) from nine different manufacturers (Roche, Basel, Switzerland; Radiometer, Brønshøj, Denmark; Siemens, Marburg, Germany; Abbott, North Chicago, IL, USA; Olympus, Center Valley, PA, USA; Beckman Coulter, Brea, CA, USA; Vitalab, Spankeren, The Netherlands; Instrumentation Laboratory, Bedford, MA, USA and Vitros, Raritan, NJ, USA) participated in the survey (Figure 1).
L-Lactate concentrations in the presence of ethylene glycol metabolites glycolate or glyoxylic acid (both 12.5 mmol/L) as measured on different analysers. A reference sample with a similar concentration of L-lactate (2.6 mmol/L) without the addition of glycolate or glyoxylic acid served as the control. The method is displayed in bold italics, (A)mperometric or (C)olorimetric; microbial origin of the enzyme is displayed in italics. 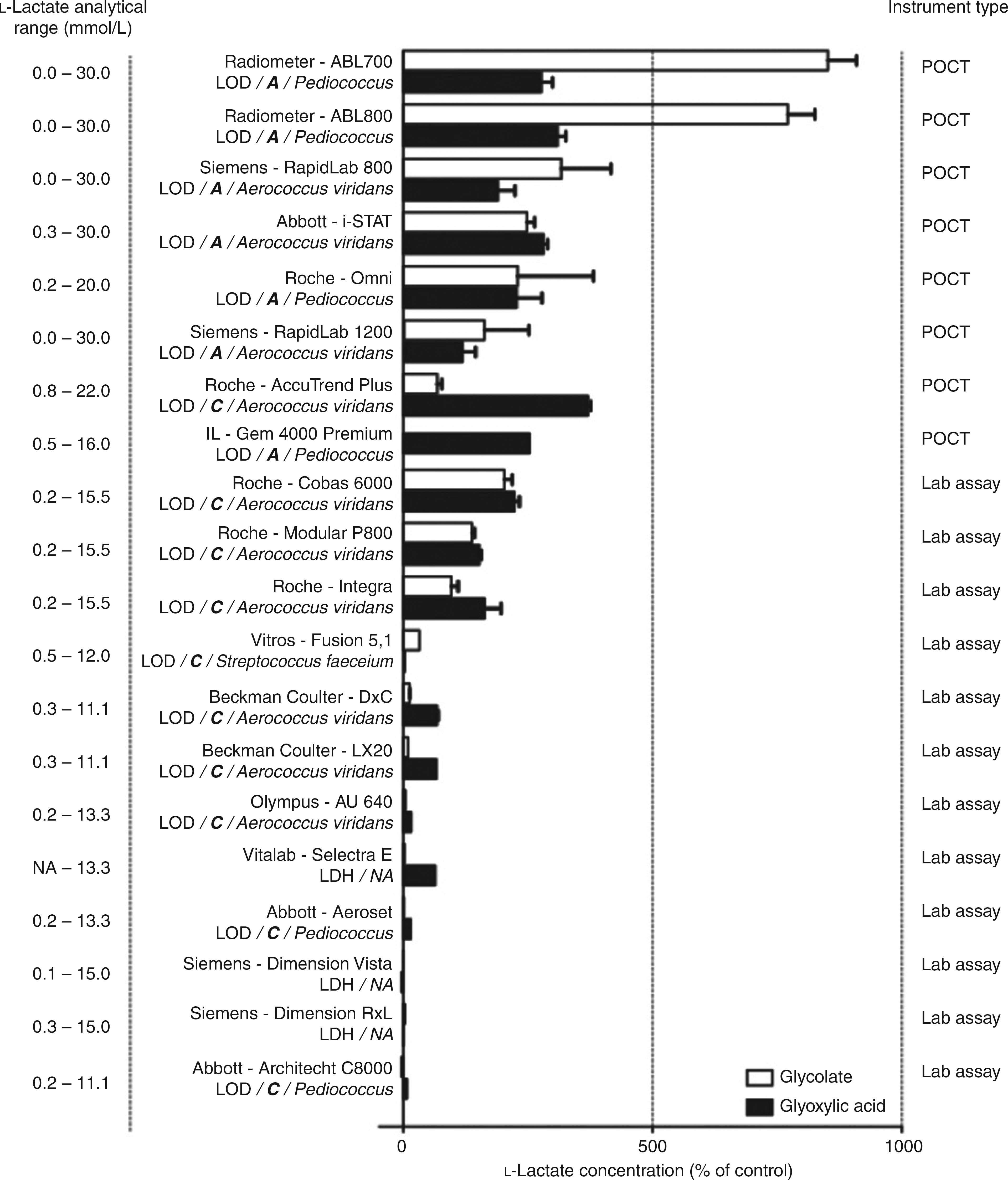
Patient serum samples were pooled and divided in seven aliquots. One served as a control (L-lactate 2.6 mmol/L). The other six aliquots were spiked with L-lactate, EG or one of the four most important EG metabolites: glyoxal, glycolate, glyoxylic acid or oxalate (all Sigma-Aldrich, St Louis, MO, USA; 12.5 mmol/L each). This concentration of EG and its metabolites is consistent with a modest ingestion of EG. 5 Each of the 31 participating hospitals received one set of seven blinded samples (1 mL per aliquot, sent at room temperature). The stability of the compounds at room temperature and post freeze-thaw (3 × during three days) was confirmed in a separate study (data not shown). The L-lactate concentrations in the seven samples were measured on the available analysers in all participating centres.
Results and discussion
L-Lactate concentrations in the control samples and in the samples spiked with L-lactate, EG, glyoxal and oxalate were measured correctly by all analysers. In contrast, the vast majority of the tested analysers reported falsely elevated L-lactate concentrations to varying degrees in the samples spiked with glyoxylic acid or glycolate (Figure 1).
L-Lactate analysis is based on the formation of pyruvate, which is catalysed by either lactate oxidase (LOD) to form H2O2 or by lactate dehydrogenase (LDH) to form NADH. Falsely elevated L-lactate levels were observed in LOD but not in LDH-based assays, suggesting that the interference is related to LOD. The similarities in molecular structures of glycolate and lactate may cause cross-reaction of glycolate with LOD, resulting in falsely elevated lactate levels in the presence of glycolate. In general, the H2O2 formed by LOD is measured by colorimetry in routine chemistry analysers, or by amperometry in POCTs and blood gas analysers. For unknown reasons, the amperometric methods experienced more interference from glycolate and glyoxylic acid than the colorimetric assays using LOD. In addition to the detection method, the microbial origin of the LOD differed between the tested analysers, including Pediococcus sp., Aerococcus viridans and Streptococcus faeceium. The combination of colorimetric methods with LOD originating from Pediococcus sp. revealed no interference with L-lactate measurement by either glycolate or glyoxylic acid. In contrast, lactate measurements were influenced by glycolate or glyoxylic acid in analysers using colorimetric assays with LOD from A. viridans or those based on amperometric methods with LOD from Pediococcus sp. Hence, these assays should not be used for the measurement of L-lactate when EG intoxication is suspected.
In addition to the microbial origin of LOD and the detection method, L-lactate measurements may be affected by other variables such as incubation time, sample volume and wavelength, which should be taken into consideration when developing an L-lactate assay. It was beyond the scope of the current report to investigate the background of the different assays of each manufacturer as this is often considered a company secret.
In conclusion, to avoid an erroneous diagnosis of hyperlactataemia in patients with EG intoxication, it is recommended to use an interference-free L-lactate assay based on either LDH or a colorimetric assay based on LOD originating from Pediococcus sp.
Declarations
Footnotes
Acknowledgements:
We thank all participating hospitals.
