Abstract
Background
Chronic cutaneous complications such as pruritus are among the very frequent complaints of sulphur mustard (SM)-exposed patients. The present trial investigated the impact of curcumin on serum inflammatory biomarkers and their association with pruritus severity and quality of life (QoL).
Methods
This was a randomized, double-blind trial among 96 male Iranian veterans (age 37–59 y) who were suffering from chronic SM-induced pruritic skin lesions. Patients were randomly assigned to curcumin (1 g/d, n = 46) or placebo (n = 50) for four weeks. Serum concentrations of interleukins 6 (IL-6) and 8 (IL-8) together with high-sensitivity C-reactive protein (hs-CRP) and calcitonin gene-related peptide (CGRP) were measured at baseline and at the end of the trial. Assessment of pruritus severity was performed using the pruritus score and QoL using the Dermatology Life Quality Index (DLQI).
Results
Serum IL-8 and hs-CRP were significantly reduced in both groups but the magnitude of reduction was greater in the curcumin group (P < 0.001). Serum CGRP was only decreased in the curcumin group (P < 0.001). No significant change was observed in serum IL-6. There were significant correlations between CGRP and IL-6 changes (P = 0.011) and between DLQI and IL-8 changes (P = 0.026) in the curcumin group. In the curcumin group, changes in serum IL-8 concentrations were found as the significant predictor of DLQI scores (P = 0.026) but none of the independent variables could predict pruritus scores.
Conclusions
Curcumin supplementation effectively mitigates inflammation in patients suffering from chronic SM-induced cutaneous complications. This anti-inflammatory effect might account for the observed pruritus alleviation and QoL improvement by this phytochemical.
Introduction
Sulphur mustard (C4H8Cl2S; 2,2-dichlorethyl sulphide; SM), is a toxic chemical warfare agent with potent alkylating and blistering properties. Since its discovery, SM has been extensively used in military conflicts. The last military deployment of SM dates back to the Iraq–Iran war (1983–1988), in which the Iraqi army repeatedly used this agent against Iranian soldiers and civilians. 1 It is estimated that over 100,000 Iranians have been exposed and chemically injured with SM, of whom about one-third currently suffer from chronic complications. 2 Although the pathophysiology of SM toxicity has not been fully clarified, DNA damage and resulting inflammatory and oxidative responses are considered as cornerstones of pathological effects. 3,4 Along with lungs and eyes, skin is among the most sensitive organs to SM that could undergo several types of damage based on the SM dose and duration of exposure. 3,5 Cutaneous complications of SM have been chronologically classified into acute and chronic. Chronic complications of SM are present even 20 y after exposure, and have been frequently reported in Iranian intoxicated patients. These complications include pruritus, xerosis, pigmentation disorders, pain, erythema, scarring and burning sensation. 6–8 Among these symptoms, pruritus has been reported to be the most common complaint, being present in about 70–90% of intoxicated patients. 9 In a recent study, Panahi et al. 10 found a significant negative impact of pruritus on the quality of life (QoL) of chemically injured patients who suffer from chronic SM-induced complications. However, current available medications for SM-induced pruritus are limited and mostly based on symptomatic therapy with topical corticosteroids. On the other hand, long-term application of topical corticosteroids is associated with significant adverse effects including steroid atrophy, rosaceae, striae, telangiectasis and increased bruisability. 11
One promising candidate to combat chronic complications of SM is the dietary polyphenol, curcumin. Curcumin [1,7-bis(4-hydroxy-3-methoxyphenyl) hepta-1,6-diene-3,5-dione; diferuloyl methane] is the yellow pigment and major bioactive constituent in the dried rhizomes of Curcuma longa L. (turmeric). There is ample evidence indicating a wide variety of biological and pharmacological activities for curcumin including anti-inflammatory, antioxidant, cardio- and neuroprotective, chemopreventive, anticancer and many more effects. 12,13 One key advantage of curcumin is its safety and tolerability. It has been shown that oral doses of curcumin up to 12 g/d are well tolerated in humans. 14
Owing to the pivotal contribution of inflammation and oxidative stress to the pathogenesis of SM-induced cutaneous complications, safe medications with dual anti-inflammatory and antioxidant activity would be highly desirable for the management of symptoms. In a recent trial, we reported remarkable improvement of pruritus, QoL and antioxidant status following supplementation with curcumin. 15 However, it remains unclear whether mitigation of systemic inflammation by curcumin has a role in the positive observed effects. Hence, the present study investigated the impact of curcumin on serum inflammatory biomarkers and their association with pruritus severity and QoL.
Methods
Participants
This study was a randomized, double-blind and placebo-controlled clinical trial, performed in the Veterans Clinic of Tehran and Ilam (west of Iran). 16 Included subjects were male Iranian veterans of the Iraq–Iran war (age range: 37–59 y) who were suffering from chronic pruritic skin lesions. The exposure was confirmed by documented development of blisters in the exposed areas of the skin and transient visual deterioration lasting for several days and associated respiratory symptoms. Patients were excluded if itching resulted from systemic or cutaneous non-chemical diseases, or if they had received any topical treatments within one month prior to the study.
Included patients (n = 96) were randomized to receive curcumin (1 g/d) (curcumin group; n = 46) or matched placebo (placebo group; n = 50) for a period of four weeks. The groups were matched regarding age (47.5 ± 10.7 and 48.3 ± 8.5 in the curcumin and placebo group, respectively). Curcumin was administered in the form of C3 Complex® capsules (Sami Labs LTD, Bangalore, Karnataka, India) containing 500 mg curcuminoids plus 5 mg bioperine®. Bioperine® is an extract obtained from black pepper (Piper nigrum L.) or long pepper (Piper longum L.), and contains 95% piperine which is a well-documented bioavailability enhancer. Placebo capsules used in the study were shape- and size-matched, and contained piperine (5 mg).
Patients were visited by a board-certified dermatologist at baseline and at the end of treatment duration. This study was conducted according to the guidelines laid down in the Declaration of Helsinki. The study protocol was approved by the Ethics Committee of the Baqiyatallah University of Medical Sciences and written informed consent was obtained from participants.
Assessment of pruritus severity
Calculation details of pruritus score
*Maximum obtainable score
Assessment of QoL
QoL was evaluated using the Dermatology Life Quality Index (DLQI). DLQI has been previously employed in Iranian patients with SM-induced chronic skin lesions. 10 This index consists of 10 questions under six headings: symptoms and feelings (questions 1 and 2), daily activities (questions 3 and 4), leisure (questions 5 and 6), work and school (question 7), personal relationships (questions 8 and 9) and treatment (question 10). Each question has a maximum score of 3 and options of ‘very much’ (scored 3), ‘a lot’ (scored 2), ‘a little’ (scored 1) and ‘not at all’ (scored 0). In addition, a ‘0’ score is allotted to ‘not relevant’ response and unanswered questions. DLQI total score is calculated by summing the scores of all questions, resulting in a scoring range of 0–30 in which higher scores are associated with more severe impairments of QoL.
Biochemical analyses
Blood samples were collected at baseline and at the end of the trial. Collected samples were centrifuged at 750
Statistical analyses
Statistical analyses were performed using SPSS software for Windows (version 11.5; SPSS Inc., Chicago, IL, USA). Data were expressed as mean ± SD. Data were assessed for normality using the Kolmogorov–Smirnov test. Group comparisons were made using independent samples t-test and paired samples t-test for normally distributed data, or Mann-Whitney U test and Wilcoxon signed-rank test for non-normally distributed data. Categorical variables were compared using chi-square or Fisher's exact test. Bivariate correlations between different parameters were assessed using Pearson's (for normally distributed data) or Spearman's (for non-normally distributed data) rank correlation coefficients in the curcumin and placebo groups. Stepwise multiple linear regression analysis was used to identify the independent parameters that were related to the evaluated efficacy measures. A two-tailed P value of <0.05 was considered to be statistically significant.
Results
Ninety-six patients entered the study, of whom 80 patients completed the study (n = 40 in each group) and were included in the final analyses. Six patients discontinued study treatment in the curcumin group (three because of gastrointestinal side-effects, one because of fear of side-effects, one because of inability to return to the clinic because of the long distance and one due to unknown reason), and ten in the placebo group (seven because of lack of treatment response or worsening of symptoms, two because of gastrointestinal side-effects and one because of inability to return to the clinic because of the long distance) (Figure 1). There was no significant difference in the dropout rate between the groups (P > 0.05; chi-square test).
Flowchart of the trial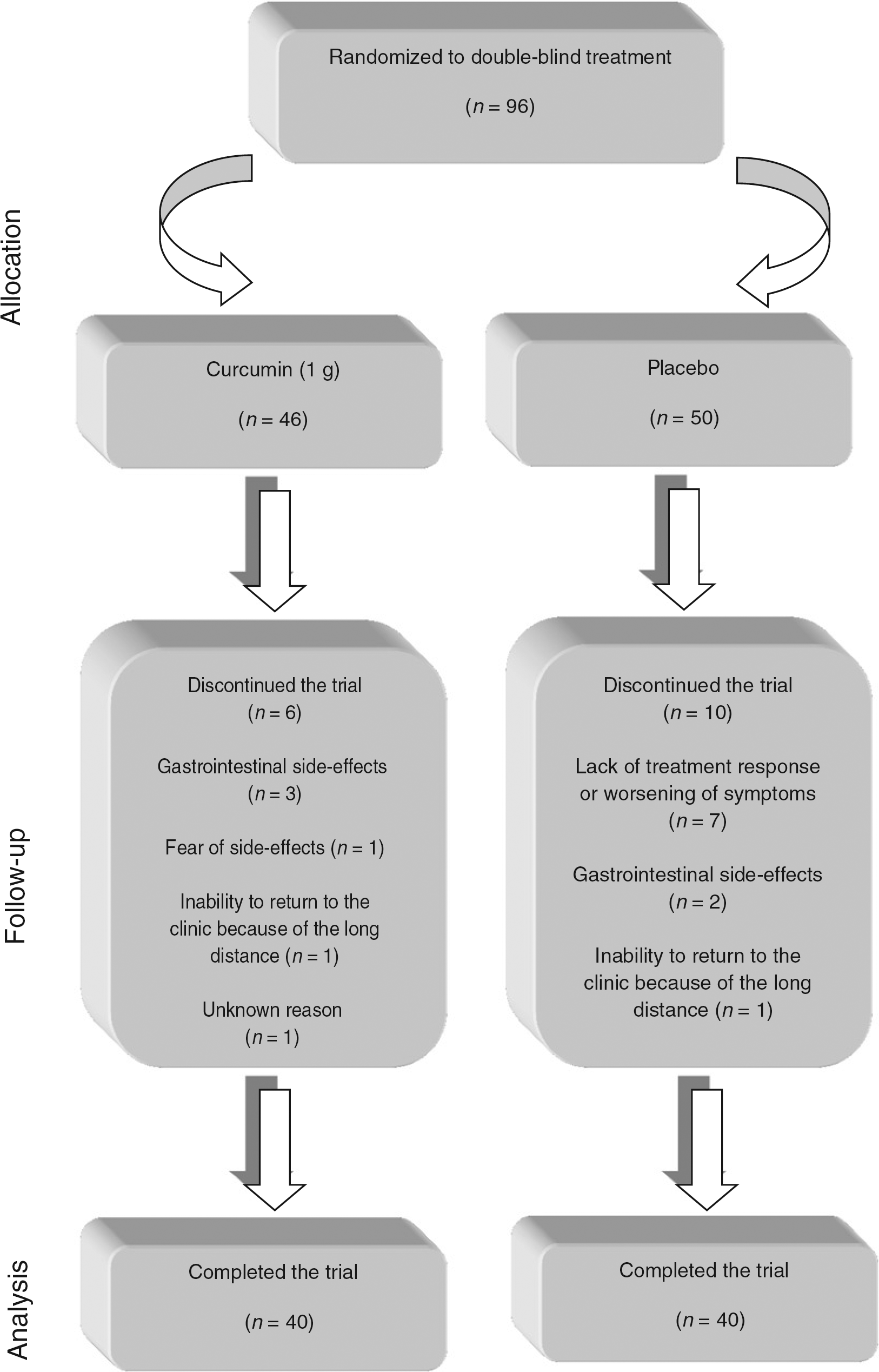
Effect of curcumin supplementation on serum IL-6
Within-group comparison of the evaluated biochemical parameters and efficacy measures
Values are expressed as mean ± SD
IL, interleukin; CGRP, calcitonin gene-related peptide; Hs-CRP, high-sensitivity C-reactive protein; PS, pruritus score; DLQI, Dermatology Life Quality Index
Between-group comparison of the evaluated biochemical parameters and efficacy measures
Values are expressed as mean ± SD
IL, interleukin; CGRP, calcitonin gene-related peptide; Hs-CRP, high-sensitivity C-reactive protein; PS, pruritus score; DLQI, Dermatology Life Quality Index
Effect of curcumin supplementation on serum IL-8
Baseline serum IL-8 concentrations were significantly higher in the curcumin compared with the placebo group (P = 0.001; Mann-Whitney U test). Serum IL-8 concentrations were significantly decreased in both the curcumin (P < 0.001; Wilcoxon signed-rank test) and placebo (P < 0.001; paired samples t-test) groups. However, the magnitude of serum IL-8 reduction was remarkably greater in the curcumin group (P < 0.001; independent samples t-test) (Tables 2 and 3).
Effect of curcumin supplementation on serum CGRP
Baseline serum CGRP levels were comparable between the curcumin and placebo groups (P > 0.05; independent samples t-test). Curcumin supplementation caused a marked decrease in serum CGRP (P < 0.001; paired samples t-test) while there was no significant difference in the placebo group (P > 0.05; paired samples t-test). With respect to the magnitude of changes, the extent of reduction was greater in the curcumin versus placebo group (P < 0.001; independent samples t-test) (Tables 2 and 3).
Effect of curcumin supplementation on serum hs-CRP
There was no significant difference in baseline serum hs-CRP concentrations between the groups (P > 0.05; independent samples t-test). Serum hs-CRP was reduced by the end of the trial in both the curcumin and placebo groups (P < 0.001; paired samples t-test). Nevertheless, the magnitude of reduction was significantly greater in the curcumin group (P < 0.001; independent samples t-test) (Tables 2 and 3).
Effect of curcumin supplementation on pruritus score
Pruritus scores were comparable between the curcumin and placebo groups at baseline (P > 0.05; independent samples t-test). Supplementation with curcumin was associated with a marked decline in pruritus score (P < 0.001; paired samples t-test). In contrast, no significant difference was observed in the placebo group (P > 0.05; paired samples t-test). In the same manner, change in pruritus score was significantly greater in the curcumin versus placebo group (P < 0.001; independent samples t-test) (Tables 2 and 3).
Effect of curcumin supplementation on QoL
Comparison of baseline DLQI scores did not indicate any significant difference between the groups (P > 0.05; Mann-Whitney U test). DLQI was significantly decreased by the end of the trial in both the curcumin (P < 0.001; Wilcoxon signed-rank test) and placebo (P = 0.003; paired samples t-test) groups. However, the rate of reduction was greater in the curcumin versus placebo group (P < 0.001; independent samples t-test) (Tables 2 and 3).
Bivariate analysis
At baseline, there were significant correlations between serum IL-6 concentrations and CGRP (Pearson's r = 0.418; P = 0.007) as well as pruritus score (Pearson's r = −0.328; P = 0.039), and between DLQI and pruritus score (Spearman's r = 0.316; P = 0.047) in the curcumin group while no significant correlation was found in the placebo group. As for the post-trial values, serum IL-6 concentrations were significantly correlated with serum CGRP (Pearson's r = 0.400; P = 0.011) in the curcumin group. There was also a significant inverse correlation between pruritus and DLQI scores (Pearson's r = −0.404; P = 0.010) in the curcumin group. The only significant correlation that was observed in the placebo group was between serum IL-8 and pruritus score (Pearson's r = −0.363; P = 0.021). Bivariate associations were also checked for the changes in the evaluated biochemical and efficacy measures. Only two significant correlations were found in the curcumin group: between CGRP and IL-6 changes (Pearson's r = 0.397; P = 0.011) and between DLQI and IL-8 changes (Pearson's r = 0.351; P = 0.026). Correlation graphs are illustrated in Figure 2.
Significant correlations between the evaluated efficacy measures. CGRP, calcitonin gene-related peptide; DLQI, Dermatology Life Quality Index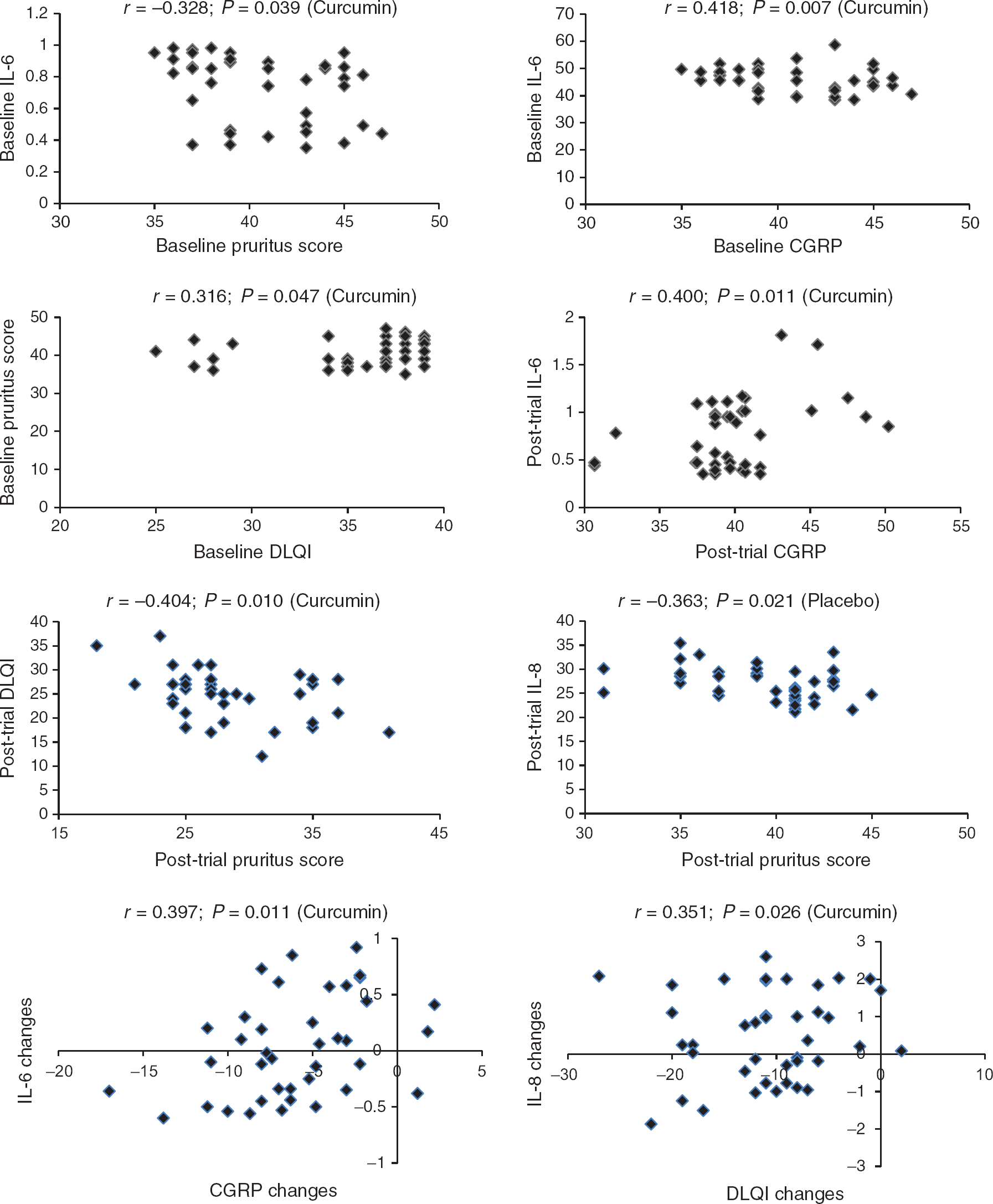
Multiple regression analysis
Stepwise multiple regression analysis was used to assess the impact of predictor variables on each of the employed efficacy measures. Changes (pretrial versus post-trial) in pruritus score and DLQI score were separately entered into the model as dependent variable. Predictor variables included changes in serum IL-6, IL-8, CGRP and hs-CRP concentrations. In the curcumin group, changes in serum IL-8 concentrations were found as the significant predictor of DLQI scores (B = 0.437; odds ratio [95% CI] = 0.351 [0.054–0.819]; P = 0.026) but none of the independent variables could predict pruritus scores. No significant determinant was found in the placebo group, neither for DLQI nor pruritus scores.
Discussion
A summary of previous reports on the up-regulation of inflammatory biomarkers by sulphur mustard
MIP, macrophage inflammatory protein; MMCP, mouse mast cell protease; TNF, tumour necrosis factor; MCP, monocyte chemotactic protein; GM-CSF, granulocyte-macrophage colony-stimulating factor; IL, interleukin; IFN, interferon
In the current research, a remarkable reduction of serum IL-8 was observed from curcumin that was significantly higher than that in the placebo group. This is consistent with previous findings on the effects of curcumin in other inflammatory disease states. 12,18 An important mechanism for the down-regulating effects of curcumin on proinflammatory cytokines is suppression of NF-κB by this phytonutrient. 19,20
Further to cytokines, elevated circulating levels of hs-CRP have been reported in patients with chronic SM complications, 21 which is indicative of a heightened burden of systemic inflammation in these patients. Hs-CRP has been documented as one of the most sensitive surrogate markers of systemic inflammation in a number of diseases, including those concerning dermatology such as psoriasis vulgaris, 22 urticaria, 23 Behçet's disease, 24 papillomatosis 25 and uraemic pruritus. 26 There is both human and animal evidence indicating CRP-lowering activity of curcumin. In a clinical trial in osteoarthritis patients, administration of a phospholipidated formulation of curcumin (equivalent to 200 mg curcumin/d) for three months was associated with a significant reduction of plasma CRP concentrations. 27 Likewise, curcumin supplementation has been reported to exert the same effect on hs-CRP in low-density lipoprotein receptor-deficient (LDLR −/−) mice fed with a high-cholesterol diet 28 as well as lead-intoxicated rats 29 and weaned piglets. 30
Another important finding of the present study was effective reduction of serum CGRP following curcumin supplementation. CGRP has been previously shown to be involved in nociception and pruritus. CGRP is expressed by keratinocytes and its levels are increased in human chronic pain or chronic pruritus conditions such as atopic dermatitis and pruritus. 31,32 CGRP also promotes inflammation through up-regulation of IL-1 and IL-8 and endothelial-leukocyte adhesion molecule 1, chemotaxis of neutrophils and translocation of P-selectin. 33,34 Regarding the contribution of this mediator to the perception of both pain and pruritus, its reduction by curcumin could represent a plausible mechanism for the promising results observed in the present study. Thus far, no study has investigated serum CGRP status and its modulation in patients suffering from chronic complications of SM. This is also the first report on the impact of curcumin on CGRP levels.
Aside from inflammation, several other mechanisms have been suggested to be implicated in the pathogenesis of SM-induced dermatotoxicity. The most important of these mechanisms include induction of DNA damage, matrix metalloproteinases, poly (ADP-ribose) polymerase signalling, nitric oxide, calcium signalling, apoptosis and oxidative stress. 17 Interestingly, there is evidence indicating the beneficial impact of curcumin on nearly all of these biochemical abnormalities. 12 For instance, curcumin not only inhibits cyclooxygenase-2, lipoxygenase and inducible nitric oxide synthase but also induces heat shock proteins that confer cytoprotection against different stimuli. 35,36 In addition, curcumin possesses well-established antioxidant and free radical scavenging activity which enables it for effective prevention of membrane lipid peroxidation as well as oxidative DNA damage and resulting genotoxicity. 37 We have previously shown in the same patients that curcumin supplementation efficiently enhances serum levels of three important enzymatic antioxidants, namely superoxide dismutase, glutathione peroxidase and catalase. 15 In light of the present findings, the efficient anti-pruritic effects and improvement of QoL by curcumin could be attributed to both antioxidant and anti-inflammatory functions of this compound.
The current findings add to a growing body of literature on the beneficial effects of curcumin in dermatological disorders. Previously, several encouraging studies have shown the efficacy of curcumin therapy in the promotion of wound healing and protection against psoriasis, scleroderma and skin cancer. 38 However, large randomized controlled trials could provide more definitive evidence using additional biomarkers of inflammation. Besides, future investigation and experimentation is warranted to clarify whether the therapeutic effects of curcumin on SM-induced cutaneous complications are enhanced at higher doses of the phytochemical. Finally, it is strongly recommended that further research be undertaken to explore the anti-inflammatory and antioxidant effects of curcumin in the amelioration of pulmonary and ocular complications of SM-exposed patients.
DECLARATIONS
