Abstract
Background
Measured unbound bilirubin concentration is influenced by bilirubin photoisomers. Bilirubin photoisomers are produced even with only a slight light exposure, and clinical samples are inevitably exposed to light. The objective of the study was to evaluate the influence of bilirubin photoisomers on the measurement of unbound bilirubin using serum of jaundiced neonates during blue light phototherapy.
Methods
Five neonates treated with phototherapy for hyperbilirubinaemia were enrolled. The samples were taken 12 h after initiation of phototherapy. Samples were processed by irradiation with blue light, by indoor ceiling light, by both blue light and indoor ceiling light or shaded. Bilirubin subfractions, total bilirubin and unbound bilirubin were measured.
Results
Compared with the non-irradiated samples, the (EZ)-cyclobilirubin concentration and (ZE)-bilirubin/(ZZ)-bilirubin ratio significantly increased in the blue light-irradiated samples, the (ZE)-bilirubin/(ZZ)-bilirubin ratio significantly increased in the indoor ceiling light-irradiated samples, and the (EZ)-cyclobilirubin, (EZ)-bilirubin and (ZE)-bilirubin/(ZZ)-bilirubin ratio significantly increased in the samples irradiated with both lights. No change was noted in unbound bilirubin in any group.
Conclusions
We consider that changes in bilirubin photoisomers induced by light exposure during clinical practice do not influence the measured unbound bilirubin concentration.
Introduction
Unbound bilirubin is unconjugated bilirubin which does not bind to human serum albumin (HSA) or other proteins, and is a useful marker in addition to total bilirubin for evaluating bilirubin neurotoxicity. 1,2 Unbound bilirubin is measured automatically by glucose oxidase and peroxidase with dilution in a UB-Analyzer (UA-2; Arrows Co Ltd, Osaka, Japan), which is widely used in Japan among other places. The equilibrium unbound bilirubin measured by this method was lower 3,4 than unbound bilirubin measured by the two-fold sample dilution method, but it significantly correlated with it. 4 Also, unbound bilirubin measured by this method is influenced by bilirubin photoisomers. 5–7 In spite of these limitations, unbound bilirubin measured by this method has correlated with various indicators of bilirubin toxicity. 1,2,8–11
The serum (EZ)- and (EE)-cyclobilirubin concentrations in neonatal hyperbilirubinaemia were very low even during phototherapy, but the unbound bilirubin increased when (EZ)- and (EE)-cyclobilirubin increased considerably in our previous studies. 5,6
(ZE)-bilirubin is a photoisomer abundantly present in serum exposed to indoor ceiling light. According to the photochemical characteristic of (ZE)-bilirubin, (ZE)-bilirubin attains photoequilibrium with (ZZ)-bilirubin during short-time light exposure.
12
The (ZE)-bilirubin/(ZZ)-bilirubin ratio is determined by the type of light source, but it is different between bilirubin irradiated with the same light source in blood and serum.
13
Itoh
In clinical practice, light exposure of samples from a jaundiced neonate occurs unintentionally since the samples are usually obtained and analysed in a lighted room. They will contain varying degrees of bilirubin photoisomers; certainly not zero.
This study was carried out in order to evaluate the influence of bilirubin photoisomers on unbound bilirubin measurement under the various photoisomers condition. Clinical samples were used to evaluate the degree of influences on measurement values in a state as close as possible to the clinical settings.
Materials and methods
Five neonates with hyperbilirubinaemia treated with phototherapy were enrolled in this study. They were born in Handa Hospital between February and April 2009. Their mean gestational age was 40 weeks (range 39.4–40.7 weeks), mean birth weight was 3274 g (range 2864–3762 g) and median 5-min Apgar score was 10 (range 8–10). They did not have haemolytic anaemia, obstructive jaundice or other anomalies.
We obtained informed consent from all their parents. They received blue light phototherapy using six fluorescent blue lights (FL20SBW-NU; Atom Medical Co., Tokyo, Japan) with a light intensity of 9.25
We collected 0.5 mL blood from patients for this study at 12 h when performing routine total bilirubin measurement after starting phototherapy. For sample collection, all indoor lighting was turned off. Serum was separated by centrifugation (1000 rpm for 10 min) under dark conditions and was divided in half into tubes protected by aluminum foil. One sample was irradiated by the same blue light for one minute with a light intensity of 9.25 Sample processing
We analysed total bilirubin and unbound bilirubin with a UB-Analyzer (UA-2; Arrows Co Ltd)
14
in the dark room under red light. In this method, 20
Statistical analysis
The two-way analysis of variance (ANOVA) was used for comparison among groups.
Results
The photoisomers, total bilirubin and unbound bilirubin concentration are shown in Table 1 and Figure 2. Significant between-group differences were noted in (EZ)-cyclobilirubin ( Comparison of effect of irradiation on (EE)- and (EZ)-cyclobilirubin, the (ZE)-bilirubin/(ZZ)-bilirubin ratio and unbound bilirubin. The bars indicate the mean value. Significant between-group differences were noted in (EE)- and (EZ)-cyclobilirubin ( Comparison of effect of different irradiation schemes on bilirubin subfractions, total serum bilirubin and unbound bilirubin Total bilirubin and unbound bilirubin was measured with a UB-analyzer (UA-2) (1:50 sample dilution). Values are mean (SD) and range (minimum–maximum). Blue light irradiation to sample for one minute, indoor ceiling light irradiation to sample for 10 minutes. The two-way analysis of variance was used for comparison among groups *Significant difference from no irradiation
†Significant difference from blue light irradiation and from both light irradiation (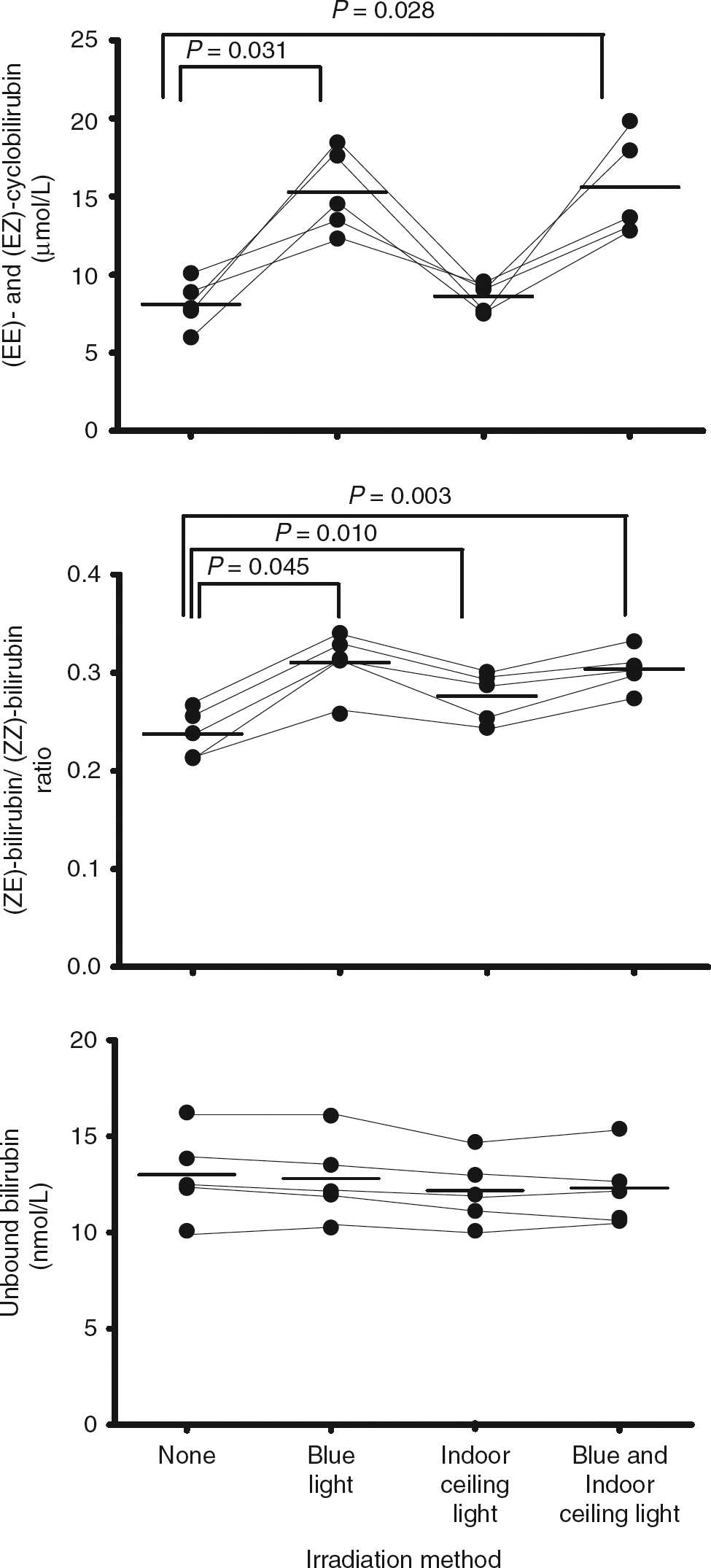
Discussion
To avoid bilirubin toxicity, neonates with hyperbilirubinaemia are treated by phototherapy. The mechanism of phototherapy for neonatal hyperbilirubinaemia involves the production and excretion of (ZE)-bilirubin and (EZ)- and (EE)-cyclobilirubin. 17
The most effective wavelength within 400–520 nm of visible light
For management of the jaundiced neonate, serum total bilirubin, measurements of serum unbound bilirubin and cutaneous bilirubin are used. A semi-automated instrument for measuring the unbound bilirubin with dilution-peroxidase method is available. A problem with this measurement method depends on the apparent binding affinity of albumin due to sample dilution. 20 In clinical studies, unbound bilirubin determined by this measurement method was correlated with auditory brainstem response (ABR) changes in term infants 1,2 as well as the probability of bilateral refer automated ABR with jaundiced newborns at more than 34 weeks of gestation. 11 In premature infants, unbound bilirubin correlated with abnormal ABR maturation 8 and kernicterus. 9 Unbound bilirubin of 17.1 nmol/L carries a risk of kernicterus with sensitivity of 100% and specificity of 98% in low-birth-weight infants, while unbound bilirubin of 13.7 nmol/L has a sensitivity of 100% and specificity of 96% in very low-birth-weight infants. 10
For evaluation of these factors, it is necessary to accurately measure unbound bilirubin. Haemoglobin,
14
vitamin C, conjugated bilirubin,
6
bilirubin photoisomers
5–7
and sample dilution
3,4,21
influence the measurement of the unbound bilirubin by the peroxidase method. The most serious error is the failure to appreciate that the unbound bilirubin determined by the peroxidase test is steady-state unbound bilirubin.
22
Bilirubin binds in the large hydrophobic cavity of sub-domain IIA on HSA.
23
The more bilirubin that binds to the first class of bilirubin sites in the HSA molecule, the more readily bilirubin reaches a condition of photoequilibrium with (ZE)-bilirubin. (ZE)-Bilirubin IX
Since Itoh
We conclude that major changes in the proportions of the different photoisomers produced by the three different irradiation schemes produced no alterations in measurement of unbound bilirubin with the UB-analyser (UA-2); unbound bilirubin values that are unchanged are unlikely to be clinically significant, but this needs to be confirmed by prospective studies of outcomes with different unbound bilirubin concentrations measured with the UB-analyser.
DECLARATIONS
