Abstract
Background
Lipoprotein-associated phospholipase A2 (Lp-PLA2) has been proposed as an inflammatory marker of cardiovascular disease. The present study investigates associations between Lp-PLA2 and other important biomarkers in Japanese patients with coronary artery disease.
Methods
We measured Lp-PLA2 levels in 141 consecutive patients (age 62.6 ± 3.8 years; men 69.2%) with angiographic evidence of coronary artery disease (acute coronary syndrome [ACS]; n = 38), stable angina pectoris (SAP; n = 72) or with angiographically normal coronary arteries (NCA; n = 31).
Results
Levels of Lp-PLA2 significantly correlated with low-density lipoprotein-cholesterol (r = 0.302), homocysteine (r = 0.528) and paraoxonase (r = 0.401) in all patients (all P < 0.01). Levels of Lp-PLA2 were significantly higher in patients with coronary atherosclerosis (ACS and SAP) than with NCA (P < 0.05). Levels of highly sensitive C-reactive protein were significantly higher in patients with ACS than with SAP and NCA (both P < 0.05). Multivariate logistic regression analyses revealed that higher Lp-PLA2 levels were independently associated with coronary atherosclerosis (odds ratio: 1.058; 95% confidence interval: 1.012–1.121; P = 0.001).
Conclusions
Higher Lp-PLA2 levels are associated with coronary atherosclerosis independently of traditional coronary risk factors. Thus, Lp-PLA2 is a novel biomarker of coronary atherosclerosis in Japanese patients.
Introduction
The role of inflammation in the pathogenesis of cardiovascular disease has become established over the last decade. Inflammatory processes significantly contribute to the initiation, progression and rupture of atherosclerotic plaques. 1 Lipoprotein-associated phospholipase A2 (Lp-PLA2, also known as platelet-activating factor acetylhydrolase) has recently emerged as a marker of cardiovascular risk. 2–4 Inflammatory cells in atherosclerotic plaque express Lp-PLA2, which then circulates in the bloodstream bound predominantly to low-density lipoprotein (LDL) and appears to impose a different inflammatory burden than C-reactive protein (CRP). 5–8 Lp-PLA2 hydrolyses phospholipids to yield proinflammatory products that are implicated in endothelial dysfunction, plaque inflammation and the formation of a necrotic core in plaque, and might be linked to the oxidative modification of LDL and the development of inflammatory responses in the arterial intima. 9–12 However, an association between circulating Lp-PLA2 concentrations and coronary artery disease (CAD) in Japanese patients has not yet been determined. In addition, relationships between Lp-PLA2 and other established biomarkers have not been examined in Japanese patients. We therefore investigated the clinical significance of circulating Lp-PLA2 in Japanese patients undergoing coronary angiography and evaluated its associations with other biomarkers.
Methods
The present prospective cross-sectional study included consecutive patients who had undergone coronary angiography at Juntendo University Hospital between July and December 2009. The entry criteria comprised no previous examination by coronary angiography, no history of coronary intervention or coronary artery bypass grafting and having precisely evaluable coronary trees. Demographic characteristics, medical history and current medications were collected from all participants at the time of enrolment.
Patients with angiographically normal coronary arteries (NCA) were placed in the NCA group, whereas those with detectable atheroma at angiography were defined as having CAD. The latter group was subdivided according to the presence of acute coronary syndrome (ACS) or stable angina pectoris (SAP). Acute myocardial infarction (AMI) and unstable angina (UAP) were included in the ACS group. The diagnostic criteria for AMI and UAP followed the respective guidelines published by the American College of Cardiology/American Heart Association in 2007. 13,14 These definitions of ACS depended on the specific characteristics of each element of the clinical presentation, electrocardiographic changes and a highly specific marker for cardiac damage. SAP was defined as effort angina with a stable profile of symptoms for at least three months before admission or no significant chest discomfort.
We initially screened 158 patients for this study. The exclusion criteria comprised maintenance dialysis (n = 5), diabetes treated with insulin (n = 4), acute or chronic infectious (n = 5) or neoplastic (n = 3) diseases. The Ethics Review Committee at our institution approved the study (number: 291); all participants signed informed consent forms and the study was registered in the UMIN protocol registration system (#UMIN000002103).
We evaluated the severity of CAD by standard coronary angiography. All angiograms were prospectively evaluated at our angiographic core laboratory. Two expert interventional cardiologists reviewed the angiograms with no knowledge of the biomarker levels and patient characteristics. Disagreement over lesion characteristics was resolved by a third expert. Angiographically significant lesions were defined as >50% stenosis in vessels with a diameter ≥ 2.0 mm. Extended CAD was classified in the standard manner as 1-, 2- or 3-vessel disease.
Arterial blood samples were obtained using a syringe and 18-gauge needles from the arterial sheaths of all patients before they were examined by coronary angiography in the operating room. Blood samples were directly collected into glass Vacutainer tubes with or without ethylenediaminetetraacetic acid (EDTA)-2Na to obtain plasma and serum, respectively. The samples were immediately placed on ice. The anticoagulated samples were centrifuged at 1000 ×
The activity of Lp-PLA2 in serum was determined based on the Lp-PLA2 hydrolysis of 1-mirystoyl-2-(4-nitrophenylsuccinyl) phosphatidylcholine to 4-nitrophenyl succinate, which spontaneously degrades under aqueous conditions and liberates 4-nitrophenol that is spectrophotometrically monitored at 405 nm. 15 The activity is expressed in IU/L, and 1 IU of Lp-PLA2 hydrolyzes 1 mmol of substrate/min. Highly sensitive CRP (hs-CRP) in the serum was measured using a validated, high-sensitivity immunoassay and an autoanalyser. Paraoxonase activity was measured using the methods of Kosaka et al. 16 and Beltowski et al. 17 Other markers were determined by routine laboratory methods.
All data were statistically analysed using SPSS version 18.0 (SPSS Inc., Chicago, IL, USA) and JMP version 9.0 (SAS Institute Inc., Cary, NC, USA). Continuous variables were expressed as means ± SD. Correlations were searched using Spearman's rank correlation. Values obtained from three groups were compared by the one-way analysis of variance, the Kruskal–Wallis test and the χ 2 analysis. The post hoc Scheffé test compared parameters within groups. The independent effect of the biomarkers on the risk of coronary atherosclerosis adjusting for potential confounders was determined using multiple logistic regression analysis. The following variables were initially incorporated into the univariate model: age, sex, hypertension, diabetes, dyslipidaemia, current smoking, LDL-cholesterol (LDL-C), glycosylated haemoglobin (HbA1c), Lp-PLA2, homocysteine (Hcy), paraoxonase, hs-CRP and statin therapy. Statistically significant variables determined in the multivariate regression analysis by stepwise forward selection and backward elimination were subsequently included in a new model. A value of P < 0.05 was considered statistically significant.
Results
Baseline demographic, clinical characteristics and biochemical measurement of all patients
ACE-I, angiotensin converting enzyme inhibitors; ARB, angiotensin receptor blockers; CAD, coronary artery disease; NCA, normal coronary artery; SAP, stable angina pectoris; ACS, acute coronary syndrome; HbA1c, glycosylated haemoglobin; Lp-PLA2, lipoprotein-associated phospholipase A2
Serum concentrations of Lp-PLA2 were significantly higher in the group with documented coronary atherosclerosis, namely the ACS (272.2 ± 143.8 IU/L) and SAP (281.4 ± 151.2 IU/L) groups than the NCA (182.6 ± 115.2 IU/L, P = 0.003 and P = 0.025, respectively) group, whereas the ACS and SAP groups did not significantly differ (P = 0.944) (Figure 1a). Serum concentrations of hs-CRP were significantly higher in the ACS (5.53 ± 14.9 mg/L) than in the SAP (1.48 ± 2.49 mg/L) and NCA (1.50 ± 2.47 mg/L, P = 0.034 and P = 0.042, respectively) groups, with no difference between the SAP and NCA groups (P = 0.999) (Figure 1b). Levels of LDL-C were also significantly higher in the ACS than in the SAP and NCA groups (P = 0.001 and P = 0.008, respectively), and the latter two groups did not significantly differ (P = 0.413). Serum concentrations of Hcy tended to be higher in the ACS than in the SAP and NCA groups (P = 0.035). However, serum concentrations of paraoxonase were significantly higher in the SAP than in the NCA group (P = 0.038), with no difference between the SAP and ACS groups (P = 0.226) (Table 1).
Concentrations of Lp-PLA2 (a) and hs-CRP (b) in patients with NCA, SAP and ACS. (a) *P < 0.01 compared with NCA. (b) †
P < 0.05 compared with NCA and SAP. Lp-PLA2, lipoprotein-associated phospholipase A2; hs-CRP; highly sensitive C-reactive protein; NCA, normal coronary artery; SAP, stable angina pectoris; ACS, acute coronary syndrome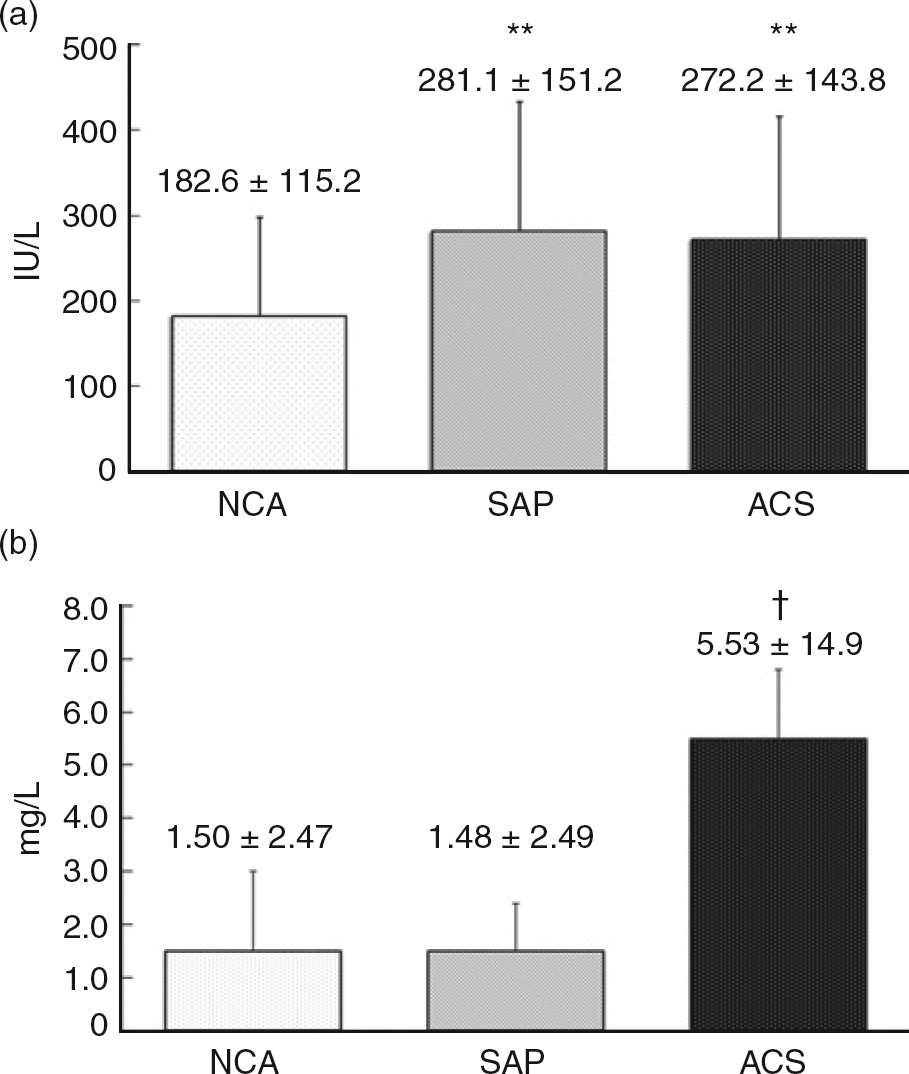
Correlations between Lp-PLA2 activity and other variables
Lp-PLA2, lipoprotein-associated phospholipase A2; HbA1c, glycosylated haemoglobin; hs-CRP; highly sensitive C-reactive protein
Correlations were searched using Spearman's rank correlation
Demographic and biochemical features of groups with lower and higher Lp-PLA2 values
Lp-PLA2, lipoprotein-associated phospholipase A2; ACE-I, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; NCA, normal coronary artery; SAP, stable angina pectoris; ACS, acute coronary syndrome; CAD, coronary artery disease; HbA1C, glycosylated haemoglobin
Univariate and multivariate logistic regression model for prediction of coronary atherosclerosis
OR, odds ratio; NS, not selected; HbA1c, glycosylated haemoglobin; hs-CRP; highly sensitive C-reactive protein; Lp-PLA2, lipoprotein-associated phospholipase A2
Discussion
The present study demonstrated an association between higher circulating Lp-PLA2 levels in patients with coronary atherosclerosis determined by coronary angiography. Furthermore, circulating Lp-PLA2 levels positively and significantly correlated with LDL-C and paraoxonase, and especially with Hcy levels.
This is the first study to evaluate associations between Lp-PLA2 and coronary atherosclerosis in a Japanese population. Levels of Lp-PLA2 were significantly higher in patients with than without documented coronary atherosclerosis. Lp-PLA2 is a novel biomarker and participant in vascular inflammation that is found in human atherosclerotic plaques. Oxidized LDL (oxLDL) is a substrate in reactions catalysed by Lp-PLA2 that result in lysophosphatidylcholine (lysoPC) and oxidized fatty acid. 10,18,19 The progressive inactivation of Lp-PLA2 during LDL oxidation leads to increased oxLDL uptake by macrophages, which could be attributed primarily to the increased uptake of the oxidized phospholipid-enriched lipid moiety of oxLDL. 20 The oxidized phospholipids of oxLDL accumulate in atherosclerotic lesions and up-regulate Lp-PLA2 production. 21 Hence, oxLDL and Lp-PLA2 are closely associated in the process of coronary atherosclerosis. On the other hand, lysoPC induces proinflammatory cytokines and chemokines such as interleukin (IL)-1β, IL-6 and tumour necrosis factor-α which trigger atherogenesis by sensitizing vascular smooth muscle cells and inducing the secretion of cellular adhesion molecules and matrix metalloproteinase by monocytes during the later stages of atherosclerosis. 22–25 Accordingly, the increase in Lp-PLA2 was associated with an increase in lysoPC, increased expression of proinflammatory genes in coronary plaque macrophages and the progression of atherosclerosis. 26 Thus, we consider that higher systemic circulating Lp-PLA2 levels associated with documented coronary atherosclerosis indicate not only the atherogenic process involved in proinflammation, but also the results of plaque formation.
Another study has independently associated Lp-PLA2 activity with clinical cardiovascular events, 27 and univariate analyses by others who have assessed the association between Lp-PLA2 levels and CAD assessed by coronary angiography also demonstrated the severity of angiographic CAD. 28,29 The results of our multivariate analysis revealed that higher Lp-PLA2 levels were as powerful a predictor of coronary atherosclerosis as either LDL-C or HbA1c. Thus, we postulate that the presence of atherosclerotic plaque could be evaluated by measuring circulating Lp-PLA2 concentrations. On the other hand, Lp-PLA2 concentrations did not differ between patients complicated with plaque instability and those with stable disease. Another study has shown that Lp-PLA2 concentrations are not increased in patients with ACS compared with those without ACS. This is in contrast to acute-phase reactants such as CRP, 28 which belongs to the pentraxin family and is the most extensively studied of the known proinflammatory factors. CRP plays a potentially pathogenic role in atheromatous plaque vulnerability, since higher concentrations closely correlate with increased numbers of thin cap fibroatheromas. 30 Consequently, we considered that Lp-PLA2 might be an important marker of, or play an active role in, the atherosclerotic process, whereas it does not play a causative role in increasing vulnerability to plaque rupture. We also believe that levels of Lp-PLA2 in patients have clinical significance as a chronic inflammation marker of plaque formation that differs from CRP, which is a marker of acute inflammation.
This exploratory study demonstrated that circulating Lp-PLA2 concentrations positively and significantly correlated with LDL-C, Hcy and paraoxonase. Circulating Lp-PLA2 is derived mainly from haematopoietic and inflammatory cells and it associates primarily with particles containing apolipoprotein B. 18,31 Hence, 70–80% of circulating Lp-PLA2 fractionates with LDL lipid and only a small ratio resides in the HDL lipid fraction. 32 The correlation between Lp-PLA2 and LDL-C reflects the binding of these factors. An elevated Hcy concentration is also considered to be a risk factor for the development of atherosclerosis. One report has suggested that Hcy influences endothelial function, which leads to a prothrombotic environment, platelet activation and endothelial leukocyte interactions. 33 Hcy was originally demonstrated as being bound to the apolipoprotein B of LDL, and the ratio of Hcy in LDL to that in HDL was elevated more than the corresponding cholesterol ratio. 34 The reactive anhydride of Hcy, Hcy-thiolactone, reacted with LDL, causing increased LDL density and aggregation and the formation of foam cells by interaction with cultured human macrophages. Hcy-thiolactone can also be disposed of via enzymatic hydrolysis by serum Hcy-thiolactonase/paraoxonase, which is a calcium-dependent extracellular enzyme transported by HDL in the blood. 35,36 In addition, Lp-PLA2 in carotid plaque significantly correlates with serum Hcy concentrations. 37 Therefore, we consider that a direct relationship between Hcy and circulating Lp-PLA2 or plaque Lp-PLA2 might accelerate atherosclerotic progression. Thus, further studies in vitro or in vivo are warranted to clarify relationships among Lp-PLA2, paraoxonase and cysteine metabolism in CAD.
In conclusion, higher Lp-PLA2 concentrations were as powerful a predictor of coronary atherosclerosis as either LDL-C or HbA1c. Thus, we believe that not only might Lp-PLA2 play an important role in Japanese patients with CAD, but it could also serve as a new systemic biomarker of coronary atherosclerosis. Furthermore, we suggest that circulating Lp-PLA2, paraoxonase and Hcy are closely related.
DECLARATIONS
