Abstract
Background
The treatment of severe sepsis highly depends on the identification of bacteria or fungi from blood and/or other body materials. Although widely available blood culturing and risk assessment scores are not completely reliable, current guidelines do not recommend the wide empirical use of antifungal medications based on questionable benefit or possible side-effects. We aimed to test whether platelet-derived microparticle (MP) measurements can improve the early detection of the infective agent behind sepsis.
Methods
Thirty-three consecutive severe septic patients from our university intensive care unit were included in our prospective study. MP number and surface antigen characteristics were followed by flow cytometry on days 1 (admission), 3 and 5. For microbiological identification, various specimens were collected on admission and in case of overall status deterioration.
Results
On admission, septic patients showed elevated annexin V and constitutive platelet marker (CD41)-positive MP numbers compared with volunteers. Mixed fungal septic patients showed significantly elevated annexin V and CD41-positive particle numbers on day 1 (P < 0.05) compared with the non-fungal septic group. Adhesive platelet marker (CD42a) harbouring vesicles were negligible in the non-fungal group, while fungal septic patients showed significantly elevated numbers in all measurements (P < 0.01). Particles from activated platelets (PAC1) had elevated numbers in the first and fifth study days compared with non-fungal septic patients (P < 0.05).
Conclusions
The measurement of CD42a- and PAC1-positive microparticles may provide important additional information which can help to improve the early instalment of antifungal therapy of severe septic patients.
Introduction
Early diagnosis and prompt appropriate treatment are still the best strategy of intensive care for severe septic patients. 1 The delay of therapy and development of multiple organ dysfunction may increase mortality above 70%. 2
One keystone of sepsis treatment is the identification of microbial agents behind the development of sepsis and the early administration of proper antimicrobial treatment. Kumar et al. 3 emphasized that patient survival could fall 7.6% in every hour without suitable medication.
For microbial identification, current sepsis guidelines recommend two haemocultures and samples from other possible sites of infection, 4 although recently published data suggest that 30% of haemocultures fail to identify the microbial source of sepsis. 5 Moreover, only 50–67% cases of blood stream fungal infections are detected by blood culturing. Novel tests, including nucleic acid amplification techniques, fungal cell wall-specific antibody and cell wall antigen detection tests, were introduced to improve the detection of fungal infections. Although the sensitivity of these tests is relatively high, cultures are still the most detailed identification method of a recent sepsis guideline. 4,6 The contribution of conventional amphotericin B in the development of renal failure can rise up to 33.2% and the role of azole medications in liver dysfunction (31.5%) questions the reasoning of pre-emptive use of these antifungal medications. 7
Following infection, the activation of the innate immune system is detectable in sepsis. Beside cells commonly described in innate immune response, there is an evolving evidence of platelet contribution in inflammation and early response of the host defence. 8,9 Following activation, platelet-derived microparticles (PMPs) are shed from the surface of platelets, harbouring markers indicating their cellular origin. 10,11 Microparticles (MPs) are 100–1000 nm in diameter, delimited by a phospholipid bilayer rich in phosphatidylserine and phosphatidylethanolamine. MPs harbouring platelet-specific surface proteins, including CD41, CD61, CD42a and PAC1, are referred to as PMPs. The hypothesized role of MPs in various physiological and pathophysiological processes includes cell-to-cell communication, induction of inflammation, cellular waste management and transfer of genetic elements. 12 Elevated numbers of particles are already described in different conditions, including coronary artery diseases, chronic inflammatory diseases and systemic inflammation response syndrome. 13
We have found no relevant information in the literature regarding the different PMP characteristics after bacterial- or fungal-induced platelet activation, but based on previous data regarding platelet activation in fungal and bacterial infections, we decided to assess if PMP measurements can provide additional important clinical information in the early recognition of the infective agent of sepsis. To test our hypothesis, we measured PMPs in severe septic patients with multiple organ failure on inclusion (day 1) and on the third and fifth days of severe sepsis, and registered the results of microbiological samples in the first week of intensive care unit stay.
Patients and methods
Patients
During our study period from 5 January 2009 until 20 December 2010, we enrolled 33 severe septic patients from our multidisciplinary university intensive care unit (Department of Anaesthesiology and Intensive Therapy, University of Pécs, Pécs, Hungary) with developed multiple organ failure and procalcitonin concentrations equal to or greater than 5 ng/mL (reference range: <0.5 ng/mL). Systemic inflammation response syndrome, sepsis, severe sepsis and septic shock were defined according to the current American College of Chest Physicians/Society of Critical Care Medicine criteria and the current Surviving Sepsis Guidelines. 14
Our study protocol was accepted by the Regional Research Ethical Committee of the University of Pécs (2406/2005) and was carried out according to the ethical guidelines of the 2003 Declaration of Helsinki. A written informed consent was acquired after detailed information regarding the study design and blood sampling from every patient included in the study. In case of consciousness disorder, the consent was provided by the next of kin according to national law. Patients admitted from different wards had greatly varying clinical history. We aimed to define our exclusion criteria to exclude certain factors which can significantly alter MP amounts: any type of known haematological malignancy, cytostatic treatment in the last 30 days, high-dose prolonged steroid medication, patients with disseminated intravascular coagulation score ≥5, 15 drugs known to alter platelet functions (i.e. acetylsalicylic acid), platelet transfusion during the study period and moribund state. We defined our endpoints as the withdrawal of consent or death during the research period. In case of death, patient data collected until the last flow cytometry measurement was assessed. The diagnostic and treatment procedures of sepsis were conducted by strictly following the recent sepsis guidelines in both study and non-study patients. 4 Laboratory parameters including C-reactive protein, procalcitonin, lactate concentration, blood cell count, electrolyte concentrations, blood gas parameters and organ function-specific parameters were registered. To follow up the clinical status of severe septic patients, Multiple Organ Dysfunction Score and Sequential Organ Failure Assessment scores were calculated every single day during the whole study period and Simplified Acute Physiology Score II was calculated after 24 h of admission. 16–18
Twenty age- and gender-matched healthy volunteers were invited to determine the baseline values of our measurements.
Blood sampling
Additional blood samples for flow cytometry measurements were drawn on admission (day 1) and on the third and fifth days with a 21-gauge cannula into a closed system blood sampling tube with Na3-citrate (0.129 mol/L) as anticoagulant. Samples were processed within a maximum of 30 min after collection. Volunteers provided blood for flow cytometry measurements on one occasion. Blood collection from volunteers was carried out through vein puncture with a 21-gauge needle into a closed system and processed as above.
MP isolation and flow cytometry measurement
Antigen specificity, fluorescent dye, clone, base concentration and the manufacturer data of antibodies used
Cy5, cytochrome 5; FITC, fluorescein isothiocyanate; PE, phycoerythrin; PerCP, peridinin chlorophyll protein
All antibodies and annexin V were titrated in preliminary experiments to determine the optimal labelling concentrations. Labelling concentrations were defined by antibody staining of samples and sample-free buffers in the presence and absence of CaCl2. Labelling was considered optimal if CaCl2-labelled sample measurement events were distinguishable from background and CaCl2-free staining.
MPs (10 μL) were incubated with 10 μL antibody previously diluted to an optimal labelling concentration in 100 μL Apo-binding buffer supplemented with 2.5 mmol/L CaCl2 for 30 min at room temperature in a dark chamber. MPs isolated after the stimulation of pooled platelet-rich plasma from five healthy volunteers without platelet function-altering medication with calcium ionophore A23186 (Sigma-Aldrich; 25 μmol/mL, 37°C, 10 min) were used as positive controls. Pooled platelet-rich plasma (200 μL) was mixed with A23186 as above and incubated at room temperature for 30 min. MP isolation was continued from the second centrifugation step accordingly. MPs stained with annexin V in the presence of 2.5 mmol/L ethylenediaminetetraacetic acid were measured for negative controls. Flow cytometry measurements and data analysis were performed on a Beckman Coulter FC500 flow cytometer with CXP software (Beckman Coulter, Budapest, Hungary). The MP gate was defined in order to distinguish the true events from electronic noise and background, using 0.3, 0.5 and 1.0 μm fluorescein isothiocyanate-labelled microbeads (a kind gift of Becton Dickinson). Side scatter, forward scatter and fluorescence channels were set in a logarithmic scale. The MP size gate was determined between 0.5 and 1.0 μm size range. Events in the MP gate were further discriminated by labelling with annexin V. MPs were defined as annexin-V-positive events in the MP gate with fluorescence intensity above the isotype control. For determination of the MP number, a known concentration (105/mL) of 3.0 μm diameter microbeads (Becton Dickinson) was used. MP measurements were carried out in the Department of Laboratory Medicine, University of Pécs, Pécs, Hungary. The intrarun variation coefficient range of our complete isolation and flow cytometry method, determined by a total of 25 measurements from five different controls, was 15.4–19.2%. Figure 1 summarizes the gating strategy of our flow cytometry protocol.
Flow cytometry gating strategy. (a) Sample tube with known concentration of microbeads (BEAD gate) along with the microparticle size gate (MP_size gate), determined in previous measurements with fluorescent microbeads. (b) Control measurement for annexin V staining. Sample was stained with annexin V in the presence of 2.5 mmol/l ethylenediaminetetraacetic acid (EDTA). (c) Selection of annexin-V-positive events. (d) Isotype control for fluorescein isothiocyanate (FITC) measurements, events in the AA1 and AA2 quadrants are recognized as positive with a FITC-stained antibody. (e) PAC1-FITC measurement from a patient's sample. Events in the forward scatter size gate (MP_Size gate), positive for annexin V (ANNEXIN+ gate) and having higher fluorescence compared with the isotype control, are regarded as PAC-1-positive microparticles (AZ1–2 quadrants). Cy5, cytochrome 5; MP, microparticle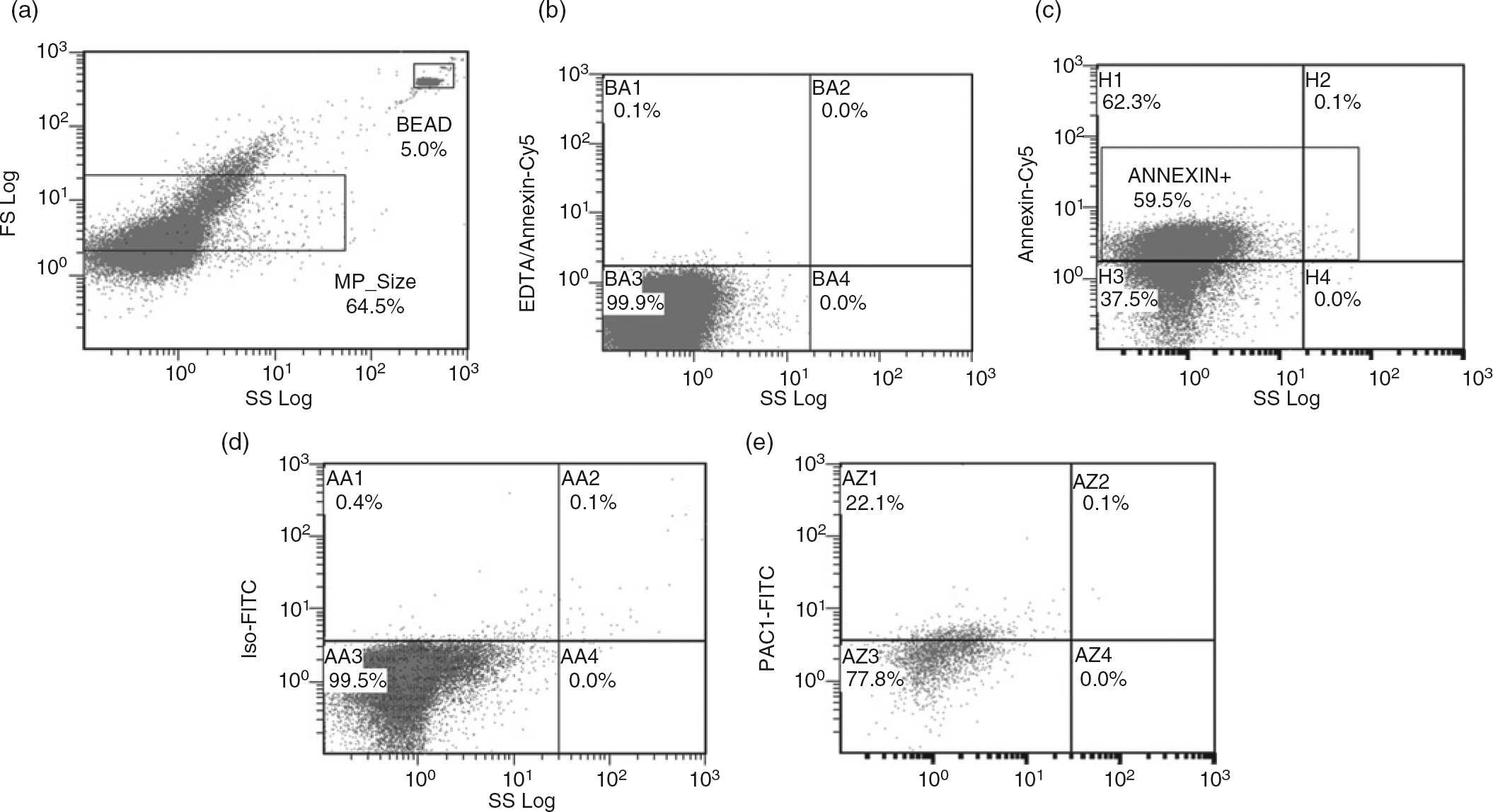
Microbiological diagnosis and treatment
For the diagnosis of the microbial agent(s) in sepsis, haemocultures, urine samples and bronchial lavage fluids were gathered. Haemocultures were obtained through a fresh vein or arterial puncture, prior to empirical antimicrobial medication, into BACTEC™ haemoculture flasks (Becton Dickinson) from patients showing signs of systemic inflammation response syndrome without recent microbiological results.
Further microbiological sampling was carried out in case of continuous or elevated fever with shivering or overall patient clinical status deterioration, including continuously elevated or increasing C-reactive protein and procalcitonin results following proper antimicrobial treatment.
Microbiological identification was performed in the Department of Medical Microbiology and Immunology, University of Pécs, Pécs, Hungary.
Empirical antibiotic therapy was started following microbiological sampling. The applied therapy generally consisted of carbapenems, respiratory fluoroquinolones, metronidazole and second- or third-generation cephalosporins. The selection of antibiotics was based on the clinical background and possible site of infection. Empirical antifungal therapy (fluconazole) was administered in case of a positive patient history for diabetes, an immunocompromised state (i.e. tumours or steroid treatment), long-term antibiotic treatment, progressive sepsis without or with only a slight procalcitonin elevation accompanied by constant fever with shivers.
Patients in our study were designated as fungal septic in the case of positive blood culture results for fungal infection accompanied by the abovementioned clinical signs, following proper antibacterial treatment during our study period. Based on these criteria, a fungal septic and a non-fungal septic group were formed.
Statistical analysis
Statistical analysis was carried out using SPSS 19 for Windows (IBM Corporation, Somers, NY, USA). For statistical comparison, the non-parametric Mann-Whitney U test was used, corrected for multiple comparisons. In statistical calculations, P ≤ 0.05 was considered significant.
Results
During our study period, 57 patients were eligible according to our inclusion criteria, but only 33 patients gave informed consent. Eight patients refused participation following detailed information about our study. Sixteen patients were excluded based on our criteria, after the reassessment of patient history (platelet inhibitor use, prolonged steroid therapy, etc).
Basic demographic data, organ dysfunctions, microbiologically proven infections and clinical status by scoring systems of our septic patients
PCT, procalcitonin; CRP, C-reactive protein; MODS, Multiple Organ Dysfunction Score; SOFA, Sequential Organ Failure Assessment Score; SAPS, Simplified Acute Physiology Score II
Data are presented as medians (first–third quartiles). Reference values: PCT: <0.5 ng/mL; CRP: <5 mg/L; lactate: 0.63–2.44 mmol/L. Infections are detailed as the total number of positive microbiological results in severe septic patients, and the number in brackets indicates the portion of mixed infections in the same microorganism group
All enrolled severe septic patients presented with elevated procalcitonin and C-reactive protein levels. The most common organ dysfunctions developing in our patients were kidney dysfunction (oliguria or anuria), haemodynamic shock, consciousness disorder and respiratory failure. A large portion of bacterial and fungal infections identified in our patients were mixed bacterial or mixed bacterial–fungal infections. Six patients comprised the mixed fungal septic group and 27 patients were in the non-fungal septic group. Microbiological identification proved Candida albicans species in all six fungal septic patients. Two patients of the mixed fungal septic group and five from the non-fungal septic group died during the study period.
Upon admission, total annexin-V-positive MPs and CD41-positive PMPs were elevated in both the mixed fungal and in the non-fungal septic group compared with our volunteer group. Most MPs (above 60% average) were positive for constitutive platelet antigen CD41, and therefore recognized as PMPs.
While mixed fungal septic patients showed elevated annexin-V-positive MP numbers throughout our study with a slight decrease on the third day, the non-fungal septic patients showed constantly decreasing MP numbers during our study period. The elevation was significant in mixed fungal septic patients compared with non-fungal septic patients on the first study day (Figure 2a, P = 0.045).
Microparticles in fungal and non-fungal severe septic patients and control baseline values. (a) The number of total annexin V-positive microparticles in severe septic patients. (b) Numbers of CD41+ platelet-derived microparticles in the fungal and non-fungal septic groups. (c) The platelet adhesion marker CD42a-positive vesicle numbers in fungal and non-fungal patient groups. (d) The numbers of PAC1, anti-fibrinogen-binding receptor antibody-positive particles in fungal and non-fungal septic patients (*P < 0.05; #
P < 0.01)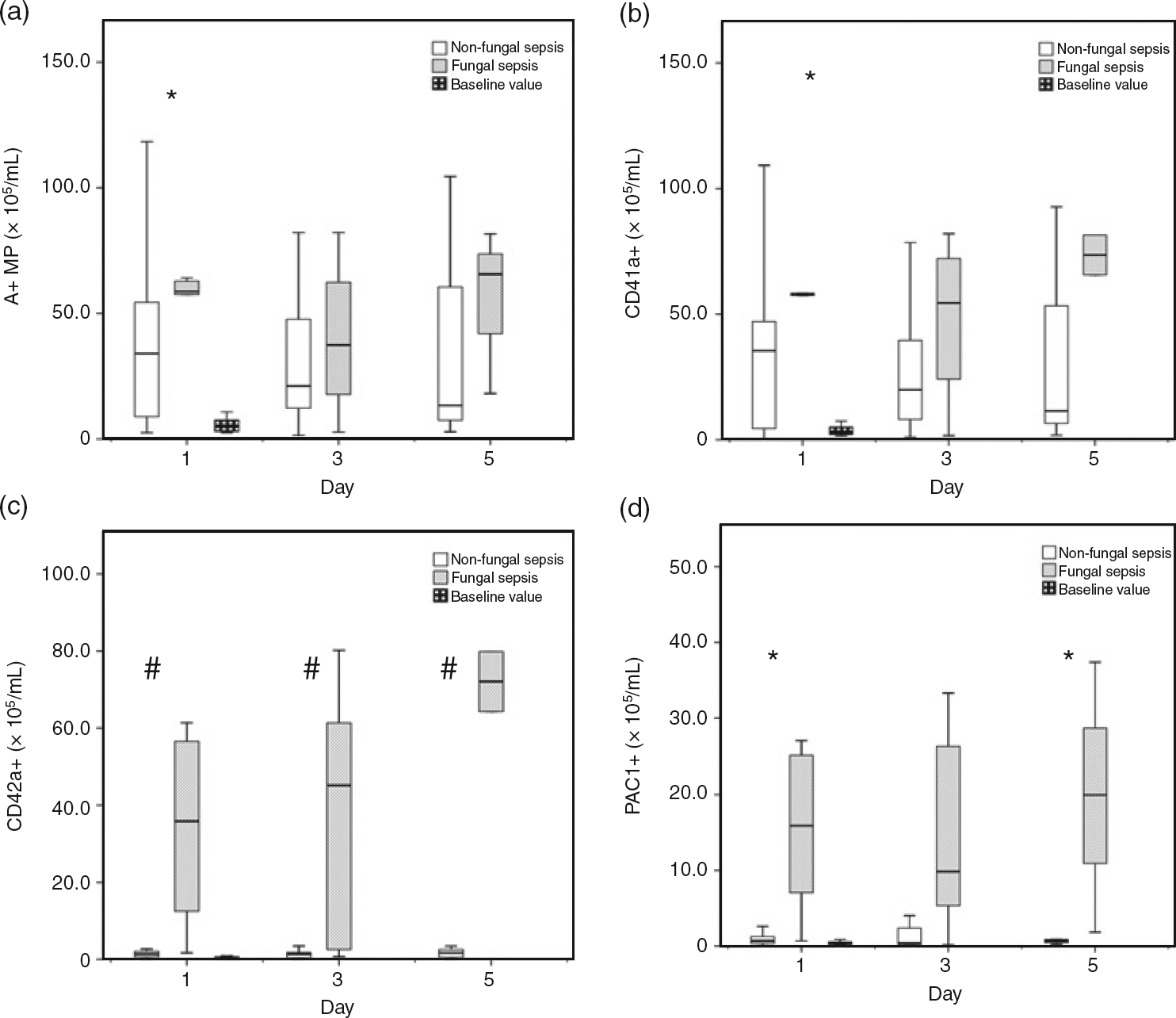
The CD41-positive PMP numbers were decreasing until day 5 in the non-fungal septic patients and were constantly elevated in the mixed fungal septic group. The elevation was statistically significant on the first day (Figure 2b, P = 0.012). CD61 results were statistically identical to CD41 results in all measurements (data not shown).
While CD42a+ PMP numbers were negligible in the non-fungal group, mixed fungal septic patients showed significantly elevated numbers in all measurements with a steep elevation until day 5 (Figure 2c, P = 0.003).
Although PAC1+ PMPs numbers showed a slight decrease and marked variety until day 5 in fungal septic patients, this group of patients had elevated numbers of PAC1-positive PMPs in the first and fifth study days compared with non-fungal septic patients. Non-fungal patients had low numbers of PAC1-positive PMPs (Figure 2d, P = 0.030).
Discussion
The role of platelets in early host defence and inflammation is long debated. Yeaman 9 extensively discussed platelet reactions to endothelial injury and the presence of microbial agents. Bacteria and fungi are known to activate platelets. Following activation, platelets can act directly by adhering to the endothelial wall or to the pathogen and forming aggregates. Also, antimicrobial proteins are released from platelets. These proteins, like thrombocidins, are highly effective against certain bacterial and fungal strains, while others successfully developed resistance against them. The increase of platelet activation markers and adhesive platelet markers on PMP surfaces may indicate platelets’ contribution in early host defence. 19–21
In agreement with Nieuwland et al., 22 all septic patients showed increased numbers of PMPs compared with volunteers. We have shown that compared with severe bacterial sepsis, severe sepsis with mixed fungal infection contributes to increased PMP levels in our multidisciplinary intensive care unit setting. Although fungal sepsis was considered rare and contributes only about 5% as a main pathogen, a current review showed a two-fold increase of Candida incidence from 1979 to 2000, with an estimated incidence of 25–30 of 100000 persons annually. 6 Most fungal infections are present as a super-infection or following antibacterial medication. 6 In addition, a large portion of patients have prolonged sepsis characterized by protein wasting, which combined with the infections, results in the overall deterioration in outcome. The assessment of a conventional automated haemoculture system found that opposed to current guidelines, 4,23 at least three cultures and a 24-h incubation time is necessary to reach an optimal infection detection rate of 95%. 23 In some cases, the incubation time for fungal haemocultures may rise up to five days while the detection rate for Candida species could still be as low as 67%. 23 Also, a recent study showed that most patients with yeast infections have inappropriate medication, 6 but empirical, pre-emptive use of antifungal therapy is questionable and not recommended. Adverse effects of antifungals may vary from renal failure from vasoconstriction (conventional amphotericin B) to visual disturbances, phototoxicity or skin rash (voriconazole) and hepatotoxicity (posaconazole, voriconazole). Itraconazole and other azole medications are well tolerated, with only rare and mild cases of hepatotoxicity, but widely used. Fluconazole is used for prophylaxis and treatment for a longer period. Besides novel laboratory techniques, multiple risk assessment scores were developed to help in the decision of whether to introduce antifungal therapy, but further validation is needed before introduction into wide clinical usage. 5,24
The abovementioned data emphasize the importance of the proper indication of antimicrobial treatment. Considering the shortcomings of generally used microbiological identification systems and risk assessment scores, our PAC1+ and CD42a+ PMP measurements can provide valuable additional information on mixed fungal sepsis-prone patients following admission to the intensive care unit.
According to our current knowledge, there is no evidence that antifungal medication alters platelets or PMPs.
The authors emphasize the low number and mixed fungal infection of severe septic patients as a key limitation of this study, but Annane et al. 5 have already discussed that fungi as a main infective agent accounts for a small portion of total cases (about 5%). Many CD41+ MPs did not stain for CD42a in the non-fungal septic group, but did in the fungal septic group. We have no clear explanation for this discrepancy. We hypothesize that platelets in the fungal sepsis group may produce CD42+ MPs in the fungal group, but alternatively, the antigen may also be hidden in some way in the non-fungal sepsis group. All patients from our mixed fungal septic group suffered from C. albicans infection, and therefore further PMP measurements from non-albicans fungal infections are needed to clarify the importance of PMPs as an early marker for mixed fungal sepsis.
Conclusions
Our data revealed that measurement of PAC1- and CD42a-positive PMPs can provide useful early information in the assessment for mixed C. albicans fungal sepsis, and thus can improve treatment decisions during the care of severe septic patients. Further extensive studies are needed to confirm the clinical usefulness and biological background of similar PMP alterations in severe septic and multiple organ failure patients.
DECLARATIONS
