Abstract
Background
Human epididymis protein 4 (HE4) has recently become an available tumour biomarker for detecting ovarian cancer along with the standard cancer antigen 125 (CA125). However, it is unknown if the levels of HE4 and CA125 may be altered in subjects who have impaired renal function with no ovarian disorders.
Methods
In 113 female patients at different stages of chronic kidney disease (CKD) with no ovarian and lung cancer and 68 subjects with normal renal and ovarian function, HE4 and CA125 concentrations were analysed by using chemiluminescent microparticle immunoassay (Architect®, Abbott) and electrochemiluminescent immunoassay (Modular E170®, Roche), respectively. Renal function was evaluated by measuring serum creatinine and urea concentrations (Cobas Integra-800®, Roche). Estimated glomerular filtration rate (eGFR in mL/min/1.73 m2) was calculated by the 4v-MDRD formula.
Results
Significantly increased HE4 concentrations (P < 0.0001) were found in individuals with differently decreased eGFR values (<90 mL/min/1.73 m2) compared with clinical controls. CA125 serum concentration was higher than normal in subjects with CKD3 (eGFR = 30–59 mL/min/1.73 m2), but significant elevation (P = 0.006) in CA125 concentrations was seen only in those who had severe renal failure (CKD4-5; eGFR < 30 mL/min/1.73 m2). These tendencies were independent of age in our study cohort, and seemed to be more evident among women in premenopausal status.
Conclusions
HE4 concentrations may be elevated in CKD patients with no ovarian and lung cancer. Thus, HE4 results should be interpreted cautiously in women with renal disorders.
Introduction
Ovarian cancer is the leading cause of death in women who have gynaecological malignancies in the Western world. 1 It is potentially curable at an early stage with ∼90% survival rate by primary surgery and aggressive chemotherapy, but patients with high-grade disease still show poor prognosis and a high mortality rate. 2 For more than two decades, cancer antigen 125 (CA125) was the only accessible tumour marker for the laboratory detection of ovarian cancer. 3 However, its sensitivity was quite low in early stage disease, and there were numerous clinical cases when CA125 concentrations were falsely elevated in patients with endometriosis, ovarian cyst, pelvic inflammation or liver dysfunction in the absence of ovarian malignancies. 4 Moreover, CA125 values were also abnormal in other malignant cancer diseases such as breast, lung or pancreatic tumours. 5 Thus, the need for a novel tumour biomarker with higher sensitivity and specificity was obvious. 6,7
Several clinical studies have recently evaluated the role of human epididymis protein 4 (HE4) analysis alone or in combination with CA125 to predict ovarian cancer in patients with pelvic mass, 8,9 to differentiate benign and malignant ovarian masses from each other, 10 or those from ovarian endometriotic cysts 11 and in order to indicate the recurrence of the disease. 12 When the levels of both tumour biomarkers are measured to evaluate the risk of ovarian malignancy, the risk of ovarian malignancy algorithm (ROMA) score can be calculated specified for either pre- or postmenopausal states. 8 In a large multicenter survey, this score demonstrated a sensitivity of 92.3% and a specificity of 76.0% in women in premenopause, whereas a 100% sensitivity and a 74.2% specificity were observed in the postmenopausal status. 13
Reasonable specificity (74.7%) and negative predictive value (93.9%) of HE4 analysis combined with CA125 measurement have been published in various benign gynaecological diseases. 8,14 Additionally, it has just been revealed that high HE4 concentrations were related to poor prognosis in lung adenocarcinoma. 15 Interestingly, only few data are known about the potential fluctuation of HE4 concentrations in healthy subjects or in those who suffer from other non-malignant, non-gynecological diseases. In healthy women, HE4 concentrations significantly varied in a menstrual cycle phase-dependent manner. 16 Here, we studied for the first time whether the levels of HE4 and CA125 were affected in different stages of chronic kidney disease (CKD) in the absence of ovarian and lung cancer.
Materials and methods
In this study, 181 patients (133 postmenopausal, 48 premenopausal) were enrolled who attended the Outpatient Clinic at the Department of Obstetrics and Gynecology (University of Debrecen) for a general check-up examination. The patients had various chronic diseases such as hypertension, cardiovascular disease, hyperlipidaemia, gastritis, type 2 diabetes mellitus, autoimmune disease, peripheral artery disease, menstrual bleeding disorders or renal dysfunction (estimated glomerular filtration rate, eGFR <90 mL/min/1.73 m2; n = 113). Of them, 12 subjects had previously undergone kidney transplantation with prolonged graft survival. For clinical controls, 68 subjects with normal renal (eGFR ≥ 90 mL/min/1.73 m2) and ovarian function were recruited.
According to their physical and radiological examination, no ovarian and pulmonary disorders were detected. The levels of additional tumour markers (CA15-3, CA72-4, CA19-9, CEA) were also measured with negative results (data not shown). Chemiluminescent microparticle immunoassay (Architect®; Abbott, Wiesbaden, Germany), and electrochemiluminescent immunoassay (Modular E170®; Roche Diagnostics, Mannheim, Germany) were used for analysing HE4 and CA125 serum concentrations, respectively. CA125 and HE4 cut-offs were set as 35 kU/L; 70 pmol/L (in premenopause) and 140 pmol/L (in postmenopause), respectively. Renal function was evaluated by measuring serum creatinine and urea concentrations (Cobas Integra-800; Roche Diagnostics), and eGFR (mL/min/1.73 m2) was calculated by the 4v-MDRD formula. 17
Statistical analysis
Data are expressed as median (range). The Kolmogorov–Smirnov test was used for the evaluation of the normality of the data. Outcome continuous parameters were non-normally distributed; therefore analyses were performed by using the Kruskal–Wallis test and Mann-Whitney U test to compare the CKD patient groups to clinical control subjects. P < 0.05 was regarded as statistically significant. Analysis was performed by using SPSS version 13.0 (SPSS Inc., Chicago, IL, USA).
Results
According to the stage of renal function, patients were separated into four groups to study the alteration in HE4 concentrations in CKD along with CA125 measurements (Table 1). CKD4 (eGFR = 16–29 mL/min/1.73 m2) and CKD5 (eGFR ⩽ 15 mL/min/1.73 m2) women were pooled into one group (CKD4-5) due to the low case number (n = 24). Since there was a significant difference (P = 0.001) in age among the whole control group (n = 68) and the different CKD patient groups, an age-adjusted (62.5 [60.8–65.5] years; P = 0.054) control subgroup (n = 12) was also set for further statistical comparisons (data not shown). In general, we found that HE4 concentrations were significantly increased (P < 0.0001) at the various stages of CKD related to decreasing eGFR compared with the control cohort (Table 1). A significant elevation in HE4 concentration was already noted in CKD2 (eGFR = 60–89 mL/min/1.73 m2) women compared with those with normal eGFR values (76.9 [55.5–101.8] pmol/L versus 44.1 [35.9–52] pmol/L). In the premenopausal CKD2 subgroup, HE4 values were abnormal (72.6 [52.9–101.3] pmol/L, P = 0.003) except for two outliers, and remained within the reference range in most postmenopausal CKD2 patients (77.6 [56.7–100.0] pmol/L, P = 0.001) (Figures 1a and b). CA125 showed no elevation in these subjects versus controls (15.5 [9.9–29.5] kU/L) regardless of their menopausal status (10.8 [10.3–17.7] kU/L [premenopause]; 12.6 [9.9–21.0] kU/L [postmenopause]) (Figures 1c and d). Among CKD3 (eGFR = 30–59 mL/min/1.73 m2) subjects, HE4 concentrations were significantly higher than normal in either the pre- or postmenopausal subgroup (180.3 [147.1–235.6] pmol/L, P = 0.0004; 205.9 [131.8–314.5] pmol/L,P = 0.0001). However, CA125 concentrations were still found around the reference range in both premenopausal (27.2 [24.4–37.6] kU/L) and postmenopausal CKD3 women (26.6 [15.4–39.1] kU/L). Further significant increases(P < 0.0001) in HE4 concentrations were observed in all CKD4–5 subjects (646.9 [369.4–952.3] pmol/L) compared with the control group (Table 1). In addition, CA125 concentrations were significantly (P = 0.021) elevated especially in most premenopausal CKD4-5 patients (40.9 [34.1–98.1] kU/L), and a minor but significant (P = 0.004) increment of this marker level was found among postmenopausal subjects (25.8 [21.6–41.1] kU/L) (Figures 1c and d).
Serum HE4 and CA125 concentrations measured in patients with normal (controls) and differently decreased eGFR parameters (CKD2-5). Subjects were divided into two main groups according to their menopausal status. (a) HE4 concentrations in premenopausal patients; (b) HE4 concentrations in postmenopausal subjects; (c) CA125 concentrations in women in premenopause; (d) CA125 concentrations studied in postmenopausal states. Kruskal–Wallis test and Mann-Whitney U test were used to compare the CKD patient groups to control subjects. Dashed line represents the reference range of either biomarker in the pre- and postmenopausal states. CKD, chronic kidney disease; CA, cancer antigen; GFR, glomerular filtration rate; HE4, human epididymis protein 4 Characteristics and laboratory parameters of all study participants recruited into different CKD subgroups based on their renal function Data are presented as median (range). During statistical analysis, patients from the pooled CKD2–5 groups were also compared with the clinical control cohort by using the non-parametric Kruskal–Wallis test CKD, chronic kidney disease; CA, cancer antigen; GFR, glomerular filtration rate; HE4, human epididymis protein 4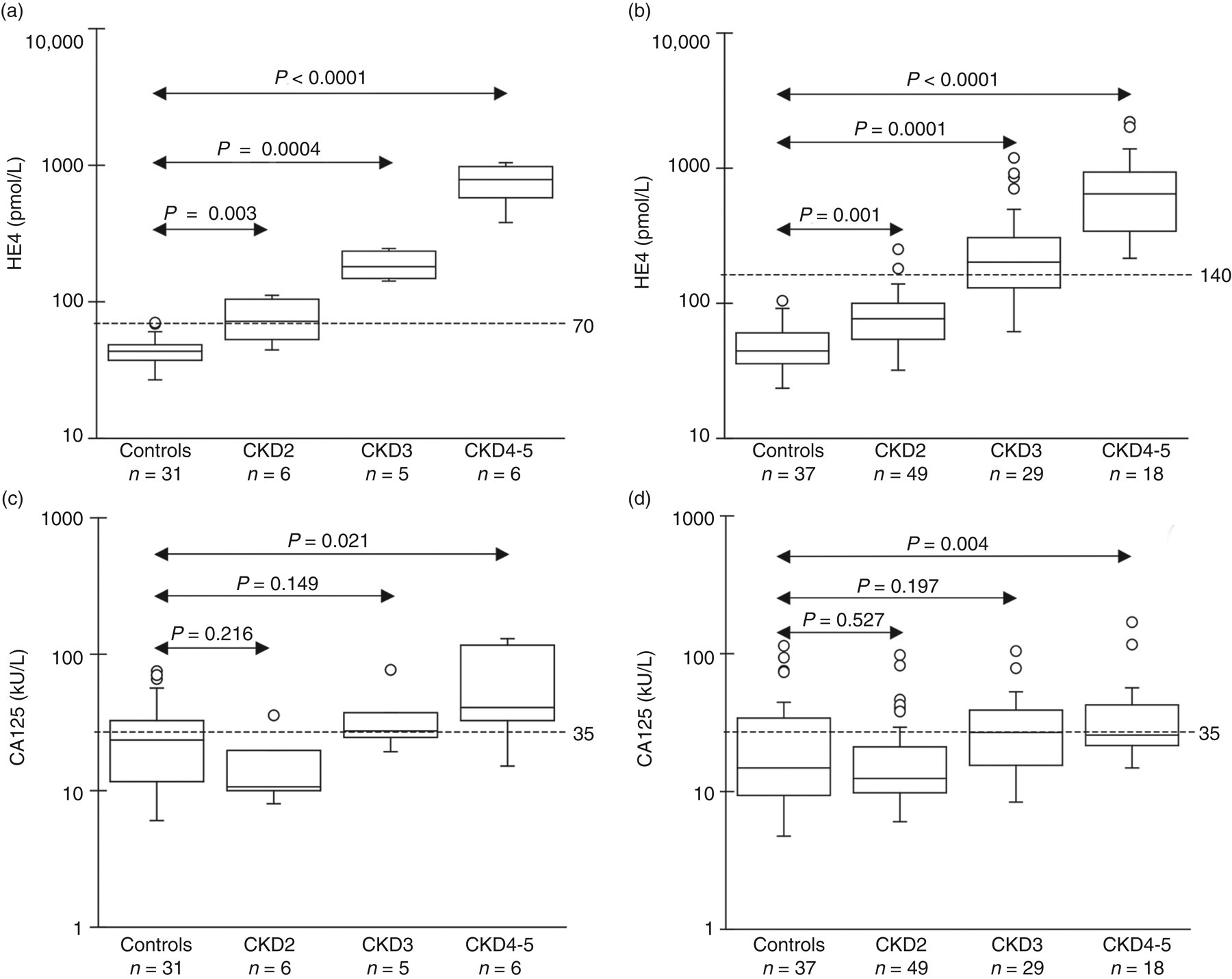
Although the female patients, after kidney transplantation (with median eGFR of 21.5 [16.1–37.3] mL/min/1.73 m2), did not have ovarian or lung cancer, they showed elevated HE4 concentrations (297.4 [174.1–652.1] pmol/L) with increased CA125 concentrations (24.2 [19.1–42.7] kU/L). In addition, age-matched control women with healthy renal function demonstrated significantly lower HE4 concentrations (46.8 [38.4–60.9] pmol/L, P = 0.0003) and normal median CA125 values (14.6 [9.4–28.2] kU/L) compared with individuals with abnormal eGFR (data not shown). Accordingly, significant differences in HE4 concentrations were independent of age in our study cohort.
Discussion
Simultaneous measurement of HE4 and CA125 concentrations provides a better discrimination of malignant ovarian tumours from benign masses, and follow-up of patients with ovarian cancer after surgery and chemotherapy. 8–14 However, the clinical utility of these tumour biomarkers is somewhat limited in certain cases: CA125 has a low sensitivity, especially in early stages of ovarian cancers, as well as being falsely elevated in different non-malignant diseases. 3,4 Here, we analysed for the first time, if kidney disorder at different stages would affect the levels of HE4 along with CA125 in pre- and postmenopausal patients with negative ovarian and respiratory conditions.
In our study, CA125 serum concentrations were significantly augmented only in severe renal failure. However, there were several outliers in terms of CA125 concentration in each subgroup, showing that the level of this biomarker may be affected by further (e.g. inflammatory) factors. In contrast, HE4 concentrations were often abnormal and significantly increased in patients even at early stages of CKD. In conclusion, HE4 concentrations are highly influenced by the alteration of renal function especially in premenopausal states.
Similarly to HE4, the level of other biomarkers such as cardiac troponin (cTn) may be affected by impaired kidney function. Both cTnT and cTnI were formerly studied in this aspect, and their serum levels were also significantly elevated in different CKD patients undergoing hemodialysis or even in those with end-stage renal failure (CKD5) without signs of acute myocardial damage. 18,19 Overall, the presence of impaired renal function needs to be considered during the interpretation of these biomarkers as well as HE4 results.
DECLARATIONS
