Abstract
Background
Prolactin has multiple forms and macroprolactin, which is thought not to be bioavailable, can cause a raised serum prolactin concentration. Gel filtration chromatography (GFC) is currently the gold standard method for separating macroprolactin, but is labour-intensive. Polyethylene glycol (PEG) precipitation is suitable for routine use but may not always be accurate. We developed a high pressure liquid chromatography (HPLC) assay for macroprolactin measurement.
Methods
Chromatography was carried out using an Agilent Zorbax GF-250 (9.4 × 250 mm, 4 μm) size exclusion column and 50 mmol/L Tris buffer with 0.15 mmol/L NaCl at pH 7.2 as mobile phase, with a flow rate of 1 mL/min. Serum or plasma was diluted 1:1 with mobile phase and filtered and 100 μL injected. Fractions of 155 μL were collected for prolactin measurement and elution profile plotted. The area under the curve of each prolactin peak was calculated to quantify each prolactin form, and compared with GFC.
Results
Clear separation of monomeric-, big- and macroprolactin forms was achieved. Quantification was comparable to GFC and precision was acceptable. Total time from injection to collection of the final fraction was 16 min.
Conclusions
We have developed an HPLC method for quantification of macroprolactin, which is rapid and easy to perform and therefore can be used for routine measurement.
Introduction
Prolactin is a hormone produced by the anterior pituitary. There are multiple circulating forms: monomeric prolactin (molecular weight ∼23 kDa), the predominant form in healthy people; big prolactin (∼60 kDa); and big-big or macroprolactin (∼150 kDa), a complex of monomeric prolactin and an antiprolactin autoantibody, usually IgG. 1,2 Macroprolactin accounts for a small but variable percentage of circulating prolactin and also reacts variably with different assays, thus contributing to the large variation in the prolactin reference intervals of immunoassay platforms. 1 Macroprolactin is thought not to be bioavailable and does not cause clinical effects or symptoms. 3 Macroprolactin concentration in the serum may be elevated (causing macroprolactinaemia) due to the reduced clearance of the prolactin–IgG complex, rather than increased prolactin production. 4 Macroprolactinaemia is estimated to account for up to 26% of all cases of elevated total prolactin. 1 However, the incidence of macroprolactinaemia may depend on how it is defined and the prolactin assay used. 3 Gel filtration chromatography (GFC) is regarded as the reference method for separation and quantification of the different prolactin forms. 3 However, this method is complex, time-consuming and does not lend itself to routine application. 1 Polyethylene glycol (PEG) precipitation is the most widely used method to screen for macroprolactin in routine practice. However, there are issues with the use of PEG, including positive interference with some immunoassay platforms leading to recoveries in excess of 100%. 3 We developed a size exclusion high pressure liquid chromatography (HPLC) method to rapidly separate the different prolactin forms.
Materials and methods
For gel filtration chromatography, 100 μL of undiluted plasma or serum was applied to a 20 cm × 10 mm column (considered to be a short-column method), containing Sephadex G100 (Pharmacia Biotech, Uppsala, Sweden). Mobile phase: 50 mmol/L Tris buffer with 0.15 mmol/L NaCl at pH 7.2. Twenty-five 0.5-mL fractions were collected and prolactin measured by chemiluminometric immunoassay on Siemens Immulite 2000 (Siemens Healthcare Ltd, Flanders, NJ, USA).
High pressure liquid chromatography was carried out using a Waters 515 HPLC pump, Waters 717plus autosampler (Waters Corporation, Milford, MA, USA) and an Agilent Zorbax GF-250 (9.4 × 250 mm, 4 μm) size exclusion column (Agilent, Wilmington, DE, USA). Mobile phase: 50 mmol/L Tris buffer with 0.15 mmol/L NaCl at pH 7.2, with a flow rate of 1 mL/min. Column eluate was collected using a LKB BROMMA 2212 Helarik Fraction Collector (Pharmacia Biotech) in 55 fractions of approximately 155 μL each. Protein markers with known molecular masses, ranging from 1.35 to 670 kDa (Bio-Rad Laboratories, Hercules CA, USA), were injected and retention times determined. Serum or lithium-heparin plasma was diluted 1:1 with mobile phase, filtered using a Phenex (Phenomenex, Torrance, CA, USA) 4 mm syringe filter (regenerated cellulose membrane 0.45 μm) and 100 μL injected. Thirty-five fractions (fractions 17–52) were assayed for prolactin on the Siemens Immulite 2000. On one occasion, in order to confirm the co-elution of prolactin and IgG in the macroprolactin peak, IgG was measured using the Abbott immunoturbidimetric assay on the Architect c16000 (Abbott Laboratories, Abbott Park IL, USA). The ratio of macroprolactin to monomeric prolactin was calculated by determining the area under the curve for each peak on the chromatogram using GraphPad Prism software (GraphPad Software Inc, La Jolla, CA, USA). Linearity was assessed using dilutions of a sample with predominant monomeric prolactin of 5766 mU/L 3:1, 1:2, 1:3 and 1:7 in mobile phase. The precision of HPLC separation was determined by daily analysis over three days of a patient sample with monomeric prolactin only, a patient sample with predominantly macroprolactin and the high prolactin adjustor supplied by Siemens (Siemens Healthcare Ltd) for the Immulite 2000, comprising of monomeric prolactin.
Sixteen patients with macroprolactin ranging from 0% to 90% of total prolactin were analysed by both HPLC and GFC and the monomeric prolactin results of the two methods were compared using Passing and Bablok linear regression analysis.
Results
The elution profile of macroprolactin forms on GFC is shown in Figure 1. Macroprolactin eluted in fractions 6–8 and monomeric prolactin in a broader peak from approximately fractions 10–16. Big prolactin was not well resolved in this short-column GFC method. The time taken from injection to collection of the final fraction in the GFC method was 23 min. The elution profile of macroprolactin forms on HPLC is shown in Figure 2. Monomeric prolactin eluted as expected between MW markers myoglobin (17 kDa) and ovalbumin (44 kDa). Macroprolactin (∼150 kDa) eluted close to γ-globulin (158 kDa). Big prolactin eluted between the macroprolactin and monomeric prolactin peaks. The time taken from injection to collection of the final fraction in the HPLC method was 16 min. The HPLC method, unlike PEG precipitation, was also able to differentiate big prolactin from both macroprolactin and monomeric prolactin.
Prolactin elution profiles in three patients on gel filtration chromatography Prolactin elution profiles in two patients on high pressure liquid chromatography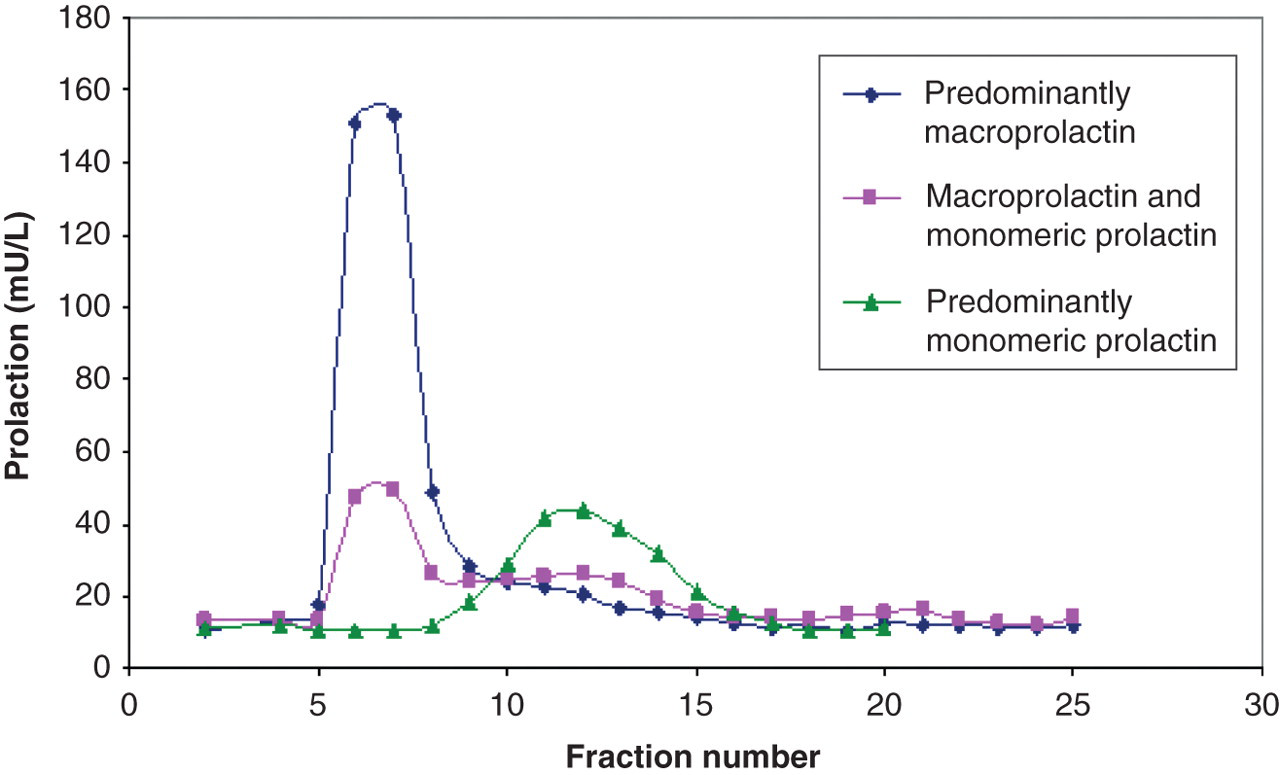
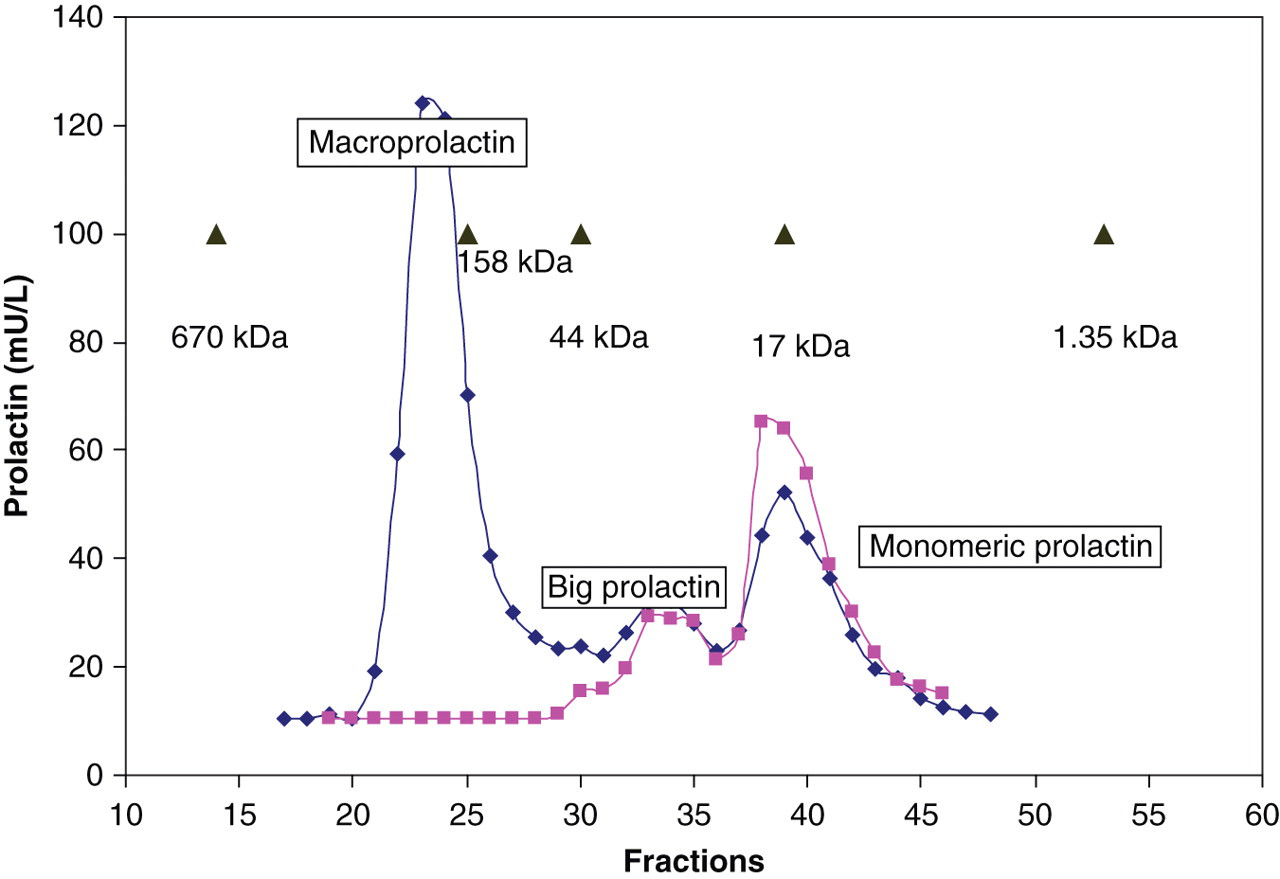
Analysis of the macroprolactin peak from a patient with macroprolactinaemia confirmed the presence of both prolactin and IgG. The precision of the HPLC method was <10% coefficient of variation (CV) for all prolactin forms; a CV of 2.9% for the sample with monomeric prolactin alone, a CV of 5.6% for the high prolactin calibrator and a CV of 7.5% for the sample with predominantly macroprolactin. Linear regression for the linearity study demonstrated a correlation coefficient of 0.997. The Passing and Bablok regression equation for monomeric prolactin percentage by the two chromatographic methods was as follows: HPLC = 1.182GFC − 1.459 (Figure 3).
Comparison (Passing–Bablok regression) of percentage monomeric prolactin measured by high pressure liquid chromatography (HPLC) and gel filtration chromatography (GFC)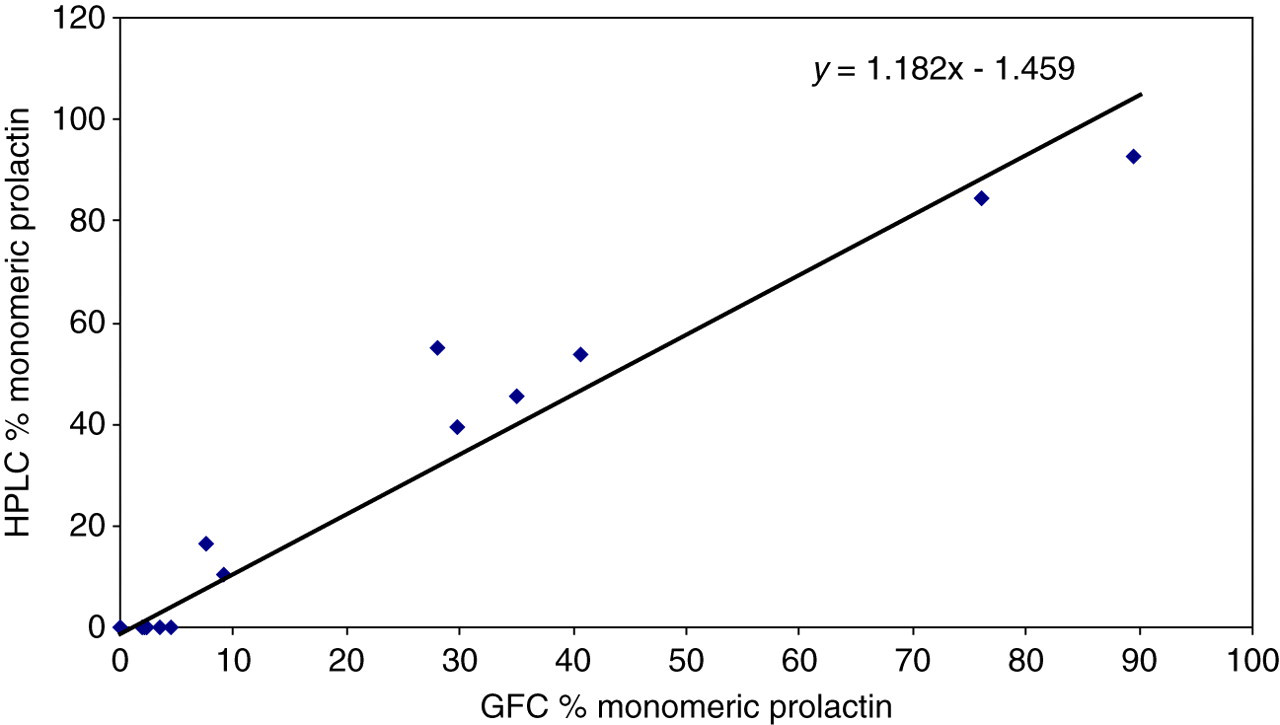
Discussion
While PEG precipitation is the most widely used method to screen for macroprolactin, it is not very accurate. 3 There is currently debate about the interpretation of PEG macroprolactin screening results. 5 Reporting results simply as a percentage of total prolactin recovered after PEG treatment, generally utilizing a cut-off point of 40% recovery in the supernatant to identify macroprolactinaemia from true hyperprolactinaemia, has limitations since increased monomeric prolactin may co-exist with the presence of macroprolactin in some patients. 1,5 Thus some authors advocate the use of post-PEG reference intervals to determine if there is a true elevation in monomeric prolactin, as this is the bioactive and therefore pathologically important entity. 1 Gibney et al. 6 identified macroprolactin in 22% of the population, with the incidence being 17% of samples with serum prolactin >3000 mU/L and 27% with serum prolactin <700 mU/L. They demonstrated that the co-existence of macroprolactin and elevated monomeric prolactin is best identified with the use of post-PEG reference intervals. While GFC may be the reference method for separation and quantification of prolactin, it is complex, time-consuming and therefore not suitable for routine application. Therefore, in our laboratory, we have used a modified GFC method using a short column; this method has reduced resolution and separates only macroprolactin from monomeric prolactin, not big prolactin. We have developed a HPLC separation method for separating prolactin species that is more rapid (16 compared with 23 min from injection to collection of the final fraction) and less labour-intensive than our short-column GFC, allowing it to be utilized for routine service in a clinical laboratory. Long-column GFC is able to separate all three prolactin species; however, the time from injection to collection of the last sample is approximately two hours. 7 This HPLC method is able to separate all three prolactin species, monomeric, big prolactin and macroprolactin, whereas PEG causes precipitation of both big- and macroprolactin. 7 However, the HPLC method, like GFC, still relies on immunoassay quantification of prolactin on multiple fractions of the eluate for each patient sample, and thus should be reserved for selected cases. The agreement between the HPLC and the GFC methods was less than expected, given the similar (size exclusion) principle used by both methods. This may be explained by the short GFC column used in our laboratory in order to reduce the elution time, which has led to the lack of resolution of big prolactin.
In conclusion, we have developed a size exclusion HPLC method to separate rapidly the three different prolactin forms, and which is suitable for routine application. In our laboratory, patient samples with prolactin above the upper reference limit undergo PEG precipitation. 1 If the post-PEG prolactin is above the post-PEG (monomeric) upper reference limit, they are reviewed by the chemical pathologist and considered for HPLC analysis based on clinical requirement. For example, HPLC is performed if there is discrepancy between the clinical presentation and the post-PEG prolactin concentration.
DECLARATIONS
