Abstract
The National Institute for Health and Clinical Excellence (NICE) has recently reviewed optimal means of early recognition and initial management of ovarian cancer, the leading cause of gynaecological death in the UK. The NICE guidelines state that general practitioners should measure serum CA125 in women presenting with persistent and continuous symptoms suggestive of ovarian cancer (e.g. abdominal distension, early satiety and loss of appetite or pelvic pain). If CA125 is ≥35 kU/L, the general practitioner should arrange an ultrasound scan of the abdomen and pelvis to enable calculation of the risk of malignancy score (RMI). Women with an RMI score of ≥250 should then be referred to a specialist multidisciplinary team. Successful implementation of these guidelines requires close liaison between primary care and laboratory medicine to ensure that CA125 is requested as a diagnostic aid only for women meeting the criteria stated in the guidelines. Preanalytical criteria must also be met and it is essential that both requestors and patients be aware of other possible causes of increases in CA125, as well as the fact that it is not raised in all patients with ovarian cancer. Guidance is being prepared to enable appropriate interpretation and implementation of the NICE guidelines. Continuous audit of the outcomes will also be essential to determine the effectiveness of this approach to early detection of ovarian cancer.
Introduction
Ovarian cancer is the fifth commonest cancer in women in the UK and has the worst prognosis of all gynaecological cancers: two-thirds of the 6800 women diagnosed annually in the UK will die within five years of diagnosis, reflecting an overall survival rate of about 35%. 1 Marked survival differences between countries are still evident in the most recent data available from the EUROCARE-4 (EUROpean CAncer REgistry-based study on survival and CARE of cancer patients), a cancer epidemiology research project on survival of European cancer patients, which has analysed three million adult cancer cases from 82 cancer registries in 23 European countries. 2 The significantly lower survival rates for ovarian cancer observed in some countries, including England and Ireland, 2 are generally attributed to late diagnosis, with as many as 70% of ovarian cancers already advanced (The International Federation of Gynecology and Obstetrics [FIGO] Stage III or IV) at diagnosis and therefore difficult to treat. The recently published National Institute for Health and Clinical Excellence (NICE) clinical guidelines on the recognition and initial management of ovarian cancer 3,4 are intended to address this.
It is, however, essential to note that the NICE guidelines relate to symptomatic women presenting to primary care. They do not in any way constitute an invitation to screen asymptomatic women for ovarian cancer. This quite separate issue is the subject of two major ongoing prospective clinical trials, the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS) 5 and the UK Familial Ovarian Cancer Screening Study (UKFOCSS). 6 These trials are assessing the impact of screening on ovarian cancer mortality in 202,638 postmenopausal women (50–74 years old) (UKCTOCS) and in 5000 women (≥35 years old) at high risk of ovarian cancer because of a strong family history or inherited genetic predisposition (UKFOCSS). Other aspects of such population screening – including physical and psychological morbidity, compliance and resource implications – are also being comprehensively evaluated in both trials. The UKCTOCS trial compares an ultrasound group (annual screening with transvaginal ultrasound) and a multimodal group (annual screening with serum CA125 as the primary test and ultrasound as a secondary test) with a control ‘no screening’ group. CA125 is interpreted using a ‘risk of ovarian cancer’ (ROC) algorithm involving initial as well as serial CA125 measurements. UKFOCSS involves measurement of CA125 every four months, interpretation using the same ROC algorithm, and transvaginal ultrasound annually or earlier if the ROC increases. Both are population screening trials, in which all subjects are asymptomatic at entry, and are not due to report until 2015. The results of those trials will add to those from the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening randomized controlled trial in the USA. Recently reported results from the PLCO trial, which differed significantly in design from the UK trials, suggest no reduction in ovarian cancer mortality when CA125 and transvaginal ultrasound are compared with usual care. 7
How do CA125 measurements currently fit into the typical patient pathway?
Together with numerical scores relating to ultrasound results and menopausal status, CA125 measurement enables calculation of the risk of malignancy index (RMI), which is used worldwide to assess the risk that an individual woman has ovarian cancer (Box 1). 8,9 The RMI has been successfully used for more than two decades to triage postmenopausal women with known ovarian cysts for further investigation and management, following a protocol similar to that shown in Table 1. 9 Patients at high risk of ovarian cancer, i.e. those with an RMI ≥250, are referred to a specialist multidisciplinary team for surgery performed by a gynaecological oncologist in a cancer centre (Table 1). With an RMI cut-off of 250, a sensitivity of 70% and specificity of 90% can be achieved in patients with ovarian cysts, so relatively few women with benign cysts will be inappropriately referred to cancer centres. 9 However, CA125 is raised in only about 50% of early Stage I ovarian malignancies so some of these will be missed. In addition, it is well recognized that ultrasound cannot always readily differentiate benign from malignant lesions and that reporting of RMI characteristics is dependent on the skill and experience of the ultrasonographer.
Calculating the risk of malignancy index (RMI) (after the recommendations of National Institute for Health and Clinical Excellence [NICE] 3 and the Royal College of Obstetricians and Gynaecologists) 9
Ultrasound scans are scored one point for each of the following characteristics: multilocular cyst; evidence of solid areas; evidence of metastases; presence of ascites and bilateral lesions.
How will the NICE recommendations regarding CA125 measurement influence current practice?
The NICE guidelines essentially reinforce previous recommendations from the UK Department of Health, 10 but differ in stating more explicitly that CA125 should be requested in primary care in women – especially those over 50 years old – who present with symptoms suggestive of ovarian cancer (Box 2), provided these symptoms are persistent and continuous. The same symptom index has previously been studied by a questionnaire in 116 women with epithelial ovarian cancer and 209 control women. 11,12 Although the study numbers were small, the symptom index was positive in 66% of women with ovarian cancer, in 31% of women with benign cysts (which also require investigation as indicated above) and in only 7% of women on routine screening. The frequency with which symptoms must be observed to prompt measurement of CA125 according to the NICE guidelines – which state that women with ovarian cancer experience some of these symptoms more frequently, more severely and more persistently than women who do not have the disease – should help to exclude those premenopausal women who experience cyclical abdominal distension or bloating as part of their monthly menstrual cycle.
Symptoms that should prompt measurement of CA125 according to the National Institute for Health and Clinical Excellence (NICE) guidelines 3
CA125 measurement is recommended only if symptoms are frequent (e.g. more than 12 times a month) and include:
Persistent abdominal bloating or distension; Feeling full (early satiety) and/or loss of appetite; Pelvic or abdominal pain; Increased urinary urgency and/or frequency.
What are the major caveats to the use of CA125 in early detection in primary care?
While early measurement of serum CA125 in the patient pathway (Figure 1) is an attractive option, providing a potentially cost-effective means of selecting women for pelvic ultrasound, it is critically important that requestors are aware of the many causes of raised CA125 other than ovarian cancer, some of which are listed in Table 2. 13 Particularly in premenopausal women, very careful consideration must be given as to whether elevations in CA125 are likely to reflect other underlying conditions. Failure to do so is likely to cause unnecessary anxiety for some women as well as leading to inappropriate requests for further investigation with significant cost implications. Reinforcing this, measurement of CA125 is only recommended by NICE in women with truly persistent symptoms that raise the suspicion of ovarian cancer, and particularly in women over 50 years of age. The majority of ovarian cancers (90% in 2007) are diagnosed in women over 45 years old, with incidence rates higher in postmenopausal women and highest in women in the 60 to 64 year-old age group. 3 CA125 is somewhat more reliable in these older age groups, with raised serum CA125 in younger women less likely to be related to a diagnosis of ovarian cancer. 3 However, CA125 is not raised in up to 30% of women with ovarian cancer, especially those with potentially curable early-stage disease. A CA125 result within the reference interval should never be used to exclude ovarian cancer.
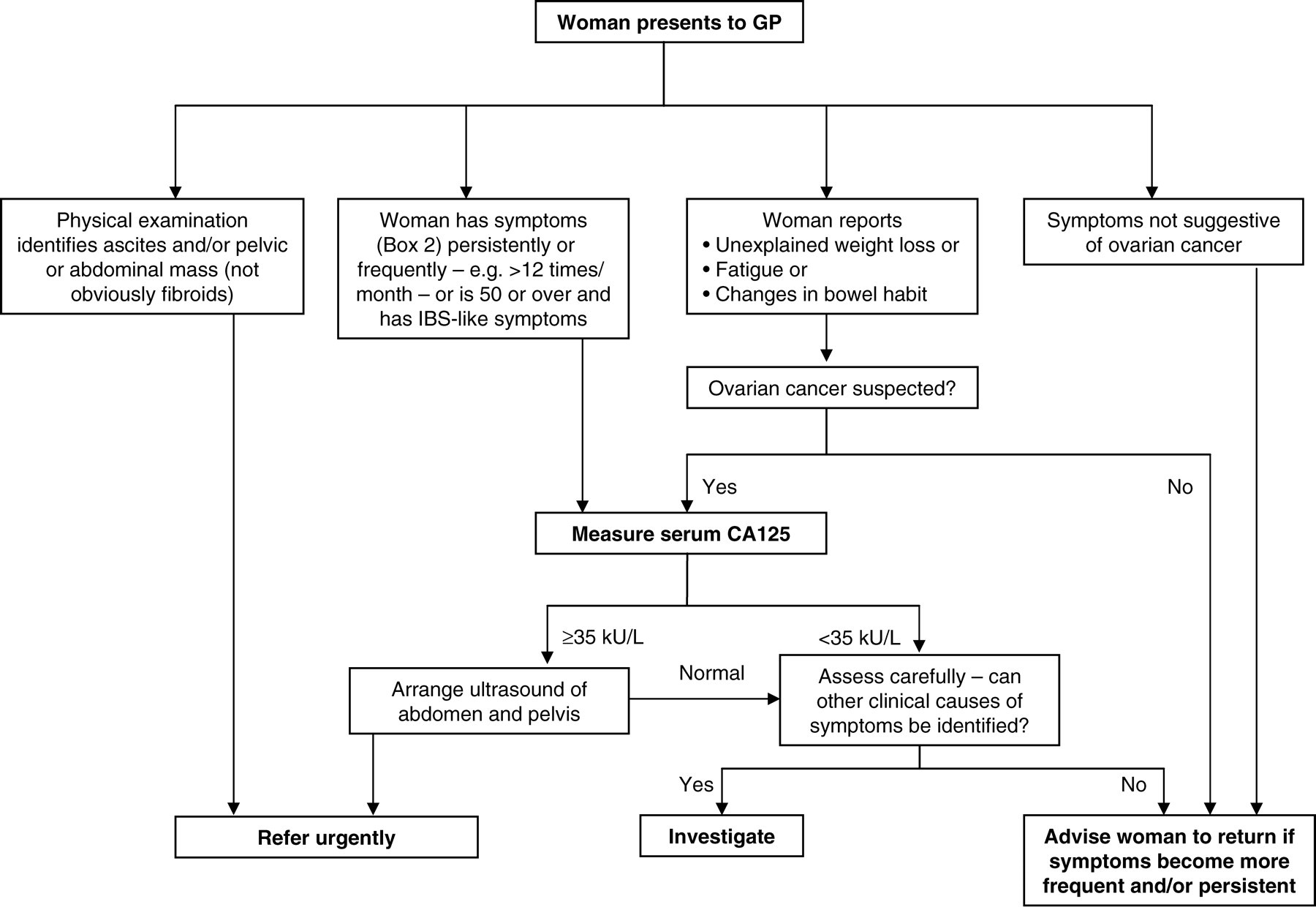
National Institute for Health and Clinical Excellence (NICE) guidelines for detection of ovarian cancer in primary care. GP, general practitioner; IBS, irritable bowel syndrome. Adapted from CG 122 Ovarian cancer: the recognition and initial management of ovarian cancer (2011), London: NICE. See
Benign and malignant conditions other than ovarian cancer which may cause elevations in serum CA125 that may result in incorrect interpretation 13 *
*Lists not exhaustive
What is the rationale behind the NICE referral guidelines?
The results of earlier studies evaluating the risk of ovarian cancer in women presenting with symptoms to primary care, 1 indicate that only 0.23% of these women will have ovarian cancer. 3 Therefore, referral of these women to secondary care on the basis of symptoms alone would mean that only one in 500 women will have ovarian cancer. 3 By including a raised serum CA125 or suspicious ultrasound scan as a criterion for referral, this ratio can be increased to one in 157, and 3% of women with ovarian cancer will be missed and subsequently not referred. However, if both CA125 and ultrasound are abnormal, one in 26 women referred to secondary care will have ovarian cancer but 78 women with ovarian cancer will be missed. This model led the NICE guideline development group to conclude that the third strategy meets requirements for cost-effectiveness within the context of the UK National Health Service. The view of the group was that although with this approach 34% of women with ovarian cancer will not be referred at initial presentation, these women should be identified when the woman returns if symptoms become more frequent (Figure 1). 3 A follow-up appointment six weeks after a normal CA125 test, to enable symptoms to be reassessed, has since been recommended in a recent letter to the BMJ from representatives of major organizations that practice, promote and advance gynaecological oncology in the UK. 14 It will be self-evident that sufficient staff resources should be available in primary care to provide support for patients going through these unavoidably stressful procedures. Studies in men undergoing screening for prostate cancer with prostate-specific antigen (PSA) have previously demonstrated that false-positive results can have an important psychological impact. 15
How can laboratory medicine specialists contribute to guideline implementation?
As is clear from the above, and echoing comments made in the same BMJ letter, 14 there are significant drawbacks to the first-line use of CA125 in early detection of ovarian cancer. These reflect the fact that – as is also the case for PSA in the early detection of prostate cancer 13 – CA125 is by no means an ideal test. However, it is the best serum marker currently available for ovarian cancer and the NICE recommendations are likely to help fast-track some ovarian cancer patients into the appropriate secondary care pathway earlier than might otherwise have been the case. It is salutary to note that 29% of newly diagnosed ovarian cancer patients come through as emergency presentations. 16 As observed in a recent editorial in the Lancet, 17 the strategy proposed, although not perfect, should raise awareness of ovarian cancer and help to promote its early diagnosis in primary care. For successful implementation, however, absolute clarity both about what the NICE guidelines actually state and of the caveats of CA125 measurements is essential prior to requesting CA125 measurement. 14 Senior staff working in laboratory medicine are in an ideal position to work with colleagues in primary care to ensure that CA125 is requested as a diagnostic aid only for women meeting at least one of the criteria listed in Box 2. This includes ensuring that those requesting CA125 are aware of the many conditions other than ovarian cancer that may increase it, sometimes transiently (e.g. concentrations may increase as much as two to three-fold during menstruation) (Table 2). It is clearly unhelpful, for example, to measure CA125 in a patient who has underlying cardiac failure (in whom serum CA125 levels may be in excess of 1000 kU/L), risking diversion of that patient into a completely inappropriate care pathway. Similarly high levels can be seen in patients with liver and lung disease, as well as others. It is essential that users remember that a result within the reference interval does not necessarily exclude ovarian (or other) malignancy and that patients are aware of this too. It would also be helpful to provide information about how the RMI is calculated as this may not be familiar to all non-specialists. Anecdotally, a recent informal poll of 40 general practitioners (GPs) in Lothian found that none were certain what it comprised. This is perhaps not surprising since a GP with an average size practice may only see one case of ovarian cancer every five years. 3
Publicizing such information through laboratory handbooks, particularly those available electronically, is of course essential, although ideally information needs to be available at the time the request is made. Links to electronic requesting systems where feasible provide an excellent means of achieving this. Through the Pathology Harmony initiative in the UK, 18 a quick reference card is currently being prepared relating to the most appropriate use of commonly requested tumour markers, including CA125. The intention is that the card will be made freely available in both printed and electronic form to requestors in both primary and secondary care. Appending comments to laboratory reports also provides a convenient means of reminding users of the appropriate use of CA125, albeit usually after the test has been requested. Above all, laboratory staff should interact with their clinical users whenever possible – whether individually, at clinical meetings or elsewhere – to encourage optimal local implementation of the NICE guidelines.
Further research into diagnostic methods and audit of the impact of the NICE guidelines in detecting ovarian cancer in the community are key priorities. The guidelines repeatedly emphasize how little solid evidence is available on a number of important and highly relevant clinical and laboratory issues, e.g. the lack of data comparing the efficacy of serum CA125, morphological ultrasound and pelvic examination in early detection of ovarian cancer. Prioritizing topics requiring further research in this and other areas would be highly desirable. Combination of measurement of serum CA125 and human epididymis protein (HE4), for example, can be used in a predictive model called the Risk of Ovarian Malignancy Algorithm (ROMA) to estimate the risk of epithelial ovarian cancer in women presenting with a pelvic mass. 19 The ROMA algorithm reportedly achieves a significantly higher sensitivity in this application than the RMI. 19 Interestingly, results of a recent study, in which 104 women with a pelvic mass and 49 healthy controls were enrolled, suggested that HE4 on its own had improved diagnostic performance as compared with CA125 and also performed slightly better than ROMA. 20 However, neither HE4 nor the ROMA model has yet been evaluated in a primary care setting and neither was considered by the NICE guideline-development group.
The RMI itself provides laboratory medicine specialists with a major opportunity to collaborate with radiologist colleagues to improve reporting procedures. Currently, typical ultrasonography reports can make calculating the RMI difficult, because they do not routinely include comments on each of the individual scoring characteristics required, i.e. the presence of multilocular cysts, any solid areas, metastases, ascites or bilateral lesions (Box 1). If ultrasonographers nationally agreed to modify their reports to meet the needs of the NICE guidelines and then to work with laboratories providing CA125 measurements to enable clear and comprehensive reporting of the RMI to GPs, this could represent a major improvement in practice.
However, an immediate priority in implementing these guidelines will be careful audit of the outcomes. Even when screening is targeted at patients selected on the basis of clinical features to have an increased probability of having ovarian cancer, the use of a low sensitivity, low specificity test such as CA125 could be said to push the boundary of what is normally regarded as acceptable in terms of screening criteria. 21 Continuous audit of the outcomes to determine the effectiveness of this approach to early detection of ovarian cancer will be absolutely essential, and will again require close collaboration between clinicians and the laboratory. Laboratories must grasp this opportunity to interact with general practitioners to influence this important care pathway.
DECLARATIONS
