Abstract
An increasing number of toxicology laboratories are choosing to expand the services they offer to include hair testing in response to customer demands. Hair provides the toxicologist with many advantages over conventional matrices in that it is easy to collect, is a robust and stable matrix that does not require refrigeration, and most importantly, provides a historical profile of an individual's exposure to drugs or analytes of interest. The establishment of hair as a complementary technique in forensic toxicology is a direct result of the success of the matrix in medicolegal cases and the wide range of applications. However, before introducing hair testing, laboratories must consider what additional requirements they will need that extend beyond simply adapting methodologies already validated for blood or urine. Hair presents many challenges with respect to the lack of available quality control materials, extensive sample handling protocols and low drug concentrations requiring greater instrument sensitivity. Unfortunately, a common pitfall involves over-interpretation of the findings and must be avoided.
Introduction
Hair has historically been regarded as an alternative matrix offered as an additional specialist test by a small number of forensic toxicology laboratories already providing routine tests for medicolegal purposes. However, over the past 20 years, there has been considerable advancement in analytical instrumentation improving the sensitivity for the detection of low drug concentrations most commonly associated with hair testing and, in addition, a greater understanding of how drugs are incorporated into hair. As a direct consequence, a growing worldwide network of laboratories now offer hair testing as a complementary technique in forensic and clinical toxicology.
Although the number of laboratories offering hair testing continues to increase and hair samples are now routinely collected during criminal investigations (drug-related deaths, drug-facilitated sexual assault [DFSA], child protection) and for monitoring drug misuse (drug rehabilitation programmes, workplace drug testing), no recognized standardized methodologies exist, resulting in considerable variation in the quality of services offered.
Challenges to expert evidence relating to hair testing are frequently due to unsuitable testing methodologies resulting in inefficient extraction of drugs from hair, poor or limited method validation with unacceptable detection limits but most commonly due to over-interpretation of the analytical findings.
The Society of Hair Testing (SoHT) 1 recognizes that the importance of ensuring standards are implemented to guarantee the future of hair testing and acceptability in courts and has published a number of guidance documents relating to the examination of drugs and doping agents in hair. 2–5 The SoHT are currently drafting guidelines for laboratories providing hair testing services and actively encourage laboratories offering hair testing services to participate in their annual proficiency testing scheme, providing laboratories with an opportunity to monitor their analytical performance.
The advantage of hair as a testing matrix is its ability to provide historical detail of an individual's exposure to drugs following chronic use or a single exposure. In order to fully understand and interpret the results of hair tests, it is important to have some basic understanding of how drugs are incorporated into the growing hair shaft and what factors affect their stability in hair. There are several fundamental principles that must be considered when introducing hair into a routine testing laboratory, including the collection and handling of hair samples, the selection and validation of the appropriate methodologies, and the understanding and subsequent interpretation of the findings. This review aims to provide a useful guide to laboratories planning to or currently offering hair testing services so that they may fully understand the requirements involved with establishing a good quality hair testing service.
Hair anatomy and physiology
Hair is a complex matrix consisting of cylindrical shafts of tightly packed cells with the cuticle forming an outer protective layer and is predominantly composed of sulphur-rich proteins. 6 Cortical cells within the interior structure of the hair shaft contain a variety of chemicals including melanin, the principal pigment of hair. Hair colour varies with the quantity, distribution and type of pigment present. The medulla, the innermost region of hair, consists of a framework of spongy keratin and air spaces and in human hair the medulla may be discontinuous or absent.
Hair follicles are embedded in the epidermal epithelium of the skin with an estimated 80,000–100,000 in the human head alone. 7 A simplified structure of the hair follicle is shown in Figure 1.
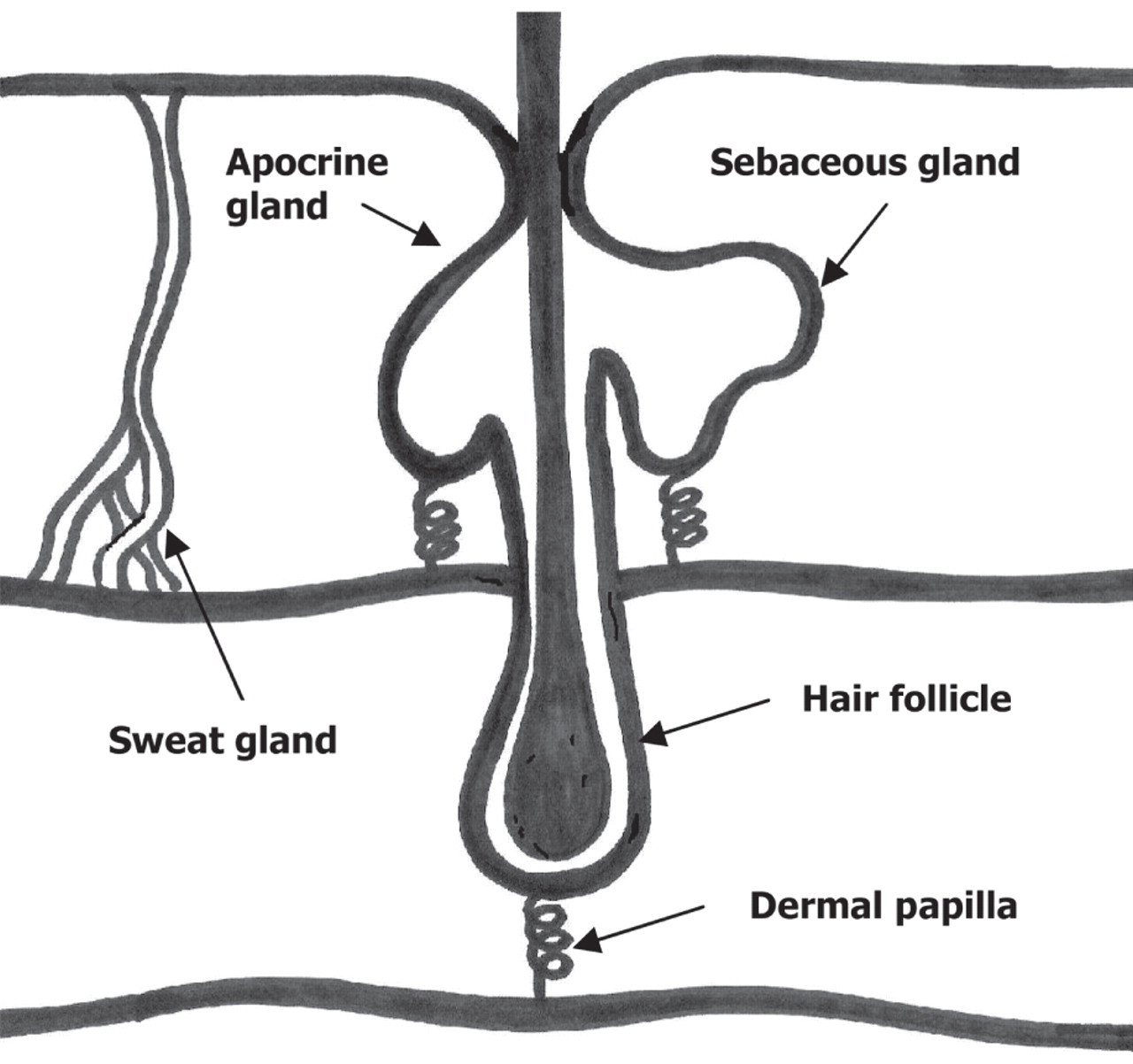
The hair follicle and surrounding structures
The sebaceous and apocrine glands secrete directly into the hair follicle, while the sweat glands secrete near the exit of the hair follicle. 8 Sebaceous glands are located over nearly the entire surface of the body while apocrine glands are localized in the eyelids, axilla (underarm), the external auditory meatus and the perineal region.
There are three basic types of hair on the human body. Vellus hair is fine, short, non-pigmented hair found on the eyelids, forehead and over the body of children and adults. Terminal hair is coarse, long and pigmented with a large cross-sectional area and is found on the scalp, beard, eyebrow, eyelash, armpit and pubic areas. Intermediate hair is found on the arms and legs of adults and has characteristics of both vellus and terminal hairs. Vellus hair is produced by non-sexual hair follicles, which are not influenced by hormones and do not change after puberty. Intermediate and terminal hair follicles are influenced by hormones and change during puberty, e.g. pubic areas, axilla and temple of scalp.
Hair growth
There are three recognized stages in hair growth: the anagen, catagen and telogen phases. On average, 85% of the hairs on the human scalp are in the anagen phase, which is associated with an increase in metabolic activity, cell division and growth. Following this period of active growth, cell division stops, the hair shaft becomes fully keratinized and the bulb begins to degenerate. This is termed the catagen phase, which is a short transitional stage, the length of which varies with the hair type. The telogen phase is a period when there is no hair growth, the follicle is short and can be easily removed by pulling. The length of time that hair is in this phase is dependent on the hair type and increases with age. Other factors affecting hair growth include pregnancy, metabolic and genetic disorders, nutrition and seasonal changes. 9
Table 1 summarizes the published ranges for hair growth rates for different hair types. An average growth rate of 1 cm per month 4 is assumed for head hair. However, this is an oversimplification, with reported growth rates ranging from as little as 0.6 cm per month to as much as 3.36 cm per month. 6 In addition, hair cut from the scalp does not represent recent hair growth as it has been estimated to take approximately 7–10 days for the growing hair to reach the surface of the scalp.
Hair growth rates (cm/month)
Hair sampled from the vertex region of the scalp is preferred because it has the fastest growth rate and the largest percentage of follicles in the anagen phase. Pubic hair grows more slowly than scalp hair and has a longer telogen phase while beard hair is thick and has the slowest growth rate.
Drug incorporation
To accurately interpret hair test results requires knowledge of the mechanisms by which drugs are incorporated into hair, and the factors which affect their binding. A great deal of research is ongoing in this key area; however, the exact mechanisms are not fully understood. During the anagen growing phase, there is an increase in metabolic activity, cell division and growth. It is at this time that drugs and trace elements are thought to be incorporated into hair. 14
Routes of incorporation
The main routes of incorporation into hair are from the blood supply, sebum, sweat and from external contamination. 6,15 It is generally accepted that each of these routes contributes to drug incorporation but to what extent each route exerts greater influence is still not clear. 16 Passive diffusion of drugs directly from the blood supply at the base of the hair follicle 17 would in theory result in a drug concentration in hair proportional to the drug concentration in blood at the time of hair synthesis. 18 However, incorporation from the blood supply alone fails to explain the different metabolic profiles seen in hair and blood. Cocaine, heroin and its primary metabolite, 6-monoacetylmorphine, are less commonly detected in blood compared with their metabolites, but are the primary analytes found in hair.
Sweat and sebum have been proven to contain drugs 19,20 and, as they bathe the growing hair follicle and surface of the hair shaft, will contribute to incorporation. Passive exposure may result from being within the same environment as individuals smoking drugs (e.g. heroin, crack cocaine, cannabis), handling drugs directly or touching surfaces contaminated with drugs and then touching your hair. Children living in this type of environment are particularly at risk from exposure to drugs 21,22 and women who continue to use drugs during their pregnancy expose the unborn fetus in the latter months of gestation. 23,24
The role of external contamination has been extensively investigated by researchers who have attempted to recreate various scenarios to mimic possible contamination risks. Drugs have been demonstrated to contaminate drug-free hair exposed to smoke-containing cannabis, 25 blood-containing cocaine 26 and from direct application of cocaine 27 to the hair sample.
Controlled dosage studies with animals and humans support the concept of a linear relationship for certain drugs, 17,28–31 but the vast majority of studies have concluded that a linear relationship does not exist between dose and drug concentrations in hair. 32–36 Intersubject variation is the most likely explanation but there are also a number of other systemic variables, including hair colour and cosmetic treatments, and their role will be discussed in more detail.
Hair pigmentation
Hair has different affinities and binding capacities for various drugs and the binding mechanisms may be unique for each drug. 37,38 Factors affecting the incorporation and binding of drugs include pKa, non-protein-bound drug molecules, size, lipophilicity, 39 structure and melanin affinity. 10,40 Researchers have demonstrated that basic drugs incorporate into hair to a greater extent than neutral or acidic drugs. Drugs such as amphetamines and cocaine are present in higher concentrations in hair compared with benzodiazepines and cannabinoids.
A number of studies have investigated the incorporation of different drugs into hair involving animal models which have provided useful information on the role of melanin. 41–44 Darker hair contains more pigmentation (melanin) than light coloured hair and so animals with both pigmented and non-pigmented hair were administered drugs of different basicity. The findings supported the greater binding affinity of basic drugs to melanin as they were present in higher concentrations in pigmented hair. Neutral and acidic drugs demonstrated no greater affinity to pigmented or non-pigmented hair.
It is important to remember when relating these findings to human hair that animal and human hair differs with respect to growth cycles and structure which may result in different drug distribution. A recent study reported differing incorporation profiles of fatty acid ethyl esters in rat hair compared with human hair. 45
Pötsch et al. 46 proposed the ‘biochemical concept’ to explain the endogenous incorporation of drug molecules into growing hair. This concept tentatively explains the high parent drug to metabolite ratio in hair, the dependence of incorporation on the physico-chemical properties of the drug, the incorporation of drugs into non-pigmented hair and the dependence of drug content on hair pigmentation.
Drug stability
Unlike blood, urine and other conventional biological matrices, chemical compounds incorporated into the hair matrix remain relatively stable in hair for many months. Centuries after their death, arsenic was detected in hair from the Emperor Napoleon Bonaparte 47 and cocaine in the hair of Peruvian mummies. 48
The stability of drugs in hair is dependent on the morphology and physicochemical properties of hair. Diffusion of drugs, in the presence of water, into and out of hair provides a route for external contamination or loss of drug concentration. 49 This was also illustrated using fluorescence microscopy to examine Rhodamine B-stained hair fibres. 50 In addition, hair is subjected to daily exposure to sunlight and weathering and the cuticle is susceptible to damage or can be destroyed by heat, light and chemical treatments.
Several researchers have investigated the effect of cosmetic treatments on hair. 51–56 Although the studies carried out varied with respect to the drugs and the cosmetic treatments investigated, all were in agreement that dyeing, perming and bleaching have a deleterious effect on the drug concentration detected in hair.
Jurado et al., 53 focused on the effects of bleaching and dyeing on the concentrations of opiates, cocaine, cannabinoids and nicotine detected in hair. The decrease in drug concentrations was more prominent for bleached hair than dyed and damaged hair showed significantly lower levels. Daily shampooing does not significantly affect drug concentration. 56,57
In addition to cosmetic treatments, exposure to light has been shown to reduce cannabinoid concentrations in hair. 58
Sample collection and preparation
Hair cut as close as possible to the scalp from the vertex region of the scalp is recommended 2 and is illustrated in Figure 2.
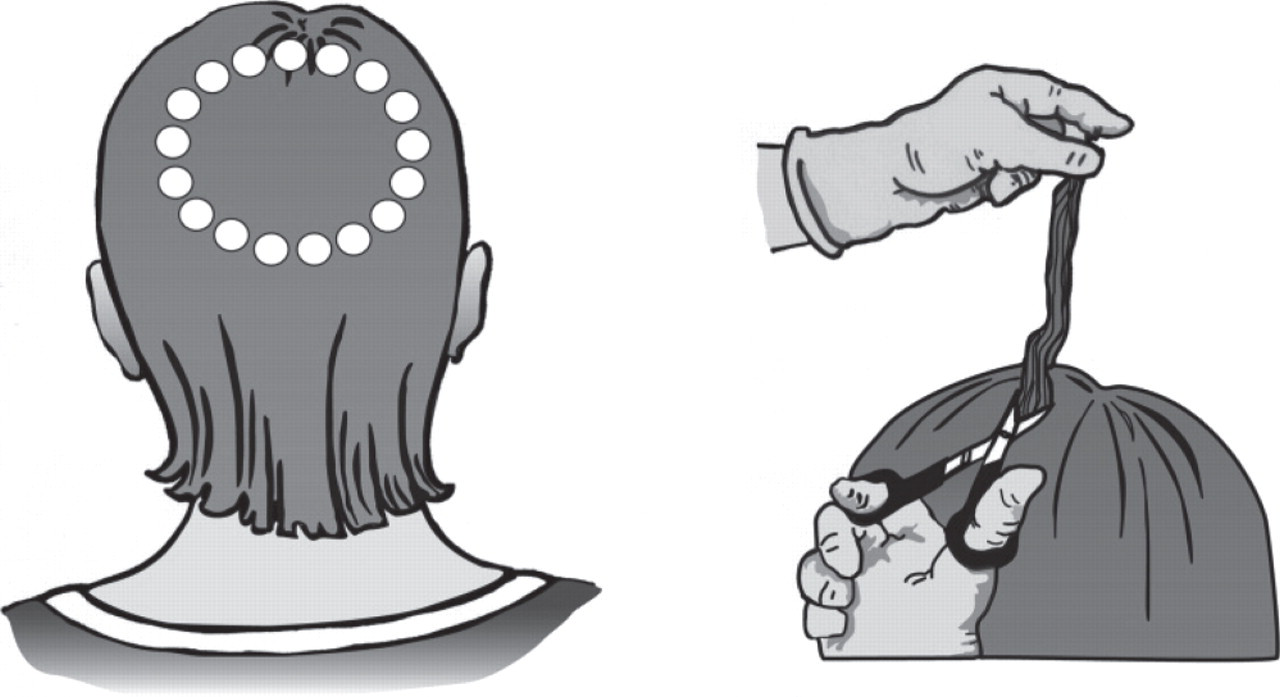
Vertex region of the scalp
The amount of hair required for analysis is often described as a ‘lock of hair’ or a pencil thickness of hair. It is important to collect sufficient hair in order to carry out routine tests, although concerns are often raised in relation to leaving a visible ‘bald patch’. This is of particular concern with small children or individuals with baldness or thinning hair. In these cases, collection of smaller hair samples from multiple sites focusing where possible around the vertex region is acceptable. The individual locks should be combined in alignment with root end identified.
Collection of scalp hair does not require specialist facilities but should be carried out by a competent collector who must observe chain of custody procedures and avoid any potential for contamination of the sample(s). Where head hair is not available, alternative collection sites should be considered, including pubic, underarm and beard hair. Collection of intimate samples requires consideration for the privacy of the donor while ensuring that the integrity of the collection process is not compromised.
Pubic hair, although not subjected to environmental or cosmetic effects to the same extent as head hair, is not always available and both urine and gland secretions are still potential sources of contamination. Beard hair is subject to the same environmental contamination as scalp hair and although sebaceous secretions may not contaminate beard hair, the specimen is likely to contain pieces of epidermis when obtained by shaving.
Collection procedure
A standard hair collection kit should contain the following components:
Chain of custody form; Foil and collection envelope; Security seals; Evidence bag; Transportation envelope (optional); Instructions for collection of a hair sample.
The chain of custody form documents details relating to the collection and handling of the sample. Table 2 summarizes the information that should be recorded on the chain of custody form.
Information recorded on a chain of custody form
Duplicate or carbon copies of the form may be required if a number of agencies are involved in the process, and in workplace drug testing, anonymized copies of the form may be required for despatch with the sample to the laboratory for analysis. Registered mail or courier service is recommended to ensure chain-of-custody. In addition, collection kits utlized for workplace drug testing and many criminal investigations require collection of two hair samples and therefore the collection kit will require duplication of several components. Further details relating to specific guidelines for workplace drug and alcohol testing of hair have been published. 59
The following step-by-step guide summarizes the collection process:
Step one: Cut a lock of hair close to the scalp from the vertex region and align the hair identifying the root end (see Figure 3).
Step two: Fold the foil lengthways and avoid folding in the middle as this will kink the hair making it difficult to handle (see Figure 4).
Step three: Place the foil-wrapped hair sample within the collection envelope, seal, initial and date.
Step four: Place the completed chain of custody form and sealed collection envelope into the evidence bag and transportation envelope and send to the laboratory for analysis.
The collection of postmortem hair samples can pose a challenge depending on the condition of the deceased's hair. Ideally, hair samples should be dry and collected with the root end of the hair identified, but this is not always possible. Hair heavily soiled with blood is commonplace following road traffic accidents or violent assaults and heavy soiling from body fluids is also routinely encountered in cases of decomposition. In these circumstances the standard hair collection kit foil and envelope may be inappropriate and an alternative more robust collection vessel required.
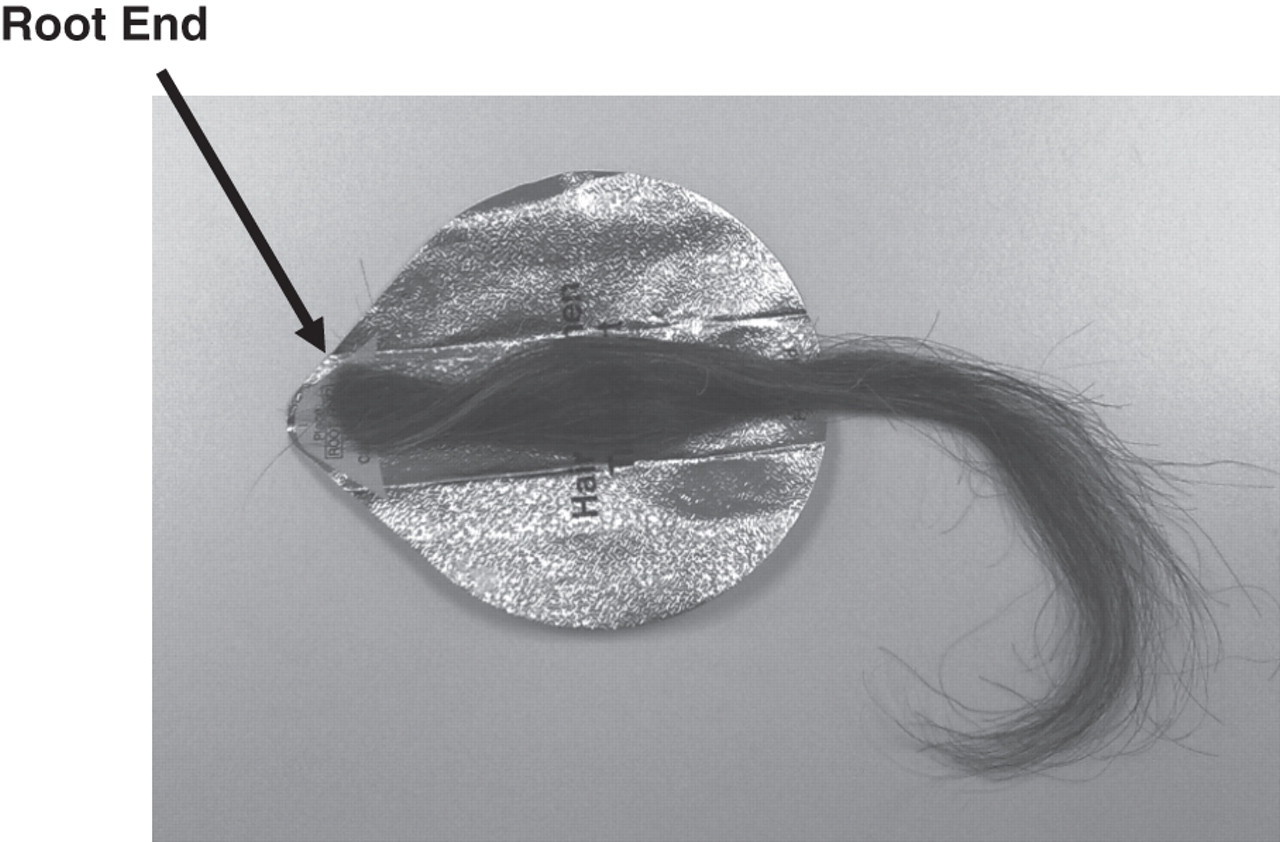
Cut hair aligned with the root end identified. Picture courtesy of Forensic Medicine and Science, University of Glasgow, Copyright © 2011

Fold the foil lengthways to secure the aligned hair. Picture courtesy of Forensic Medicine and Science, University of Glasgow, Copyright © 2011
In some circumstances the analysis of hair roots will help provide additional information relating to drug use prior to death. In addition to the collection of a cut hair sample, the collection of a pulled hair sample with roots intact is also recommended as illustrated in Figure 5.
Collection of a lock of pulled hair to preserve the roots. Picture courtesy of Forensic Medicine and Science, University of Glasgow, Copyright © 2011
Hair samples should be stored at room temperature, preferably dry and in a dark place away from direct sunlight.
Segmental analysis
Hair cut into segments provides an opportunity for a more detailed historical profile of an individual's drug exposure. Hair is cut into measured segments of between 1 and 3 cm prior to washing to remove gross environmental contamination.
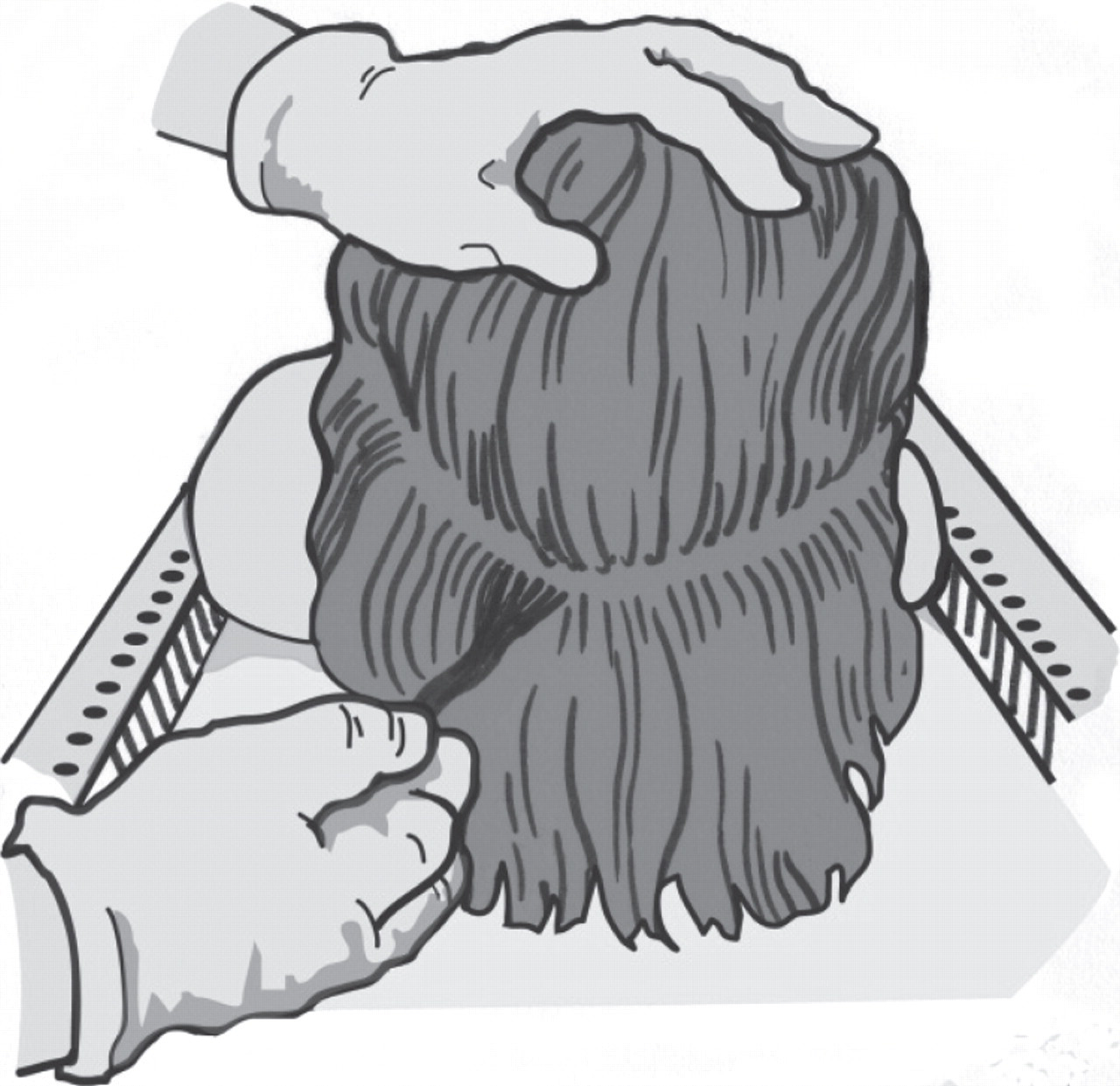
Hair samples soiled with body fluids may require extensive clean-up procedures prior to segmental analysis. Figure 6a illustrates an example of a large matted hair mass submitted for analysis. During the postmortem examination, it was not possible to identify the root end and so the decision was made to submit the entire hair which was already detached from the skull. In this case a piece of scalp was identified, which facilitated the identification of the root end of the hair (as illustrated in Figure 6b) but this is not always the case. Where the root end cannot be identified, a sample of the hair should be analysed in bulk.
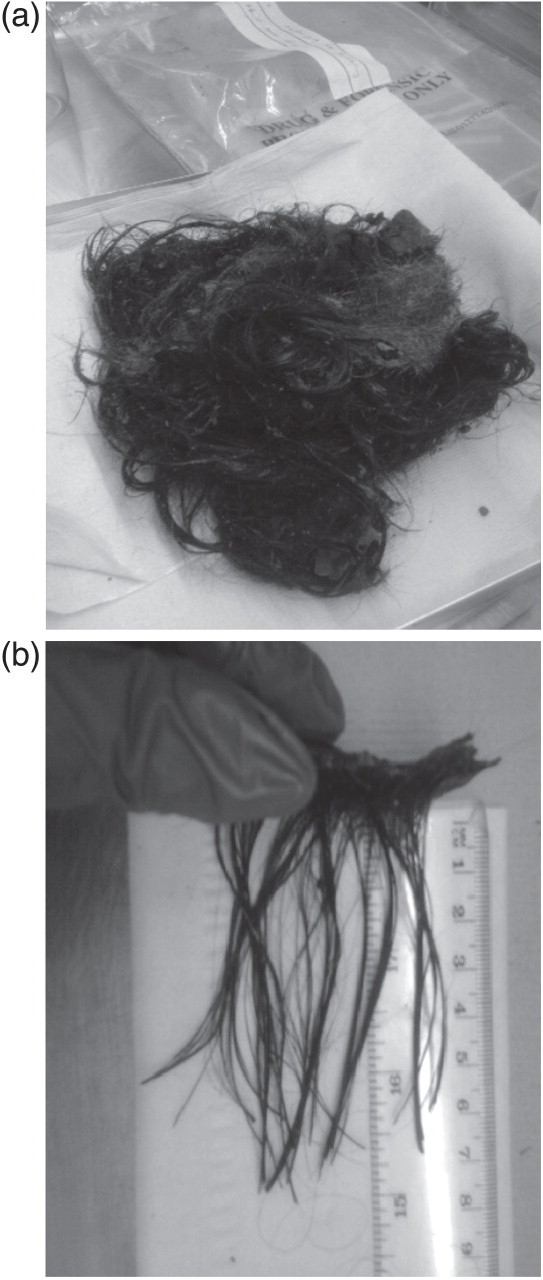
(a) Large matted hair mass collected from a murder victim. Picture courtesy of Forensic Medicine and Science, University of Glasgow, Copyright © 2011.
Washing
Washing hair prior to analysis is an important stage in the sample preparation process not only to clean visibly dirty hair samples but also to remove gross environmental contamination from the surface of the hair that may not be visible to the naked eye. Examples of contamination include hair products (e.g. shampoo, styling gel, hairspray), dandruff, head lice, body fluids and drugs.
The SoHT recommends washing hair with an organic solvent, followed by an aqueous solution and then a final wash with organic solvent. 2,4 Researchers have evaluated numerous combinations of different water-based solutions and solvents to optimize wash procedures. 60–62 Ideally, wash procedures will selectively remove all trace of surface contamination without removing drugs incorporated into the hair shaft. Samples are agitated for a few minutes submerged in each of the wash solutions which are then retained for further analysis. Positive wash solutions indicate external contamination as the likely source but caution must be exercised as a number of wash procedures may actively remove drugs from within the hair shaft in addition to surface contamination. Hair swells when submerged in heated water for extended incubation times (sonicated wash step), resulting in loss of incorporated drugs. The strength of the organic solvent used to wash the hair could also contribute to the extraction of drugs from within the hair.
A complex wash procedure, requiring a number of discriminating criteria to be met during the decontamination process, has been proposed as a means of differentiating between contamination and incorporated drugs. 63 Although a multicriterion approach has potential, this process has not been fully accepted as a standardized methodology. This is further supported by a study involving the analysis of hair samples that were contaminated with cocaine. The participating laboratories employed different wash procedures, some extensive, but they failed to decontaminate the hair. 64
Standardized decontamination wash procedures that will effectively remove any trace of external contamination without actively removing the drugs incorporated into the hair are not currently available and, as such, the possible role of external contamination must be considered when interpreting hair testing findings.
Sample homogeneity
Once the hair samples have been washed and dried, they should be cut into smaller pieces or milled to a powder and then typically 10–50 mg of hair accurately weighed prior to analysis. Increasing the surface area of the hair increases the potential for recovery of drugs from hair. 65 Although use of a ball mill will improve sample homogeneity, it should be limited to preparing larger volumes of hair as the process can result in sigificant loss of sample. This is not a practical option for preparing numerous individual hair samples or segments as the mill requires extensive clean-up procedures between each sample and loss of hair is of particular concern, especially when the majority of hair samples submitted for analysis are limited in volume.
Cutting the hair into small pieces of 2–3 mm in length using a pair of sharp-tipped scissors is by far the most effective and efficient method of hair preparation, although researchers have recently reported an alternative micro-pulverized extraction procedure for small sample volumes. 66,67
Scissors used to cut hair should be cleaned between samples using a water-based detergent and/or wiped clean with a solvent (e.g. methanol).
Analysis
The analysis of hair involves an initial pretreatment step to release the drugs from within the hair matrix prior to either direct analysis (immunological screening) or further clean-up using liquid–liquid or solid-phase extraction. Separation and detection of the analytes of interest is achieved using chromatographic methods coupled to mass spectrometry, e.g. gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-mass spectrometry (LC-MS).
Drug levels in hair are considerably lower than those found in routinely tested matrices such as blood and urine, and although standard bench top single quadrupole systems do have sufficient sensitivity for some hair testing applications, this is not the case for drugs poorly incorporated into hair.
The historical development of hair testing techniques was described by Sachs 68 as having progressed through different phases beginning with the ‘pre-mass selective detectors’ (MSDs) period where techniques used were predominantly immunoassay-based, e.g. radioimmunoassay. 69,70 Following this was the ‘uncritical’ use of MSDs and then the ‘critical evaluation’ of methods involving MSDs from 1993 to 1996. Tandem mass spectrometry systems were available at this time offering increased sensitivity, and a handful of papers were published reporting the benefits, 71,72 but their implementation into laboratories offering hair testing was generally viewed as cost prohibitive.
Over the past decade, methods incorporating tandem mass spectrometry for both screening 73–74 and confirmation 75,76 of drugs in hair have become commonplace, as the price of these instruments has decreased and many laboratories now use them routinely.
Incubation and extraction
Method sensitivity is compromised significantly through the use of inefficient extraction procedures not targeted to specific drugs. Release of the incorporated drugs from the hair matrix is achieved through incubation of the hair in solutions that remove drugs without damaging the hair (methanol or buffered solutions) or result in complete digestion of the hair (alkaline solution). 77
It is important to consider the potential deleterious effect on the drugs in question when choosing the extraction solutions. In vitro hydrolysis of cocaine and heroin has been reported under acidic and alkaline conditions. 60,78
Incubation procedures with methanol on the other hand do not have the same limitations associated with hydrolysis but drug recoveries are lower. 60–65 Researchers have reported the use of a variety of incubation conditions for different drugs and a selection of these are listed below in Table 3.
Selection of extraction conditions for releasing drugs from hair
Laboratories introducing new hair testing methodologies must investigate the efficiency of different incubation conditions to optimize their in-house extraction procedures, while minimizing hydrolysis of labile drugs.
Screening
Radioimmunoassay (RIA) was initially employed for screening hair samples for drugs of abuse, 68,89–91 but the majority of laboratories have now replaced RIA with enzyme-linked immunosorbent assays (ELISA). 92 Immunoassays originally developed for whole blood have been adapted or optimized specifically for hair. 93–95 Immunological screening assays for hair should be sufficiently sensitive to detect low drug levels in hair and should be targeted to the parent drug and also the metabolite, to reflect the drug profile in hair. The hair matrix should not interfere with the assay as this may result in false-positives.
Automation of ELISA has provided high-throughput laboratories with the opportunity to screen large numbers of hair samples for multiple analytes, quickly assigning and eliminating negative samples requiring no further analysis. 96 Increasingly, chromatographic methods are being introduced for rapid screening of drugs in hair. 73,74,97 Although these techniques are expensive and less practical for screening large numbers of samples, they have the advantage of simultaneous screening and identification for a wider range of analytes in a single analysis.
Confirmation techniques
Following incubation to release the drugs of interest from the hair matrix, the resulting extract requires further clean-up using the same techniques employed for blood or urine, namely liquid–liquid extraction or solid-phase extraction. The majority of published hair testing methods are for drugs of abuse, including opiates, 42,60,69 cocaine, 64,87,98 amphetamines 66,67,75,84 and cannabinoids, 58,85,86,99 employing GC-MS or LC-MS-MS.
Although new methods have been reported using specialist analytical instrumentation including quadruple time-of-flight and matrix-assisted laser desorption/ionization time of flight, 97,100 standard instrumentation and method parameters (temperature programme, ions selected for monitoring, collision energies, etc.) that are currently in place within laboratories can also be utilized for hair. The main issues that must be considered are whether or not the method has sufficient sensitivity for low drug levels in hair and whether the target analytes are specific for hair. Table 4 summarizes the target analytes for each of the common drugs of abuse.
Target analytes for common drugs of abuse in hair
In addition to drugs of abuse, methods have been published for steroids, pharmaceuticals and alcohol biomarkers. 62,101–105 It is not the purpose of this paper to carry out a detailed review of the methodologies. A number of review papers and books are already available summarizing the range of analytical methods used for different applications and these are an excellent resource when looking to develop new hair testing methods. 62,106,107
Cut-offs
There are currently no recognized cut-offs employed in hair testing. The SoHT has proposed cut-off values for drugs of abuse and alcohol markers in hair and these are listed in Table 5. These cut-offs are currently under revision and new guidelines are expected to be published in 2011. The US Mandatory Guidelines for Federal Workplace Drug Testing Programs have also proposed cut-offs for hair but have not been uniformly accepted. 108
SoHT proposed cut-offs 4
SoHT, Society of Hair Testing; THC, Δ 9 -tetrahydrocannabinol; THC-COOH, 11-nor-Δ 9 -tetrahydrocannabinol-9-carboxylic acid; BZE, benzoylecgonine; EME, ecgonine methyl ester; CE, cocaethylene; NC, norcocaine
*Alcohol biomarkers ethyl glucuronide (EtG) or fatty acid ethyl esters (FAEEs) can be used independently to assess chronic excessive use of alcohol but both parameters are recommended
Quality assurance
Implementation of quality assurance is recognized as a fundamental principle for all testing laboratories and accreditation to the international standard ISO/IEC 17025
109
is an industry requirement. The United Kingdom Accreditation Service (
In 2008, the Home Office appointed a Forensic Science Regulator whose remit includes ensuring those providing a service to the Crown Prosecution Service are adhering to recognized standards. This includes laboratories offering toxicological analyses and ultimately hair testing. Although the Regulator has specified that forensic science laboratories should adhere to ISO/IEC 17025, the medical laboratories standard (ISO/IEC 15189) is likely to be included. Specific guidance is available from the International Laboratory Accreditation Cooperation for forensic science laboratories 110 and, more recently, for forensic toxicology laboratories from the United Kingdom and Ireland Association of Forensic Toxicologists (UKIAFT). 111
Although drug testing laboratories worldwide recognize the importance of implementing quality systems, a recent study 112 highlighted that only a small proportion of forensic laboratories throughout Europe are currently accredited to ISO/IEC 17025. The majority of those questioned were planning to introduce accreditation into their laboratory and this was also reported in a survey of toxicology laboratories offering hair testing. 113 The main barrier facing laboratories wishing to introduce accreditation is the cost. A typical European laboratory tests less than 500 hair samples per year and this constitutes a small fraction of the general toxicology tests offered by the laboratory.
The entire accreditation process requires significant financial commitment involving additional staff resources to manage the quality management system, purchasing consumables to carry out additional validation parameters in accordance with ISO/IEC 17025 and the costs of the annual audits (biannual for the first 1–2 years). A small testing laboratory could expect to invest an initial £20–40,000 in set-up costs with a minimum of £10,000 annual fees including expenditure on consumables. 114
Proficiency testing schemes for hair
Accrediting methods for the analysis of drugs in hair represents a significant challenge to the analyst with respect to the lack of certified reference material and limited guidelines on best practice. The SoHT (
Laboratories accredited to ISO/IEC 17025 must participate in proficiency testing schemes compliant to ISO/IEC Guide 43 and purchase reference materials from manufacturers compliant to ISO Guide 34:2009. 117,118 There are currently two European proficiency testing schemes for hair, the SoHT and the German Society of Toxicological and Forensic Chemistry (GTFCh), neither of which are accredited to ISO/IEC Guide 43. However, there are currently no alternatives available and the benefit of both schemes is that the samples are prepared using hair from known drug users.
Quality controls for hair
External quality controls are expensive and many laboratories prefer to prepare their own controls in-house. 119,120 Ideally, quality control samples should be prepared from ‘real’ hair samples collected from known drug users. Preparation of controls in-house can be problematic with issues surrounding homogeneity and lack of availability of old case samples following restrictions imposed by the introduction of the Human Tissue Act (2004) and the Human Tissue (Scotland) Act (2006). 121,122
Purchasing additional proficiency testing scheme samples to use for quality controls is one option available to laboratories and the GTFCh provide this service through suppliers and distributors (Medichem® Diagnostica,
Laboratory guidelines for hair
Guidelines are readily available for urine workplace drug testing, 123,124 doping control in sport 125 and postmortem toxicological investigations. 126 The UKIAFT Forensic Toxicology Laboratory Guidelines cover the areas of postmortem forensic toxicology, human performance forensic toxicology and criminal forensic toxicology. 111 The SoHT is committed to ensuring a consistent approach to the analysis of drugs in hair and the accreditation process with overwhelming support from the SoHT members who already adhere to the current guidelines for best practice in hair testing. 113 Specific guidelines for workplace drug and alcohol testing in hair are also available. 59
Method validation
The accuracy and reliability of test results are frequently challenged during cross-examination in court, resulting in questions relating to the suitability of the laboratory service. The international quality standard ISO/IEC 17025, documents the criteria for testing laboratories wishing to demonstrate their technical competence in generating valid test results. To satisfy the requirements of ISO/IEC 17025, laboratories must provide data to support the use of the method as fit-for-purpose.
In addition to standard validation parameters of linearity, limits of detection and quantification and precision, laboratories must also investigate the robustness of the method, investigate analyte stability, bias and estimate uncertainty in measurement. A number of ISO documents are available to provide guidance (
Applications and interpretation
Hair testing has been offered throughout mainland Europe and the USA for over two decades and is now well established as an alternative to urinalysis. An estimated 27 million employees are tested for evidence of drug misuse each year in the USA. 124 Although urine remains the most widely tested sample, a growing number of employers are choosing to test hair which offers several advantages over urinalysis. 129,130 Hair has the benefit of confirming long-term exposure to drugs over a period of weeks to months depending on the length of hair collected, while urine provides short-term information over a period of hours to several days. However, concern over infringement of individuals' civil liberties has raised questions in relation to the use of hair in workplace drug testing. The argument relates to an individual's choice to use drugs recreationally when not in work and, as such, not of concern to the employer.
Hair is a strong, stable tissue unaffected by adulterants or short-term abstinence and has proved invaluable when re-granting driving licences. 131–133 Drivers in Italy and Germany who have been convicted of being under the influence of drugs while driving have the opportunity to re-apply for their licence after a period of six months. Over this time period, the driver must submit several urine samples for analysis or provide a hair sample for re-analysis to prove abstinence. The disadvantage of using urine as a testing matrix in these instances is that drivers must submit several samples over the assessment period and those who continue to use drugs can provide a drug-free urine sample. Abstaining from drug use in the days prior to sample collection will ensure a negative urine sample, allowing the individual to evade detection of their drug use. In comparison, intermittent abstinence will not affect the results of a hair test.
Additional advantages relate to the collection and storage of hair in comparison to traditional biological matrices. Collecting head hair is less intrusive and causes less embarrassment than observed urine collection and hair does not require refrigeration and can be stored indefinitely. Drugs are relatively stable in hair and, as such, a second representative hair sample can be collected and analysed.
The role of hair testing as an alternative or complementary matrix has expanded across the spectrum of toxicological investigations and Table 6 summarizes key applications of hair testing.
Applications for drug testing in hair
Initially, there was considerable reluctance among the majority of toxicology laboratories in the United Kingdom to offer hair testing and this was centred predominantly on the perception that hair was a technically difficult matrix to handle and analyse, and concerns relating to efficacy. Although a number of issues that were initially raised in relation to contamination and ethnic bias have not been entirely resolved, the past decade has seen a significant increase in the number of laboratories now offering hair testing. Similar in profile to the European laboratories surveyed in 2007–2008, most laboratories in the UK are small providers who already offer forensic toxicology services. 113
Workplace drug testing in the UK is expanding and although urine and oral fluid are the preferred matrices for testing, there are companies who have chosen to test their employees' hair as a complementary technique. By far, the most common applications of hair testing in the UK involve the investigation of drug-related deaths, drug-facilitated crime (DFC), child custody and, most recently, assessment of alcohol abuse.
Drug-related deaths
The ability to detect drugs in hair has enormous potential, particularly with respect to postmortem hair samples. In many cases, the drug use history of the deceased is either not known or not available for the investigation of the death. Drug levels detected in blood provide information regarding the short-term drug use of the decedent prior to death, while hair analysis provides an invaluable source of information relating to antecedent drug use history in the months prior to death. 135 Establishing evidence of chronic drug use through hair analysis can greatly aid the investigative process and help establish the role of drugs with respect to the cause of death. 136 In addition, providing reliable evidence to support previous drug use through hair analysis can also help family and friends of the deceased, who may have been unaware of the deceased's previous drug use, come to terms with their death.
Establishing abstinence in the months prior to death provides supporting evidence of a lack of tolerance to the drugs implicated in the cause of death. For example, a prison inmate was found dead with a needle in situ and following toxicological investigations, the cause of death was established as heroin intoxication. Although the deceased had a history of heroin misuse, he had successfully completed a detox programme 18 months prior to his death and subsequent and regular urine tests indicated occasional cannabis and diazepam use, but not heroin. Analysis of hair samples collected postmortem confirmed previous use of cannabis and diazepam only in the months prior to death, consistent with anecdotal evidence provided by prison staff and fellow inmates.
The collection and analysis of hair roots has the potential to provide information relating to acute poisoning. Amphetamine, methamphetamine 146 and codeine 147 were detected in hair roots 30 min after administration and 3,4-methylenedioxymethamphetamine was identified in the root bulb of rat hair five minutes after administration. 148 Hair roots collected at postmortem were analysed, confirming the presence of drugs detected in paired postmortem blood samples. 149,150 However, the collection and handling of hair roots is problematic and the majority of hair samples submitted for analysis in postmortem cases are those cut close to the scalp.
Collection of hair roots involves pulling the hair from the scalp while ensuring the roots are aligned. On receipt in the laboratory, the roots are cut from the hair which requires significant skill and patience. It is advisable to prepare hair samples away from air conditioning units and this is particularly important when handling hair roots. The analysis of hair roots may provide crucial information in cases of advanced decomposition where there is limited availability of samples suitable for analysis and justifies the additional effort required to prepare hair roots.
Drug-facilitated crime
Drugs most commonly associated with DFCs cause sedation and amnesia, and are rapidly absorbed following administration. Victims may not report the crime for a considerable time after the event due to the amnesic properties of the drug. Analysis of blood and urine is unlikely to identify the drug adminstered if more than 72 hours has passed while hair provides an opportunity to identify the drug or drugs many months later.
Chronic administration of drugs for criminal gain has been reported 142,151 but by far the most common DFC reported is DFSA. This presents investigators with a considerable challenge as the identification of a single dose requires sensitive instrumentation to detect low pg/mg levels in hair. Gas chromatography-tandem mass spectrometry (GC-MS-MS) and liquid chromatography-tandem mass spectrometry (LC-MS-MS) provide the required senstivity.
The collection of hair samples is critical and dependent on the time that has elapsed since the incident. 143 A hair sample should be collected within 4–6 weeks of the incident and if positive a second sample collected one month later. The second sample acts as a control sample and, as such, the complainant must not have any cosmetic treatments or cut their hair between hair collections or until the investigations have concluded.
Gamma hydroxybutyrate (GHB) is an endogenous compound present in low concentrations in the body but is also widely available and associated with the club scene and in cases of DFSA. As GHB is endogenous, GHB may be detected in hair at low concentration and, as such, the presence of GHB alone is insufficient to confirm exogeneous administration. An elevation of the GHB concentration in the hair segment consistent with the approximate timeframe of the incident is required to differentiate between endogenous and exogenous GHB. Hair segments of between 3 and 5 mm have been proposed 142,143 but as yet there is no recognized concensus.
Child custody
Many children live in an environment where drug use is commonplace with the associated social welfare issues and are at an increased risk of accidental poisoning. Reports of deliberate administration of drugs to infants and young children by parents or carers are well documented and include fatalities. 151–155 Hair analysis provides a means of evaluating long-term drug exposure in infants and children; however, there are a number of potential sources of the exposure that must be considered.
Mothers who continue to misuse drugs and alcohol during pregnancy expose the unborn fetus as demonstrated by drug-positive meconium and hair collected from the newborn. 33,137,138 Drugs are also transfered in maternal milk to babies who are breastfed, and young children who are crawling or starting to walk can access drugs or drug residue on contaminated surfaces. Additional potential sources include smoke in the atmosphere and sweat from close contact with adults or siblings who use drugs.
When considering the potential sources of a positive hair sample from an infant or child, the contribution from gestational exposure must be explored. Many infants are born with hair of varying lengths and although many lose their hair in the months after birth, some will not lose their hair. If the child has not had their hair cut since birth and the mother was known to use drugs during her pregnancy, any drugs identified in the distal region of the hair must be attributed to exposure in the womb. Full details relating to the infant or child's hair and age should be requested and interpreted with caution. An average hair growth rate of 1 cm/month applies to adults but this cannot be assumed for infants and children whose growth rates will vary considerably.
Chronic excessive alcohol consumption
Chronic alcohol use is recognized as a significant worldwide health issue impacting across the spectrum of health, family and work life. The use of specific diagnostic biomarkers for chronic alcohol consumption, ethyl glucuronide (EtG) and fatty acid ethyl esters (FAEEs), has been investigated in various matrices including hair. 45,62,145 EtG and FAEEs are affected by cosmetic treatments; however, their incorporation into hair is not affected by hair pigmentation. 45,156
The SoHT published a consensus statement in 2009 recommending identification of either EtG or FAEEs in hair for assessing chronic alcohol consumption while encouraging identification of both. 5 Recommendations are given with respect to analytical instrumentation, GC or LC tandem MS for EtG and headspace solid-phase microextraction with GC-MS for FAEEs and the cut-offs to employ, as listed in Table 5.
Analysis of the section of hair closest to the scalp (0–3 cm) is recommended; however, the use of non-head hair has demonstrated good correlation with EtG concentrations in head hair. 157 Pubic hair concentrations were higher and as such should be interpreted with caution. Interpretation of alcohol biomarkers in hair should also consider the role of contamination from ethanol-containing cosmetics which have resulted in the identification of FAEEs in hair. 158
Over-interpretation
Drug testing in hair is an established complementary technique with a range of applications in both clinical and forensic toxicology. Evidence relating to hair testing has long been accepted in courts in the USA and Europe, 159,160 and increasingly so in the UK. 161,162 There remains, however, a number of factors that present issues relating to the interpretation of hair testing findings. This invariably leads to over-interpretation and a difference of opinion between experts that fundamentally questions the value of hair testing as forensic evidence. These issues include the incorporation of drugs and metabolites in hair, including the role of environmental contamination, hair colour bias and the effect of cosmetic treatments.
It is accepted that hair testing is an invaluable tool when assessing an individual's exposure to drugs, and although the exact mechanisms surrounding the incorporation of drugs into hair is not fully understood, it is accepted that a multimodel approach is most likely. Routes of incorporation from blood capillaries at the root bulb, sweat, sebum and external contamination may all contribute to the drug concentrations measured in hair and therefore must be considered when interpreting the findings. Washing hair samples will remove gross environmental contamination but may not remove all trace of drugs on the surface of the hair and therefore this must be considered as a potential source, or contributing source to the levels detected.
In 2009, the Federal Bureau of Investigation took steps to suspend cocaine analyses for all cases, with the exception of criminal investigations into the exposure of children to the drug. 163 They cited concerns relating to the role of environmental contamination leading to false-positives. The SoHT responded to these concerns by highlighting the importance of considering the likelihood of cocaine samples containing metabolites in sufficient quantity to produce a false-positive. 164
The determination of metabolites is important to help distinguish between passive contamination and ingestion. 2 The SoHT recommends evaluation of metabolite to parent drug ratios, but these must be considered in light of variation between laboratory methods. 2 The hydrolysis of labile drugs during method analysis must be taken into account when utilizing metabolite to parent drug ratios. 2,165
Melanin plays an important role in the incorporation of drugs in hair and many drugs and their metabolites will bind preferentially to heavily pigmented hair. This is, however, an oversimplification as the binding of a number of drugs and metabolites is not influenced by melanin content and some researchers do not believe that there is a substantial hair colour bias. 166
Cosmetic treatments, whether regular shampooing, dyeing or relaxing/bleaching, will have a deleterious effect on the drug concentrations in hair but to different extents. Drug concentrations in untreated hair decrease from the root to distal end of the hair, supporting the natural ‘wash-out’ of drugs from hair. Aggressive bleaching treatments reduce drug concentrations and therefore all declared hair treatments must be considered.
For all of the reasons given above and the variation in hair growth rates, drug concentrations in hair cannot be related to dose. It has been suggested that individual hair growth rates should be calculated by bleaching or colouring a hair strand directly above the scalp. The growth rate can then be estimated by examining the hair four weeks later. This approach may have some merit in relation to criminal investigations but not withstanding the considerable intersubject variability, it is not a practical approach for most cases.
Segmental analysis of hair cut close to the scalp can provide an approximate historical profile of drug exposure but it is not possible to specify exact dates. Consideration must also be given in relation to the possibility of a ‘false-positive’ hair test. Following cessation of cocaine use, researchers have demonstrated a delay of up to three months before hair samples are negative as a direct consequence of varying hair growth rates. 167,168
Increasing or decreasing concentrations along the length of hair may be a result of increasing or decreasing drug use but may also relate to a combination of the variables discussed above or simply variation in the analytical measurements.
Future developments
The drive for greater understanding of the mechanisms of drug incorporation continues to excite and motivate researchers in the fast evolving field of hair testing. Research and development has a direct influence on the path taken by practitioners with respect to services offered in drug and alcohol testing in hair. The introduction of hair testing methodologies utilizing advanced instrumentation has opened up the potential for the identification of diagnostic biomarkers, and expanded the application of hair from identifying chronic exposure to detecting a single acute exposure.
Improvements in extraction techniques and further developments in analytical methodologies will continue to advance understanding, while the introduction of guidelines and quality systems will underpin the acceptance of hair as a powerful investigative tool with both clinical and forensic applications.
The introduction of hair testing services requires careful consideration and investment. Implementation of accreditation requires significant financial and resource commitment, and laboratories planning to offer hair testing services should consider carefully the implications of purchasing specialist equipment and the time required to validate methods for analysis of drugs in hair.
DECLARATIONS
