Abstract
Introduction
Neutrophil gelatinase-associated lipocalin (NGAL) is emerging as a promising new biomarker for the early identification of acute kidney injury (AKI). We have determined a reference range in a large healthy population. In addition, as NGAL is a neutrophil-related protein, we investigated whether the presence of leukocyturia has the potential to significantly alter the specificity of NGAL in the diagnosis of AKI.
Methods
One hundred and seventy-four subjects (100 men, 74 women ranging from 19 to 88 y) were included in the reference population. Urinary NGAL was analysed on the Abbott ARCHITECT and results expressed in mass (μg/L) and also normalized to urinary creatinine (μg/mmol). Fifty-two leukocyturic urine samples were also analysed for NGAL.
Results
The 95th centile for NGAL was determined to be 107 μg/L (13 μg/mmol). There were significant gender-related differences for NGAL, with women having higher concentrations. There were significant age-related differences for NGAL between the 40–59 and 60–88 y age categories. There were significant age-related differences between the <40 and 60–88 y categories when NGAL was normalized to creatinine. In addition, we found significantly higher concentrations of NGAL in leukocyturia (P < 0.0001).
Conclusions
We have established a 95th centile cut-off for urinary NGAL in a reference population. We have demonstrated the important potential interference of leukocyturia in confounding the interpretation of NGAL in the diagnosis of AKI.
Introduction
Acute kidney injury (AKI) is a common condition in hospitalized patients and is associated with high morbidity and mortality. 1 Currently, diagnosis of AKI is made by a rise in serum creatinine concentration, which is a delayed and unreliable measure in the acute setting. 2,3 Muscle mass, gender, age and aggressive fluid resuscitation in critically ill patients, all affect serum creatinine, rendering it a suboptimal marker of AKI. A biomarker of AKI that is easily measured, unaffected by other biological variables and capable of both early detection and risk stratification would be highly desirable.
Using genomic and protein microarray technology, a series of molecules have been identified as potential markers for AKI, among them, neutrophil gelatinase-associated lipocalin (NGAL). NGAL is a 25 kDa protein which is generally expressed in low concentrations but is increased in the presence of epithelial damage. 4–6 Several studies have demonstrated that NGAL is significantly increased in patients with AKI with the rise in NGAL occurring 24–48 h before a rise in creatinine is observed. 7–9 NGAL can be measured in both urine and plasma. 10 NGAL requires further evaluation in different clinical settings but evidence so far suggests it to be a promising biomarker. 11–14
Abbott Diagnostics (Abbott Park, IL, USA) has developed an automated urinary NGAL assay for use on the ARCHITECT analyser. 15 One of the requirements for clinical use of any biomarker for AKI would be to determine cut-offs for the normal population and decision points for defining abnormality. We have determined the 95th centile value in a large, healthy, non-hospitalized population for NGAL. In addition, since NGAL is a neutrophil-related protein, we have studied whether the presence of leukocyturia has the potential to significantly affect the NGAL concentration and affect its specificity for AKI.
Methods
The reference population included 174 urine samples from healthy subjects attending a routine health check clinic from August 2010 to October 2010. The clinic targets the general population who have not been previously identified as at risk from common conditions. One hundred men and 74 women, with an age range from 19 to 88 y, were included. Random midstream urine samples were collected into sterile containers without preservative, on the morning of their clinic visit. Subjects with either a glomerular filtration rate (GFR) of <60 mL/min/1.73 m2 as determined by the Chronic Kidney Disease-Epidemiology Collaboration (CKD-EPI) equation, or white blood cells of >10 × 109 cells/L or an erythrocyte sedimentation rate of ≥30 mm/h, were excluded. Urine samples with an albumin-to-creatinine ratio (ACR) of ≥30 mg/mmol or leukocyturia of ≥1 × 107 cells/L were also excluded.
After routine analysis was completed, an aliquot of the redundant urine sample was collected. The samples were centrifuged and the clarified urine aliquoted and stored at −70°C until required for analysis. The maximum time from collection of sample to freezing of an aliquot was six hours.
Urinary NGAL was analysed on the Abbott ARCHITECT i1000sr system. The ARCHITECT urine NGAL assay utilizes a non-competitive, sandwich format with chemiluminescent signal detection. The assay is reported to have a measurement interval of 10–1500 μg/L. Seventy-two samples had concentrations <10 μg/L; these were dealt with statistically by assigning them a value of 5 μg/L. Claimed within- and between-series imprecision at 20 μg/L is 1.4% and 4.4%, respectively. Within- and between-series imprecision at 1200 μg/L is 1.4% and 2.2%, respectively. Prior to study commencement, assay performance was assessed following published guidelines and found to perform within the manufacturer's specification. 16
Urinary creatinine was analysed by the Jaffe rate reaction on a Beckman Coulter DxC800 system (Beckman Coulter Inc, Brea, CA, USA). Urinary NGAL results were expressed in mass (μg/L) and also as normalized to urinary creatinine (NCR) (μg/mmol).
In addition, as the presence of urinary neutophils can be a source of NGAL, we evaluated urine samples from patients with leukocyturia. Midstream urine was collected into a sterile container and analysed for leukocyturia within four hours of collection. Urinary white cell count was determined by microscopy, using the Fast-Read counting chamber (Immune System Ltd, Paignton, UK). All subjects providing infected urine had a CKD-EPI estimated GFR of ≥60 mL/min/1.73 m2 and leukocyturia of ≥1 × 107 white cells/L.
All statistical analysis was performed using GraphPad Prism software, Version 5.03 (GraphPad Software Inc, La Jolla, CA, USA). All data were initially assessed for normality by applying the D'Agostino and Pearson omnibus normality test. All data failed this test and were therefore analysed using non-parametric methods. The groups were compared using the Mann-Whitney U test and presented as median and interquartile range.
Results
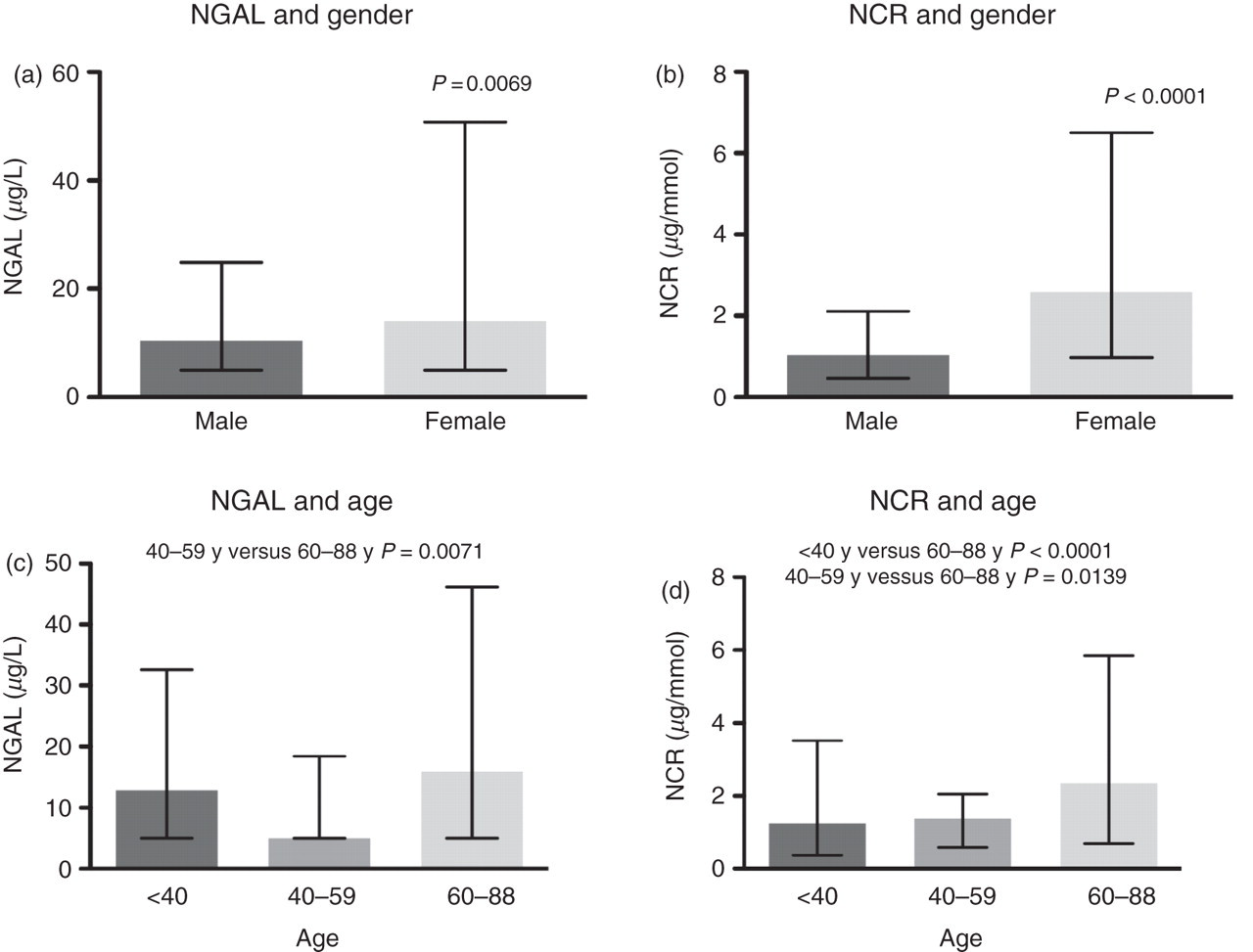
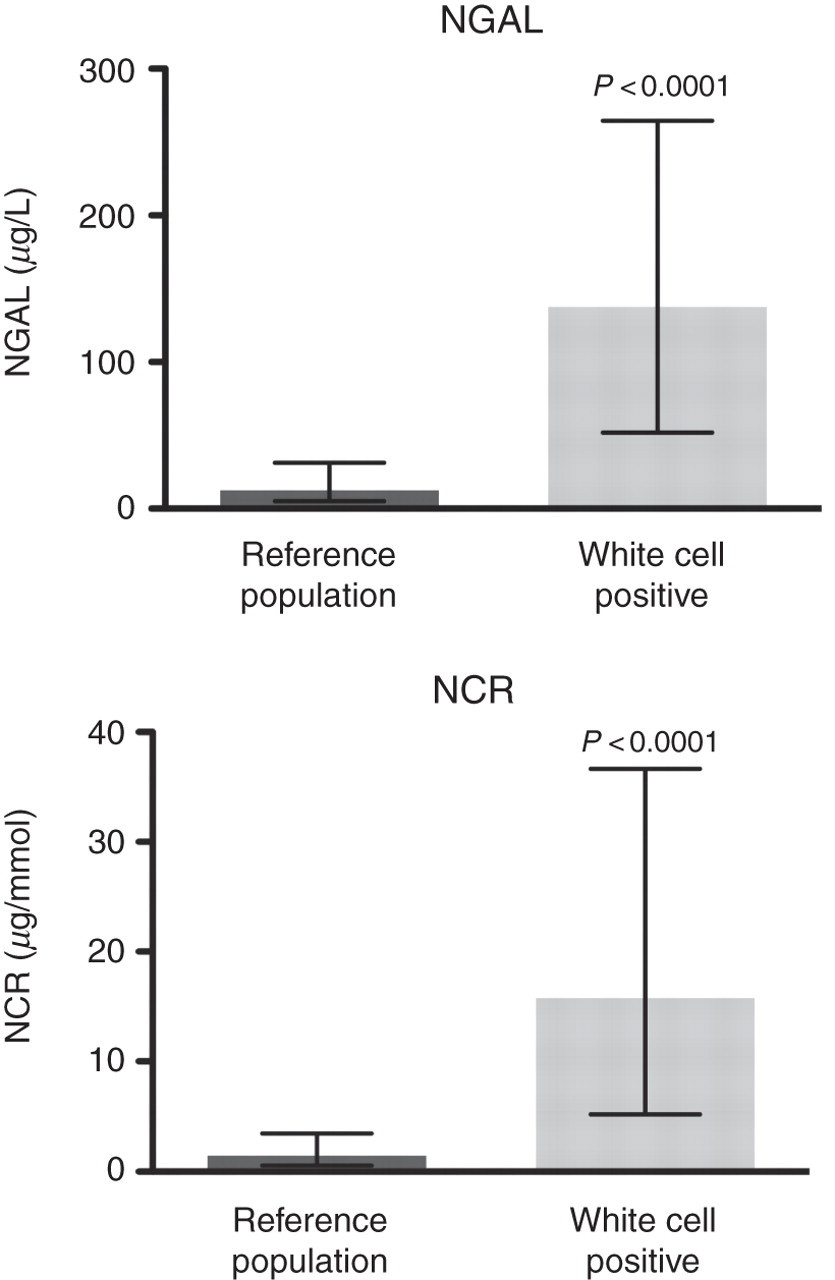
The dispersal of NGAL and NCR was found to display non-Gaussian distribution (P < 0.0001) (Figure 1). Women were found to have higher concentrations of NGAL (P = 0.0069) and NCR (P < 0.0001) in the reference population (Figure 2). There were significant age-related differences for NGAL between the 40–59 and 60–88 y categories when analysed as mass or when normalized to creatinine (P = 0.0071 and P = 0.0139, respectively). There were significant age-related differences between the <40 and 60–88 y categories only when NGAL was normalized to creatinine (P < 0.0001) (Figure 2). The 95th centile for NGAL and NCR in the total reference population, together with the gender- and age-related 95th centile results, are presented in Table 1. NGAL was also measured in an additional 52 urine samples containing ≥1 × 107 white cells/L. There were significantly higher concentrations of NGAL (P < 0.0001) and NCR (P < 0.0001) in white cell positive urine samples compared with the reference population (Figure 3). The white cell positive urine samples were then categorized further into urine samples that contained ≤1 × 108 white cells/L (n = 24) or >1 × 108 white cells/L (n = 28). Concentrations of NGAL and NCR were found to differ significantly between these categories, with higher concentrations of both NGAL (P = 0.0038) and NCR (P = 0.0050) in urine samples that contained >1 × 108 white cells/L.
Reference population NGAL and NCR frequency distribution. NGAL, neutrophil gelatinase-associated lipocalin; NCR, normalized to urinary creatinine Gender (a, b)- and age (c, d)-related differences for NGAL and NCR. NGAL, neutrophil gelatinase-associated lipocalin; NCR, normalized to urinary creatinine Differences in NGAL and NCR concentrations between the reference population and white cell positive urine samples. NGAL, neutrophil gelatinase-associated lipocalin; NCR, normalized to urinary creatinine NGAL and NCR 95th centile reference intervals NGAL, neutrophil gelatinase-associated lipocalin; NCR, normalized to urinary creatinine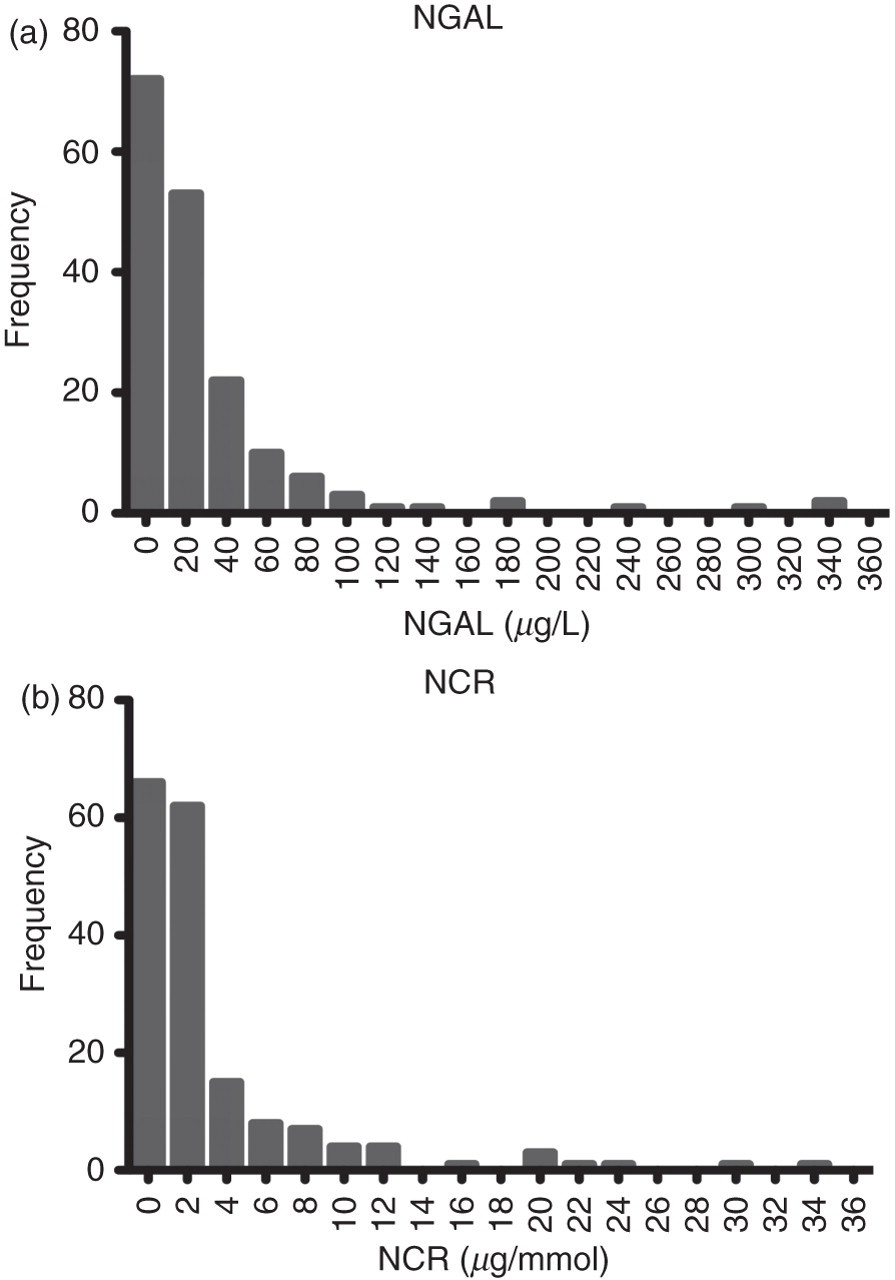
Discussion
This is the first independent study to report results for the 95th centile of NGAL in mass and normalized to creatinine in a sizeable population spanning all age groups (including those over 70 years). Our population represents a healthy cohort of subjects, rather than a population which is under clinical investigation. Several studies in which patients developed AKI have demonstrated the use of NGAL in the early diagnosis of AKI and proposed cut-offs for optimum utility of this test ranging from 100 to 270 μg/L. 17 Singer et al. 18 have recently reported an optimum cut-off of 104 μg/L, above which patients were likely to develop intrinsic renal failure. This is in close agreement with our proposed 95th centile of 107 µg/L. However, our 95th centile is slightly lower than the value reported by Abbott Diagnostics. In a population of 196 non-hospitalized donors, they found the 95th centile to be 132 μg/L. They did not investigate the influence of age or gender on NGAL expression. Our data suggest that NGAL concentrations vary with age and that decision-making cut-offs should be age-related. Our data suggest that when NGAL is expressed in mass or when normalized to creatinine, there are significant gender differences. Whether NGAL should be normalized to creatinine is still an area of debate. 19 There is no clear guidance as to whether it offers an improvement in the performance of NGAL in AKI. The argument against this is that normalization to creatinine is inaccurate as patients must be in a steady state and this is clearly not the case for patients with AKI. Therefore, normalization may be unnecessary and misleading in some clinical settings. On the other hand, acutely ill patients often have significant urine output, following aggressive fluid resuscitation, and thus baseline post-resuscitation NGAL results may be underestimated as a result of urinary dilution. The inclusion of both results in future clinical studies are required to determine the sensitivity and specificity of the 95th centile cut-offs and to determine the benefits of normalization to creatinine.
We have demonstrated the significant potential interference of leukocyturia in confounding the interpretation of NGAL in the diagnosis of AKI. NGAL is currently an expensive biomarker and therefore requires optimal clinical utility to justify the cost. Our data for normal subjects with leukocyturia demonstrate that the determination of urine white blood cells must form part of the protocol for evaluation and interpretation of urinary NGAL. Decavele et al. 20 propose a mathematical adjustment to improve the diagnostic efficiency of NGAL in the presence of white blood cell counts >100 × 109/L. Further studies are required to determine the significance of urosepsis on the specificity of NGAL and how it impacts on the clinical utility of the test.
The optimal value of NGAL is in the early identification of patients at risk of renal tubular damage and evolving intrinsic AKI. This study defines clear cut-offs for NGAL in a healthy population. Higher concentrations should alert clinicians to the need for close clinical monitoring of kidney function and facilitate timely initiation of renal-protective therapies in patients with rising NGAL.
DECLARATIONS
