Abstract
The objective of this study is to report a new manifestation of acute stroke following antifibrinolytic agent administration in young women carrying heterozygosity for methylene-tetrahydrofolate reductase (MTHFR) C677T. The study included two young women who developed an acute ischaemic stroke following three days of tranexamic acid administration for bleeding gynaecological disorders. Case 1, a 44-year-old woman, presented left hemiplegia, mild dysarthria and anosognosia. Brain magnetic resonance imaging showed right ischaemic fronto-temporal lesion due to subocclusion of the right middle cerebral artery. Case 2, a 49-year-old woman, developed aphasia and right hemiplegia. Neuroimaging showed left capsular and periventricular infarcts due to near occlusion of the left internal carotid artery. Thrombophilia screening, coagulation parameters, homocysteine testing, 12-lead electrocardiography, and transthoracic and transoesophageal echocardiography were unremarkable. Genetic assay showed that both patients carried heterozygosity for MTHFR C677T, in which cytosine (C) is replaced by thymidine (T) at base position 677. To our knowledge, this is the first report describing the association between genetic factors and the onset of stroke following antifibrinolytic drugs intake. These data suggest a synergic effect of plasminogen activator inhibitor and heterozygosity for MTHFR C677T on the pathogenetic mechanisms leading to ischaemic stroke in young people.
Introduction
Prothrombotic and proatherogenic polymorphisms have been suggested as genetic predisposers for cerebral ischaemia in young patients. 1 However, predisposing genetic background on stroke pathogenesis is poorly understood and continues to be rigorously investigated.
A common genetic factor is represented by the methylene-tetrahydrofolate reductase (MTHFR) 677T variant. The homozygous form MTHFR 677TT was found to increase the risk of arterial thrombosis in a meta-analysis 2 but not in a large study. 3 The heterozygosity for MTHFR C677T, also called thermolabile variant, with approximately half-normal activity of the enzyme, has been associated to increased migraine susceptibility. 4 So far, there has not yet been clear evidences that the thermolabile variant increases the risk of stroke.
Here, we report two cases of young women who developed acute ischaemic stroke following tranexamic acid treatment. Interestingly, both women showed heterozygosity for the MTHFR C677T polymorphism.
Case report
Case 1
A 44-year-old woman was admitted to our Stroke Unit because of the acute onset of left hemiplegia, dysarthria and anosognosia. Her family history was entirely negative for thromboembolic diseases. Her past clinical history was positive for migraine with aura but no hypertension, diabetes or dyslipidaemia were reported. Her recent clinical data reported bleeding due to curettage for voluntary abortion one week before, and the intake of tranexamic acid (500 mg oral/daily) from three days. No other pharmacological treatments were reported. At admission, neurological examination revealed moderate–severe hemiplegia with hypotonia in the left side, absence of tendon reflexes on the left side and hypoelicitability on the right side. Mild dysarthria and anosognosia were also observed. Brain magnetic resonance imaging (MRI) (T1, T2, fluid attenuated inversion recovery [FLAIR] and diffusion-weighted [DWI]) was performed showing right ischaemic fronto-temporal lesion exhibiting restricted diffusion on DWI (Figure 1a). Intra-and extracranial angio-MRI showed subocclusion of the right middle cerebral artery (Figure 1b). A complete routine chemical blood analysis including biochemical and coagulation parameters, cell count and thrombophilia screening was performed and resulted clinically irrelevant. Similarly, homocysteine (7.27 μmol/L, range: 5–15 μmol/L), folate (13.3 ng/mL, range: 7.2–15.4 ng/mL) and vitamin B12 levels (334 pg/mL, range 200–950 pg/mL) were in the reference range. Complete urine analysis including chemical (bilirubin, haemoglobin, glucose, ketones, leukocytes, nitrite and proteins, specific gravity, pH, urobilinogen), physical (colour, appearance) and microscopic examination did not show significant change from the reference range.
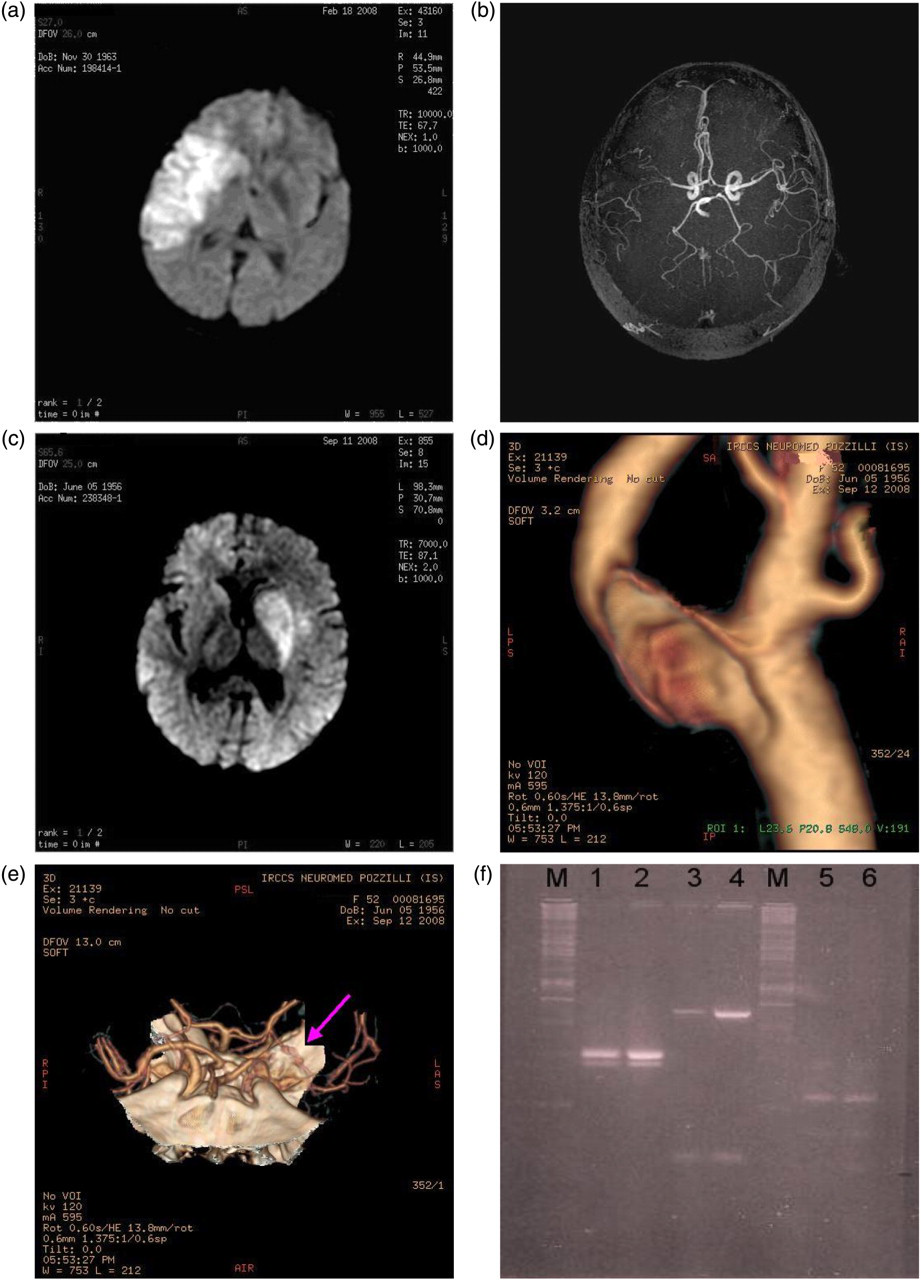
Neuroradiological findings and genetic analysis. (Case 1): (a) Right ischaemic fronto-temporal lesion in brain magnetic resonance imaging (MRI) (diffusion-weighted [DWI]), (b) subocclusion of the right middle cerebral artery (angio-MRI). (Case 2): (c) Left ischaemic capsular and periventricular lesion in brain MRI (DWI), (d) occlusion of the left middle cerebral artery and (e) arrow showing left subocclusive internal carotid stenosis (computed tomography angiography). (f) Patients showed heterozygosity for MTHFR C677T (lanes 1 and 2, respectively), wild type for Prothrombin G 20210 A variation (lanes 3 and 4) and Factor V Leiden (lanes 5 and 6) (gel electrophoresis of polymerase chain reaction–restriction fragment length polymorphism on 3% agarose). M, marker
Finally, electrocardiogram (ECG), and transthoracic and transoesophageal echocardiography were executed and were unremarkable. An additional peripheral blood sample drawn into EDTA tubes was obtained for genetic assays investigating the MTHFR C677T, factor V Leiden (G1691A) and prothrombin (20210A) polymorphisms. Genomic DNA was extracted from EDTA blood probes using the QIAamp DNA Blood Mini Kit (Qiagen, Milan, Italy). Genotyping was carried out by polymerase chain reaction–restriction fragment length polymorphism. Genetic assay revealed heterozygosity for MTHFR C677T, in which cytosine (C) is replaced by thymidine (T) at base position 677 (Figure 1f).
Case 2
A 49-year-old woman was referred to the Stroke Unit after the acute onset of aphasia and right hemiplegia. The patient's family history was negative for thromboembolic diseases. Her past clinical history was positive for migraine without aura, and no cardiovascular risk factors such as hypertension, diabetes or dyslipidaemia were recorded. Recent clinical data showed hospitalization for excessive uterine bleeding due to curettage for uterine fibromatosis eight days before, and the intake of tranexamic acid (500 mg oral/daily) from three days. No other pharmacological treatments were reported. At admission, neurological examination revealed moderate–severe hemiplegia in the right side, with hypotonus. Tendon reflexes were absent on the right side and were normally elicited on the left side. Severe aphasia was observed. Brain MRI (T1, T2, FLAIR and DWI) revealed left ischaemic capsular and periventricular lesion exhibiting restricted diffusion on DWI (Figure 1c). The extracranial ultrasound examination revealed a near occlusion of the left internal carotid artery confirmed by computed tomography angiography that showed also the occlusion of the left middle cerebral artery (Figures 1d and e). A complete blood and urinary analysis was performed (as reported in case 1). Blood analysis revealed an increase in leukocyte number, which was reduced in the following days and microcytic anaemia (Hb: 8.7 g/dL, range: 12–15.5 g/dL; mean cell volume: 73.5 fL, range: 80–94 fL). Other blood and urine parameters were clinically irrelevant. Homocysteine (5.69 μmol/L, range: 5–15 μmol/L), folate (8.90 ng/mL, range: 7.2–15.4 ng/mL) and vitamin B12 (331 pg/mL, range: 200–950 pg/mL) levels were in the reference range. Finally, ECG, and transthoracic and transoesophageal echocardiography were unremarkable. Interestingly, also in this case, genetic analysis revealed heterozygosity for MTHFR C677T (Figure 1f).
Discussion
Stroke is a complex multifactorial disorder resulting from an interaction between genetic background and environmental (non-heritable) factors. The incidence of stroke increases with age and is therefore low in young adults, defined as aged less than 40, 45, 50 or 55 years. 5
Common risk factors such as hypertension and diabetes are not very frequent in young adults and understanding of risk factors for ischaemic stroke evidence is based on case reports and some case-control studies.
Here, we report two cases of young adult women who developed acute stroke. Interestingly, both women carried heterozygosity for MTHFR C677T and had been taking tranexamic acid for gynaecological disorders.
A perusal analysis of our data revealed that both patients suffered only from migraine and did not show traditional vascular risk factors.
Some studies candidate migraine as a possible risk factor for stroke. On this issue, Linetsky et al. 6 showed in six patients that stroke occurred during the ictal phase of the migraine. In these patients, carotid or vertebral vasospasm with consequent hypoperfusion and infarct 6 was demonstrated by means of cerebral angiography.
In our described cases, the patients did not reveal migraine within the last month before the stroke event. 6 In contrast, both women had taken tranexamic acid three days before the onset of stroke.
Tranexamic acid is a plasminogen activator inhibitor that has been promoted as an effective and safe treatment in a variety of bleeding disorders, including menstrual blood loss in menorrhagia, and is usually prescribed to treat or prevent heavy menstrual bleeding. 7 Clinical studies performed in women with idiopathic menorrhagia showed that tranexamic acid for 4–7 days reduced menstrual blood loss by 34–59%. 8
From their experience of a single or a limited number of cases, several authors have expressed concern about an increased incidence of thrombotic and cerebral ischaemic complications associated with antifibrinolytic drugs. 9–12 Previous reports described the stroke onset following tranexamic acid administration. 13–16 In particular, Rydin and Lundberg 11 and Davies and Howell 12 reported intracranial arterial thrombosis in two women treated with tranexamic acid for menorrhagia. Agnelli et al. 16 described the case of a young women who died of left internal carotid artery thrombosis after taking tranexamic acid to prevent uterine bleeding. In both studies no genetic analysis was reported.
In our cases, we found that the ischaemic stroke following tranexamic acid is associated with heterozygosity for MTHFR C677T. Based on this evidence, we hypothesized that the tranexamic acid administration could be related to the stroke onset because of a permissive genetic background.
Experimental models and human studies have demonstrated that the gene MTHFR C677T is actively involved in maintaining the normal homeostatic properties of vascular endothelium, including endothelium-dependent regulation of vascular tone, inflammation and haemostasis. 17 In this regard, alterations of the haemostatic system may be more prominent in the presence of a plasminogen activator inhibitor, thus suggesting a synergistic effect on the pathophysiological mechanisms leading to cerebral ischaemia.
Our data increase the knowledge about the causes of stroke in young patients specifically related to drug intake. However, more evidences are needed to clarify the direct interaction between these factors. Future studies will need to be performed in animal models to define whether the heterozygosity for MTHFR C677T could be considered as a gene modifier of stroke, related to the use of antifibrinolytic agents.
DECLARATIONS
