Abstract
The transformation of the antineutrophil cytoplasmic antibody (ANCA) specificity in the absence of specific drug treatment has not been reported in the literature. A few studies have suggested changes in the epitopes recognized by the ANCAs. We describe two patients who switched from myeloperoxidase-positive to PR3 (proteinase 3)-positive ANCA during the course of their disease process and subsequently remained unchanged. One patient developed ulcerative colitis following the appearance of PR3-ANCA while the other remains quiescent. Regular follow-up and close monitoring of ANCA specificity are essential. A change of specificity may indicate the development of a new ANCA-related disease.
Introduction
Antineutrophil cytoplamsic antibody (ANCA) may contribute to the pathogenesis of vasculitis. 1 Change of antibody specificity is very unusual as only a few reports have described its induction as a consequence of specific drug treatment. We describe two patients with renal vasculitis, both of whom spontaneously switched from myeloperoxidase (MPO)-ANCA to proteinase 3 (PR3)-ANCA antibodies. They had no clinical features of active vasculitis at the time of apparent antibody switch. Several months later, one developed inflammatory bowel disease.
Case 1
A 54-year-old man had a six-week history of ankle oedema, cough, joint pains, night sweats and flu-like symptoms. There was no nasal symptom or haemoptysis. Past history included choroido-retinopathy and kidney stones. On examination, he was apyrexial and his blood pressure was 136/86 mmHg. He was also noted to have petechiae over the ankles. Initial investigations included haemoglobin 10.3 (reference range 13.0–18.0) g/dL, urea 23 (1.7–8.3) mmol/L, creatinine 302 (45–120) μmol/L and C-reactive protein (CRP) 64 (0–5) mg/L. Urinalysis revealed blood 3+, protein 3+ and cellular and granular casts. Immunofluorescence showed a P-ANCA pattern. MPO-ANCA was greater than 100 ELISA units (reference range 0–7 U/mL) and PR3-ANCA (reference range 0–7 U/mL) was 0.8 U/mL. Antiglomerular basement membrane and antinuclear antibodies were not detected. A chest X-ray was normal. A renal biopsy contained 23 glomeruli; two had cellular crescents. Immunohistochemistry was negative for immunoglobulins and complement components and electron microscopy did not show immune deposits. Treatment comprised of methylprednisolone, prednisolone and cyclophosphamide. He achieved clinical remission within 10 weeks when the MPO-ANCA became negative. He was commenced on azathioprine after three months and prednisolone was tapered and stopped after two years.
One year later, he developed low-titre PR3-ANCA while the MPO-ANCA remained undetectable. CRP at this moment in time was 1 mg/L. Fifteen months later, he developed rectal bleeding and diarrhoea. Investigations included haemoglobin 14 g/dL, serum creatinine 91 μmol/L, CRP 22 mg/L and PR3-ANCA was positive at 22 U/mL. Sigmoidoscopy revealed chronic active proctitis and colonic biopsies showed active chronic inflammation. Treatment comprised of prednisolone enemas, mesalazine and an increased dose of azathioprine. Symptoms resolved after a few weeks. Since then he has had no symptom of vasculitis or inflammatory bowel disease, although PR3-ANCA remains positive.
Case 2
A 64-year-old man complained of generalized arthralgia and cough. Three months later, he developed a purpuric rash with nail fold infarcts. His serum creatinine increased from 129 to 182 μmol/L. Three years prior to this presentation, he had anterior resection of the colon for adenocarcinoma. The recorded blood pressure was 135/50 mmHg. He had a colostomy and a solitary ulcer of the right lateral malleolus. Investigations included haemoglobin 10.6 g/dL, erythrocyte sedimentation rate 65 mm/h, urea 13.5 mmol/L, creatinine 210 μmol/L, CRP 146 mg/L and 24-h urine protein 0.69 g. Urinalysis revealed blood 4+ and protein 1+. Serum complement C3 was 1.6 g/L and C4 was 0.46 g/L. ANCA fluorescence showed a C-ANCA pattern; PR3-ANCA was borderline (7.0 U/mL) while MPO-ANCA was >100 U/mL. Antinuclear antibodies and glomerular basement membrane antibodies were not detected. A chest X-ray was normal and the ultrasound showed normal kidneys. A renal biopsy contained eight glomeruli; all showed cellular crescents and two contained segmental necrosis. Immunocytochemistry was negative and no dense deposits were observed by electron microscopy. He was treated with intravenous methylprednisolone followed by oral prednisolone, monthly intravenous cyclophosphamide and irbesartan. This regimen appeared to improve his symptoms as the serum creatinine decreased from 238 to 200 μmol/L, and the CRP became normal. The MPO-ANCA became negative and he was converted to azathioprine. Eighteen months after the presentation, PR3-ANCA became detectable. He continued prednisolone and azathioprine treatment. In October 2008, CRP increased to 9 mg/L and PR3-ANCA increased to 80 U/mL, although he remained well. Hence, the prednisolone dose was increased. Consequently, his CRP fell to 3 mg/L and PR3-ANCA decreased to 15 U/mL. In April 2010, joint pains recurred. At this time, PR3-ANCA had increased to 124 U/mL and CRP to 26 mg/L. He was treated with an increased dose of oral prednisolone and symptoms rapidly improved. Between 2005 and 2010, his blood pressure was consistently below 130/70 mmHg, but serum creatinine increased to 331 μmol/L. In 2010, he had a repeat kidney biopsy. This contained 24 glomeruli, 11 of which were totally sclerosed and the remainder showed mild increase of mesangium. There was moderate tubular atrophy and fibrosis. Immunocytochemistry was negative and there was no evidence of active disease.
Methods
Following standard protocol, 1,2 indirect immunofluorescence (IIF) was performed using commercial ethanol-fixed neutrophil slides (Binding Site, Birmingham, UK) as the substrate, with detection using polyclonal anti-human IgG conjugated to fluorochrome. Enzyme-linked immunosorbent assay (ELISA) was used for the detection of specific antibodies. Polystyrene wells precoated with either purified human MPO or PR3 proteins (Phadia Ltd, Uppsala, Sweden) for anti-MPO/PR3 ELISA, respectively, were used. Detection was by using enzyme-linked (β-galactosidase) anti-IgG mouse monoclonal antibody. The reference range for MPO- and PR3-ANCA ELISA assay is deemed to be 1–7 U/mL using Phadia Immunocap 250 instrumentation. We performed both IIF to observe the ANCA pattern and ELISA to confirm antigenic specificity on each occasion serum samples were analysed from the two patients.
Quality assurance and result reliability
One may question the accuracy and reliability of the analytical measurements taken, as it appears that both patients roughly switched during the same time period. Although this is correct, the switch did not occur in exactly the same month or the same batch. In addition, several measurements were taken over a period of few months that were consistent prior to the change in antigenic specificity. The same methodology and assay system has been in use throughout the study period, thereby eliminating possibility of inter-assay variation. Stored serum samples from initial presentation or from the time of switch were not available for a repeat analysis. Nevertheless, we have clear evidence in terms of internal (known positive and negative controls) and external quality assurance (National External Quality Assessment Scheme – NEQAS) data for both IIF and the ELISA to demonstrate that the results were accurate and reliable at the time and since the switchover occurred.
Furthermore, several competent readers for IIF have read and examined both patients’ fluorescence patterns during the past six years and if any discrepancies were observed; these would have been highlighted and amended accordingly. All our readers regularly participate in the regional quality assurance programme (NAEQAG – Northern Autoimmunity External Quality Assurance Group), which provides further evidence that the ‘switch’ phenomenon is genuine rather than an artefact or as a result of a technical error.
Immunofluorescence patterns
By using the IIF method, mainly two types of ANCA patterns (P-ANCA and C-ANCA) can be observed. Figure 1a shows internal quality control images (known positive P-ANCA and C-ANCA) and Figure 1b shows both patients’ most recent fluorescence patterns, i.e. after the switch.
(a) P-ANCA: fluorescence is observed in the perinuclear region of the polymorphic neutrophils. The bright green fluorescence appears as the nuclear outline due to the migration of the target antigen (myeloperoxidase) in the negatively charged outer nuclear zone. C-ANCA: fluorescence is observed in the cytoplasm of the polymorphic neutrophils as the target antigen (proteinase 3) is located within the cytoplasm. The nuclei appear as dark non-fluorescent regions. (b) Case 1 shows bright fluorescence of the perinuclear region consistent with the description of a P-ANCA pattern. There may be a degree of antinuclear antibody reactivity as the nuclei are slightly fluorescing. In contrast, case 2 shows fluorescence of the intracellular region (cytoplasm) and dark dense negative nuclei which is consistent with the description of a C-ANCA. *Images taken using a Nikon Eclipse E400 Fluorescent Microscope (Kawasaki, Japan). Overall magnification×400 (×40 objective lens,×10 eyepiece). Commercially available ethanol-fixed neutrophils slides were used. ANCA, antineutrophil cytoplasmic antibody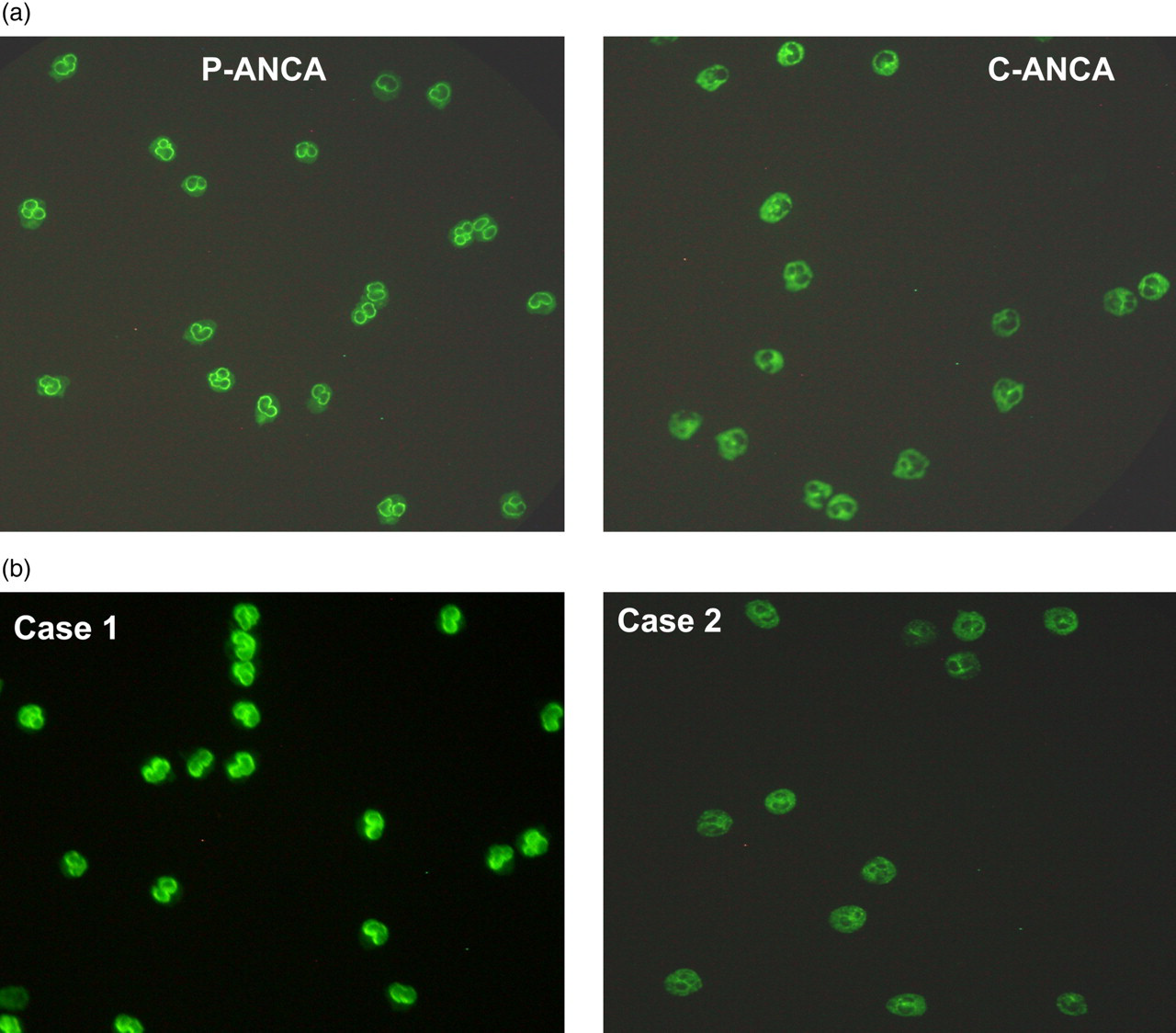
Discussion
Our patients spontaneously switched from having MPO-ANCA to PR3-ANCA antibodies (Figure 2). Previous reports of changing ANCA specificity were only associated with propylthiouracil or hydralazine. 3–5 Herein, we present an unusual phenomenon of the change of antigenic specificity without a change in the indirect immunofluorescent patterns. Furthermore, quality assurance data, both internal and external, provide evidence for it to be a genuine ‘change’ as opposed to an artefact.
ANCAs are usually IgG autoantibodies against intracellular constituents of neutrophils
1,2
and are found in small vessel vasculitides: granulomatosis with polyangiitis (Wegener's granulomatosis), Churg-Strauss and microscopic polyangiitis. Granulomatosis with polyangiitisis is usually associated with PR3-ANCA whereas microscopic polyangiitis and Churg-Strauss are associated with MPO-ANCA.
2
Rarely, patients can display a reversed pattern, as seen in our second patient (case 2) who had a classical C-ANCA fluorescence pattern but a high MPO-ANCA concentration at initial presentation.
2
Furthermore, dual MPO- and PR3-ANCA positivity has also been described in the literature, although very rare.
6–8
Our second patient could be considered a variant of this phenomenon at initial presentation; however, this changed to single-positivity within a month.
MPO- and PR3-ANCA antibody concentrations (U/mL) measured over a five-year period. (a) Case 1: MPO-ANCA increased from 50 to 100 U/mL prior to a rapid decline and subsequent rise in PR3-ANCA antibodies. (b) Case 2: MPO-ANCA was >100 U/mL which reduced gradually and has remained completely negative since the switch while PR3-ANCA has increased progressively. MPO, myeloperoxidase; ANCA, antineutrophil cytoplasmic antibody; PR3, proteinase 3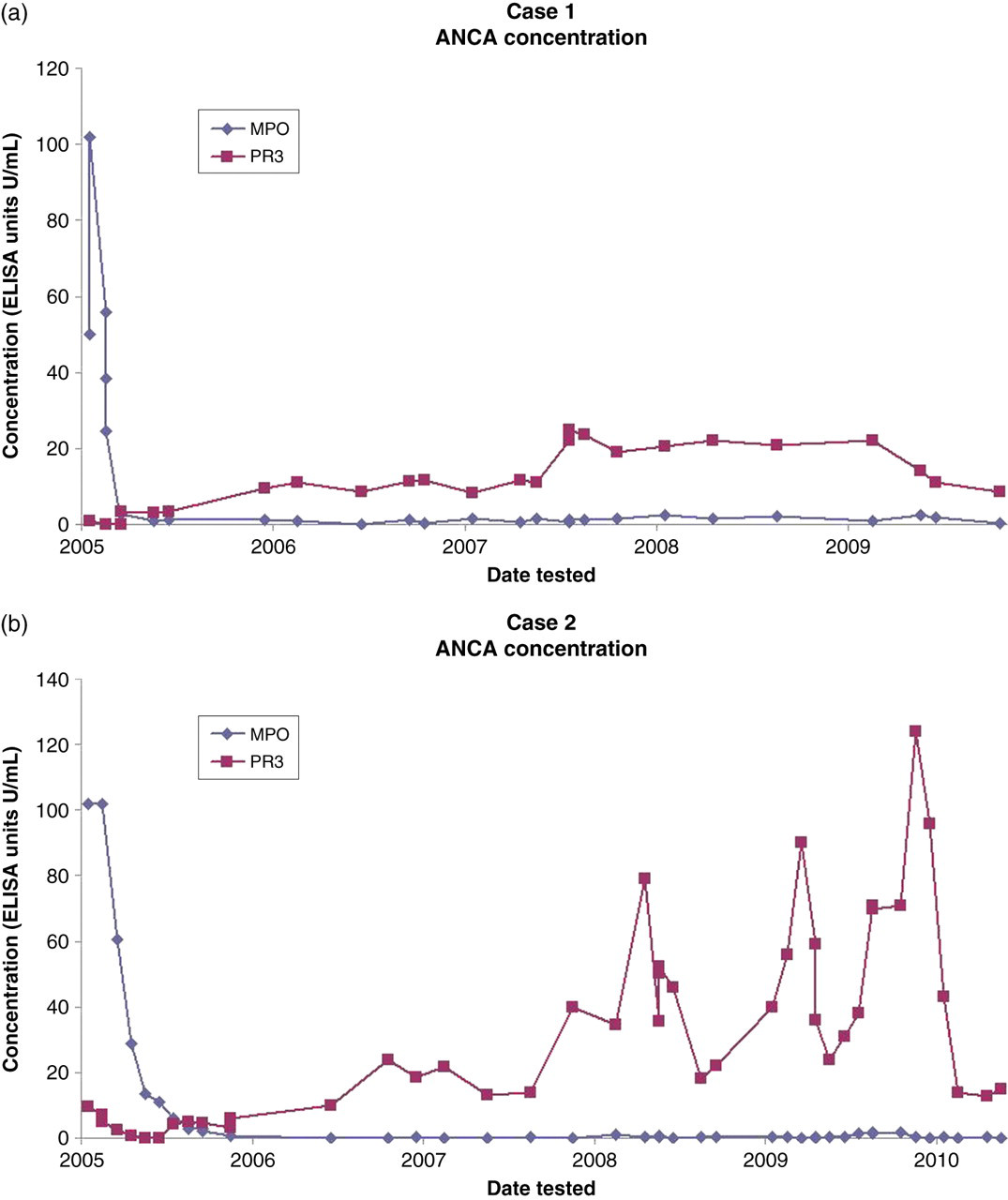
Occasionally, ANCA are found in non-vasculitic disorders including inflammatory bowel diseases, rheumatoid arthritis, malignancies and infections. 1,2 Inflammatory bowel conditions are associated with rare atypical P-ANCAs, against cathepsin G, elastase, and bacterial permeability increasing proteins. 9,10 The Case 1 photomicrograph (Figure 1b) perhaps illustrates an atypical P-ANCA, which could be due to the presence of atypical antigens. Recently, ulcerative colitis patients have been described to have PR3-ANCA. 11,12 Similarly, our first patient also developed PR3-ANCA and later ulcerative colitis.
The specificity of ANCA nearly always remains unchanged. Relapses are usually preceded by an increase and remission by reduction of ANCA titre. 13 A few studies have reported changes in the epitope recognition but not with the target antigen. 14 However, some drugs may induce changes in the antigenic target itself. 3–5 Choi et al. 15 reported a 25-year-old man with granulomatosis with polyangiitis who switched from PR3-ANCA to MPO-ANCA when treated with propylthiouracil and reverted back to PR3-ANCA upon stopping this treatment.
The mechanism of switch in our patients is unknown. Possibilities include exposure of ‘cryptic’ antigens or formation of ‘neoantigens’, to which the immune system was intolerant. Another possibility is that immunosuppression eliminated some auto-reactive B-cells while others proliferated, underwent affinity maturation and produced ANCA in a T-cell-dependent manner. 16 Patients with vasculitis have an increased number of ‘primed’ neutrophils expressing high levels of PR3 on their cell surface. 17 This may have driven B-cells towards a secondary auto-reactive phase and the development of other ANCAs. Hence, alteration of the surface expression of MPO or PR3 during the course of vasculitis may have changed antigenic specificity. Finally, one has to consider that two ANCA-associated conditions developed independently in the same individual.
We describe two patients whose ANCA specificity changed spontaneously from MPO-ANCA to PR3-ANCA. In one patient, this was associated with the onset of inflammatory bowel disease. Hence, a change in ANCA specificity should alert to the possible development of a new ANCA-associated condition.
Take-home message
Change of ANCA specificity can occur in patients with vasculitis and may indicate onset of a new ANCA-associated condition; Two distinct diseases processes can occur in the same individual, both of which are independently associated with ANCA antibodies; PR3-ANCA was detected prior to the development and diagnosis of clinically significant ulcerative colitis in one of our cases.
DECLARATIONS
