Abstract

It was five years ago that the idea of Pathology Harmony was conceived in Birmingham, UK. At that time it was realized that three major organizations undertaking clinical biochemical analyses in the city all used the same analytical platforms and reagents but offered different reference intervals to help interpret the results they produced. On initial inspection, it was not possible to discern any scientific explanation for these differences. While this variation was not unexpected and was longstanding, it was seen as an issue that needed to be addressed. Indeed, the specific area of reference range non-uniformity has recently been highlighted in the Journal. 1 While Pathology Harmony is a UK initiative, its basis, drivers and implementation will resonate in other countries.
External drivers to Pathology Harmony
There are several clear external drivers encouraging us to consider harmonization of various areas of the laboratory's output. Of particular note is the drive to link up laboratory results to enable them to be viewed by a range of users in primary and secondary health care. As we move towards full electronic reporting of pathology results, we appreciate more fully that variation in things such as test names, reference intervals and units of measurement associated with our results is something that hinders progress.
Unfounded variation between laboratories is now being questioned by those around us, and where it does not stand up to scrutiny, we need to address it as quickly as we can. In particular, users in primary care no longer accept unnecessary variation of our pathology service, especially where it interacts with their own work. This is not just related to reference intervals but to many other areas of our work. So, for example, if there are differing policies between laboratories on when to phone abnormal results out of hours to a primary care location this is increasingly likely to be questioned by the end user. Different organizations working across health care boundaries clearly need to have some joined up thinking. One role of Pathology Harmony has been to bring back together pathology laboratories that had moved apart as a consequence of more competitive interactions with each other. We also need to ensure that the issues around harmonization are fully understood and the processes we use to undertake review and make recommendations for harmonization are scientifically based. Only with this assurance will local implementation proceed.
Harmonization techniques
Pathology Harmony evolves through three phases of work, using techniques that enable logical and scientific approaches to the harmonization process. Firstly, we identify the variation. This includes sampling what actually happens in a number of laboratories to determine the extent of the differences. We then look for scientific reasons supporting the differences we have identified. Identifying a valid scientific basis to variation curtails any further consideration of harmonization. Creatinine, with very different analytical approaches still in use, is an example of a test that we have not been able to harmonize. Where the underlying science does not convincingly explain identified variation, the harmonization process moves forward (Figure 1).
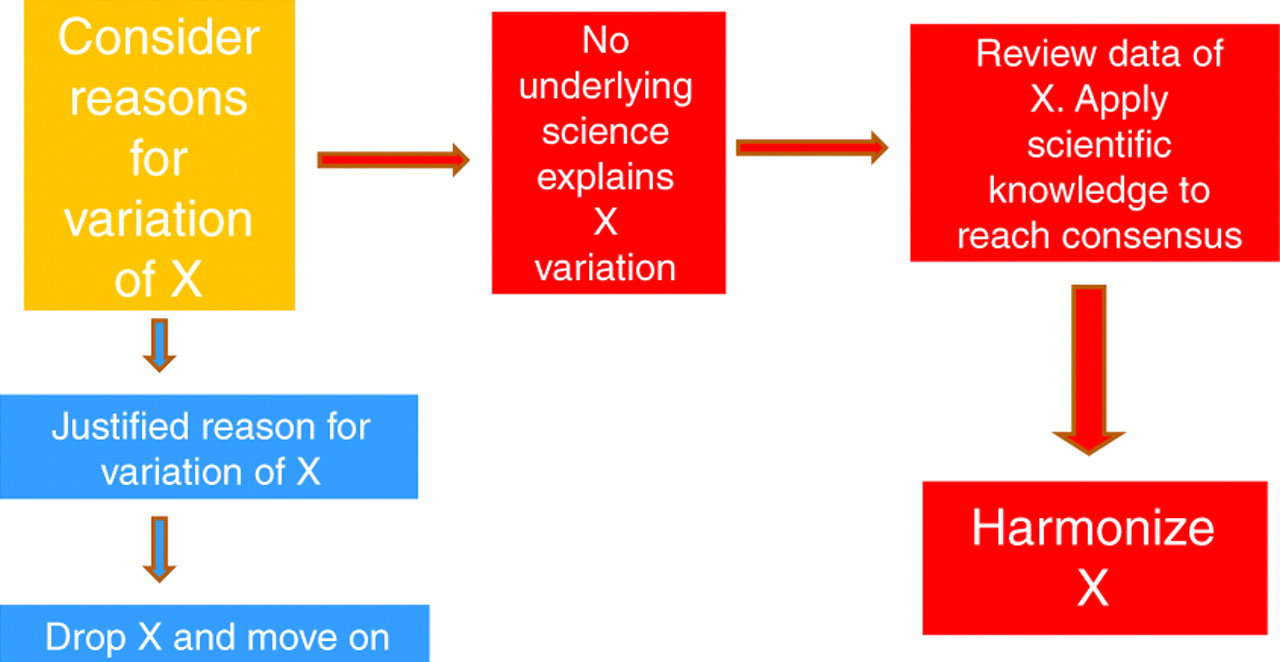
The process of Pathology Harmonization
Harmonization of reference intervals has taken up a lot of our time so far. When investigating how laboratories have ended up with their current reference intervals, it is clear that there has been very little standardization of technique. Current reference intervals can be based on historical ranges developed using very different analytical equipment many years ago, on ranges provided by the reagent manufacturer or on published reference intervals in standard textbooks, sometimes modified locally, based on a whole range of reasoning. Interestingly, in most situations the variation seen for reference intervals has, with the exception of an occasional outlier, generally been very small. For example, the data collected for serum potassium reference intervals for laboratories in the central region of England are shown in Figure 2. This, together with other evidence led to the Pathology Harmony recommendation of a serum potassium reference interval of 3.5–5.3 mmol/L. Recommendations for some other common biochemistry tests are listed in Table 1.
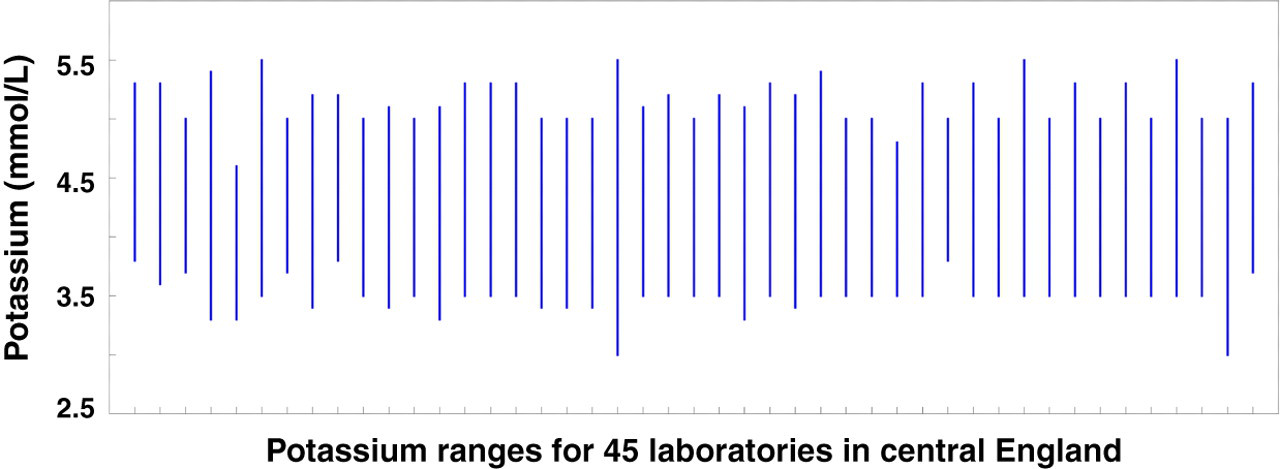
Variations in serum potassium reference intervals in 45 laboratories in central England
Examples of agreed Pathology Harmony clinical biochemistry reference intervals for adults
IFCC, International Federation of Clinical Chemistry and Laboratory Medicine; p-NPP, p-nitrophenyl phosphate; AMP, 2-amino-2-methyl-1-propanol
The Pathology Harmony approach has primarily centred on the study of test reference intervals, firstly in clinical biochemistry and more recently in haematology and immunology. The outputs have been widely circulated and commented on and this has included presentation of the recommendations at meetings with representatives from across the UK. The later stages of the Pathology Harmony process have had strong support from the Department of Health in England and also from key professional groups. This political dimension is absolutely vital for successful local implementation and has been a key part of the success of the Pathology Harmony approach. Professional groups in the UK (the Association for Clinical Biochemistry, the Institute of Biomedical Science and the Royal College of Pathologists) have now asked all laboratories to implement harmonized ranges with a deadline of June 2011.
Units of measurement
At present, there is an urgent need to help UK pathology harmonize the units used to report test results. Studies in haematology highlighted potential issues with different test units. Of particular concern was the reporting of haemoglobin concentration in both g/dL and g/L units, with the potential for clinical interpretation errors. This is a clear clinical risk that can be reduced by harmonization. The fact that the Pathology Harmony recommended unit for haemoglobin is g/L, when most laboratories currently report g/dL makes the point that we need to carry people with us for local implementation to proceed. Using the same basic technique of data collection and review, Pathology Harmony has now moved on to study units of measurement. The first 600 recommendations for harmonization of clinical biochemistry test units are now open for review on the Pathology Harmony website. 2
Harmonization of reference intervals, test names and units of measurement offers many practical advantages to help optimize current services. Harmonization can also be helpful when we generate new evidence about appropriate reference ranges from population studies, so that when we decide that a change is required it can be implemented from a basis of current consensus. The pathology harmonization process requires a mix of science, pragmatism and also political buy-in from key professional groups. The success so far has been due to an understanding of the importance of each of these elements to help achieve our fundamental aim of continual service improvement in the changing world around us.
DECLARATIONS
