Abstract
Although generally robust, immunoassays remain vulnerable to occasional analytical errors that may have serious implications for patient care. Sporadic errors that occur as a result of properties of the specimen are particularly difficult to detect. They may be due to the presence of cross-reacting substances, antianalyte antibodies or antireagent antibodies, all of which may lead to erroneously high or low results. Low results may be observed for tumour markers due to high-dose hooking in the presence of very high analyte concentrations. Erroneous results can occur unexpectedly with any specimen and there is no practical means of identifying specimens likely to cause problems in immunoassays. The possibility of interference should always be considered when results do not appear to be in accord with the clinical picture. Errors can occur in even the best-managed laboratories and their early investigation is always desirable. If there is any doubt whatsoever about a result, clinical staff should be encouraged to contact the laboratory. Investigations for possible interference that can be undertaken in most laboratories include testing for linearity on dilution, recovery experiments, treatment with heterophilic blocking tubes and confirmation using a different method. It may be desirable to consult specialist laboratories if more complex studies are necessary. Informing clinical and laboratory staff of the ever-present possibility of unexpected interference, ensuring brief clinical details are available to laboratory staff, and above all facilitating excellent communication between laboratory and clinical staff are key to minimizing the risk of clinical mismanagement due to unsuspected interference.
Introduction
Immunoassay remains the method of choice in the clinical laboratory for analysis of many analytes, particularly complex heterogeneous molecules. It is not surprising, since immunoassays involve the reaction of complex biological reagents (usually antibodies) with other complex biological reagents (the analyte) in a variable biological matrix (often serum or plasma), that they are inherently vulnerable to different types of interference. 1–6 However, the frequency with which such interference occurs – and most importantly, the proportion of consequently erroneous results that may significantly and adversely affect clinical management – is rather difficult to assess. 7,8 Serious clinical errors are most likely to occur when decisions about patient management are directly linked to laboratory results, alternative means of corroborating those results are not readily available and they are not part of a panel, which is often the case for tumour markers (e.g. human chorionic gonadotropin [hCG], prostate specific antigen [PSA], CA125, thyroglobulin and calcitonin), 9–11 troponin 12 and possibly thyroid stimulating hormone (TSH). 3 Awareness by laboratorians of the types of interferences most likely to be encountered for particular assays and analytes, together with excellent and proactive communication with clinical staff, is essential to increase the likelihood of such errors being detected in time to prevent unwarranted or inappropriate clinical treatment. Whether the risks of waiting for confirmation of a result outweigh the risks of failing to take immediate action is a decision for the clinician, always remembering that having the wrong result may be worse than having no result.
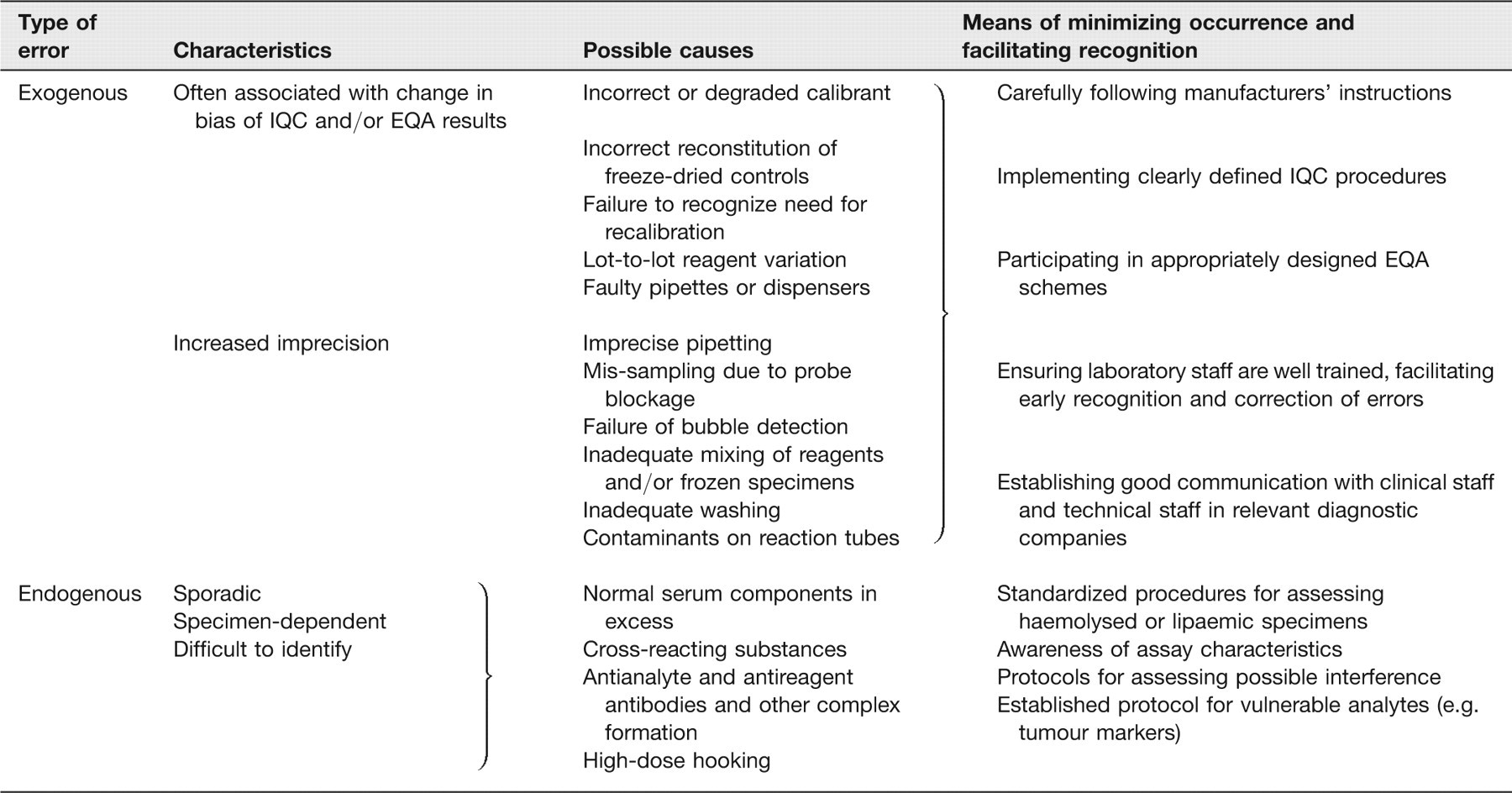
Exogenous errors, which are not associated with properties of the individual specimen and which may reflect system failure (e.g. a blocked probe on an automated analyser), differ from errors due to endogenous interferences, which are usually specimen-dependent and are much more difficult to detect. Causes of both types of error are briefly reviewed here but the main focus of this practically oriented article is on how to investigate suspected endogenous interferences and minimize the risk of them adversely affecting clinical management (Box 1).
Substances such as immunoglobulins, other proteins, lipids and bilirubin present in some blood serum samples can interfere in some immunoassays to give falsely high or low results. Immunoassays can be designed by careful selection of reagents, addition of blocking agents and reaction kinetics to minimize, but probably not eliminate, the effects of such interferents. Falsely high or falsely low results due to interferences endogenous to the specimen present a particular risk to patient care because they (a) are not detectable by normal laboratory quality control procedures, (b) are reproducible within the test system, (c) are often clinically plausible and (d) are relatively rare. The diagnostics industry has done much to design assays that are robust to endogenous interferents and laboratories should include test robustness in their selection criteria for assay platforms. As with all investigative procedures, both laboratory and clinical staff should maintain a high index of suspicion in inspecting test results, especially where major clinical interventions are based on test results alone. Good laboratory-clinical liaison will assist this. Procedures (e.g. use of blocking reagents) are available for the laboratory to check whether a suspicious result might be due to endogenous interferents.Minimizing risk of interference – key points
Although not considered here, it is of course also essential for laboratory and clinical staff to be well-informed about how laboratory test results may be influenced by other factors including pregnancy, severe illness or medication. Examples include the variable effect of heparin, phenytoin, frusemide, carbamazepine or salicylate on free thyroid hormone results,
13,14
finasteride and other 5-
Frequency and type of errors occurring in clinical laboratories
Fewer errors now occur in the analytical phase of analysis than in the pre- and postanalytical phases, reflecting major advances in automation and sample handling. 16,17 With total error rates reportedly in the range 0.012–0.6% for laboratory tests, 16 the frequency of errors in the analytical phase, expressed as a percentage of those occurring in all phases of analysis, has been estimated as between 7% and 13%. 17 These analytical errors include those due to equipment malfunction and undetected failure in quality control as well as those classified as exogenous or endogenous. 17 Erroneous results of either type may be inappropriately high or low.
Errors due to exogenous factors
Exogenous errors may be random (e.g. due to variability in sample pipetting or other manipulations) or systematic (e.g. consistent deviations from the ‘true’ value due to calibration errors), may affect many results, and should usually be detected by good internal quality control 18 and occasionally retrospectively by careful examination of external quality assessment (EQA) results 1 (Table 1). It has recently been suggested that study of the outlier rate is a useful parameter with which to assess the robustness of assays. 19 The effectiveness of such procedures is likely to have improved with increasing focus on continuous quality improvement in clinical laboratories, as exemplified by the Clinical Laboratory Improvement Act in the United States and stringent requirements for laboratory accreditation in the United Kingdom.
Comparison of exogenous and endogenous types of errors
IQC, internal quality control; EQA, external quality assessment
Errors due to endogenous interferences
In contrast, endogenous interferences are almost always sporadic and specimen-dependent, and hence more difficult to identify. Moderate interference is generally more difficult to detect than gross interference, where results are more likely to arouse clinical suspicion. If unrecognized, interference may cause errors in interpretation of results due to the analytical specificity of the method used (e.g. unintended detection of cross-reacting substances closely resembling the analyte being measured), due to the presence of abnormally high concentrations of normal serum constituents (e.g. haemolysed or lipaemic specimens), or due to formation of antibody or other macromolecular complexes. Whatever the cause, the identification of endogenous interferences – many of which are associated with measuring substances other than those intended – always depends on the vigilance of well-informed clinical and laboratory staff (Table 1).
Cross-reacting substances
An understanding of what an individual immunoassay measures and of its vulnerability to clinically relevant cross-reacting substances is essential, in order to ensure that the assay used is appropriate for the clinical application as well as to minimize the risk of misinterpretation of results. The specificity of any immunoassay is primarily determined by the specificity of the antibodies used and the assay format. 1 It is important to remember that cross-reaction may be either positive or negative as illustrated in Figure 1. Although cross-reactivity is often quoted as a percentage of the assay result of the interferent as compared with the signal obtained by an identical concentration of analyte, the concentrations of both analyte and interferent likely to be encountered are often very different. In order to provide a more clinically useful means of assessing the likely degree of interference that would be encountered in routine practice, it has been suggested that cross-reactivity should be calculated at the concentrations of the cross-reactant likely to be encountered in health and disease and then expressed as the apparent percentage change in the measured endogenous analyte concentration. 20
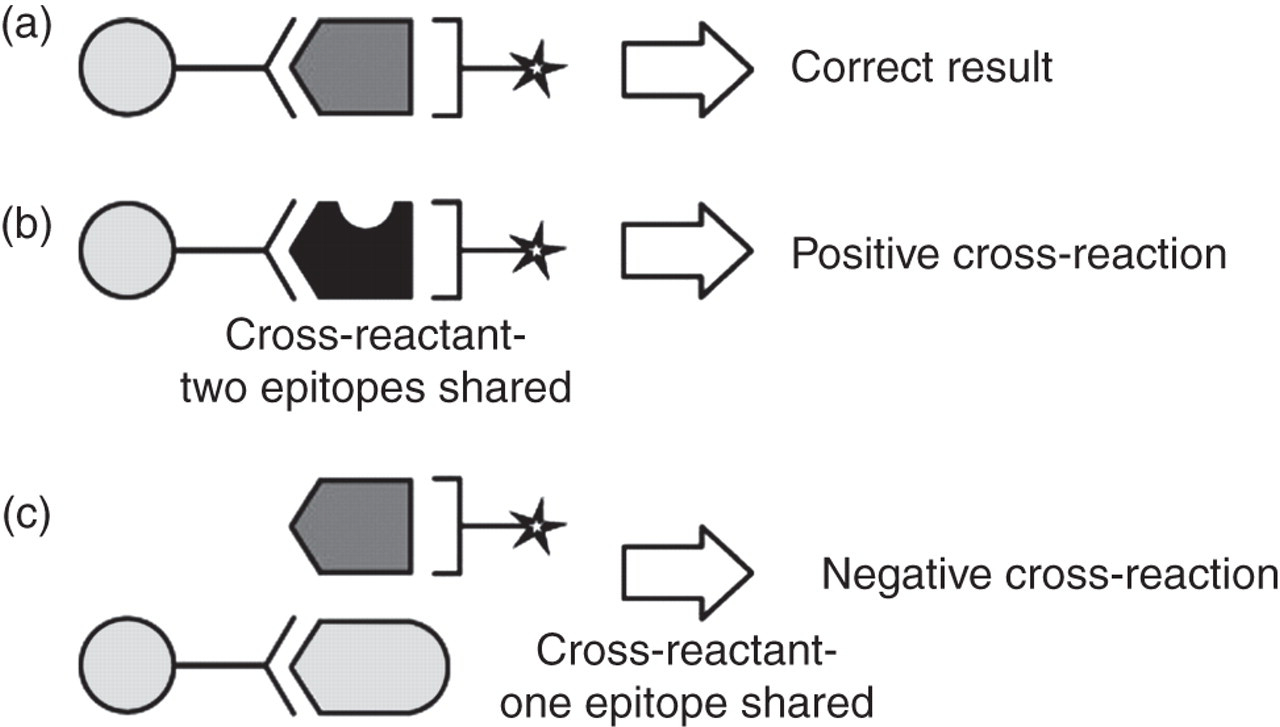
Cross-reactions in two-site immunoassay. (a) Antibodies bind to specific analyte – correct result obtained; (b) cross-reactant sharing two epitopes in common with analyte – positive cross-reaction; (c) cross-reactant sharing only one epitope in common with analyte – negative cross-reaction. (Reprinted with permission from Seth J, Sturgeon CM. Pitfalls in immunoassays in endocrinology. Endocrinology and Metabolism In-Service Training and Continuing Education. 1993;
For many analytes what it is most clinically relevant to measure is still not well-established, although concerted international efforts are being made to address this for analytes as diverse as insulin, parathyroid hormone (PTH), thyroid hormones, troponin and vitamin D.
21
In a prototype International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) project, six International Reference Reagents for clinically important forms of hCG were prepared to enable better characterization of what current hCG immunoassays measure.
22
Availability of these Reference Reagents, together with complementary antibody mapping studies,
23
should facilitate development of clinically more relevant assays in the future.
22
For use as a tumour marker, for example, immunoassays should recognize both intact hCG and its free beta-subunit (hCG
A clearer understanding of what forms of an analyte are present in physiological specimens will be much facilitated by the increasing availability of well-validated mass spectrometric methods. Already in use in many routine laboratories for measurement of relatively simple molecules such as steroids, drugs of abuse and therapeutic drugs, technological advances mean that mass spectrometry is increasingly being applied to study complex heterogeneous molecules. A recent report describes the identification of new clinical variants of PTH and their measurement, together with that of previously identified forms of PTH, using quantitative mass spectrometric immunoassays. 24
While availability of highly specific assays is undoubtedly necessary to improve understanding of the effect of disease processes on circulating analyte concentrations, in clinical practice, use of assays with broader specificity is sometimes advantageous. Table 2 provides some examples illustrating the importance of sound understanding of the specificity of the immunoassay used. For some analytes, observed cross-reactions may be clinically beneficial (e.g. cross-reaction of hCG in an assay for luteinising hormone [LH] may be helpful in identifying unsuspected pregnancy in a woman with amenorrhoea 1 or of recombinant insulin analogues in assays for insulin 25 ) provided the characteristics of the immunoassay are appreciated by the user, while for other analytes they highlight a need for immunoassays with improved specificity (e.g. digoxin, growth hormone [GH]) or better standardization (e.g. PSA, PTH). Encouragingly, the latter is being proactively addressed for PSA under the auspices of the NHS Cancer Screening Programme. 26 Comparability of PTH results should be similarly improved by calibration of PTH immunoassays in terms of the recently established International Standard for PTH(1-84), IS 95/646. 27
Examples of analytes for which the analytical specificity of the immunoassay method used is likely to affect clinical interpretation
Lipids, haemoglobin and other serum constituents
There is considerable literature relating to the effect of excess concentrations of normal serum constituents on immunoassays. 20 This has been comprehensively collated by several authors 20,28,29 and is also presented in data sheets for most commercially available tests. Manufacturers’ advice should always be followed, and documented procedures for treating specimens identified as haemolysed or lipaemic should be available in all laboratories. Turbidity in a lipaemic sample can usually be visually detected and, if required, triglycerides removed from the sample either by precipitation or by ultracentrifugation before repeating the analysis on the clarified sample. Haemolysis is also readily detectable provided specimens are visually examined before analysis. Automated detection of haemolysis, icterus and lipaemia is possible: the advantages of this approach, including a helpful algorithm, have been reported for 28 analytes including some proteins. 30 Immunoassay results for some analytes (e.g. adrenocorticotrophic hormone, gastrin, insulin and PTH) are particularly likely to be affected adversely by haemolysis 20 and a repeat specimen should always be requested. The possible effect of additives in specimen collection tubes should also be considered. 28
Antibody- and antibody-like interferences
Since immunoassays rely on interactions between the analyte being measured and reagent antibodies to produce the assay signal, it is not surprising that they are particularly susceptible to interference from other unrelated antibodies that may be present in the reaction mixture. It is important to recognize that interfering antibodies may be present only transiently in a patient's serum, and that their characteristics and reactivity may vary, such that no immunoassay can be considered to be completely robust to all possible interference. Such interference, which again may be positive or negative, is more likely to occur in two-site ‘sandwich’ assay formats, in which interfering antibodies may either interact with the analyte to prevent binding or may form a bridge between the signal and capture antibodies in the absence of analyte (Figure 2). In a large study of 11,261 serum samples, carcinoembryonic antigen (CEA) was measured using a two-site, two-step immunometric assay using mouse monoclonal antibodies. 7 The frequency of interference in these specimens was found to be 4%, a proportion that could be reduced to 0.10% by removing the Fc fragments from the capture antibody. 7 This was reduced even further by adding heat-treated, non-specific murine immunoglobulin to the assay buffer. Some manufacturers of commercial immunoassays do their best to minimize the risk of such interference, usually by adding non-immune animal serum samples or other blocking agents to the reaction buffer. While appreciating that no measure can be relied upon to be effective in removing interference in every specimen – reflecting the biological variability described above – more could certainly be done by some manufacturers to reduce the risk of interference.
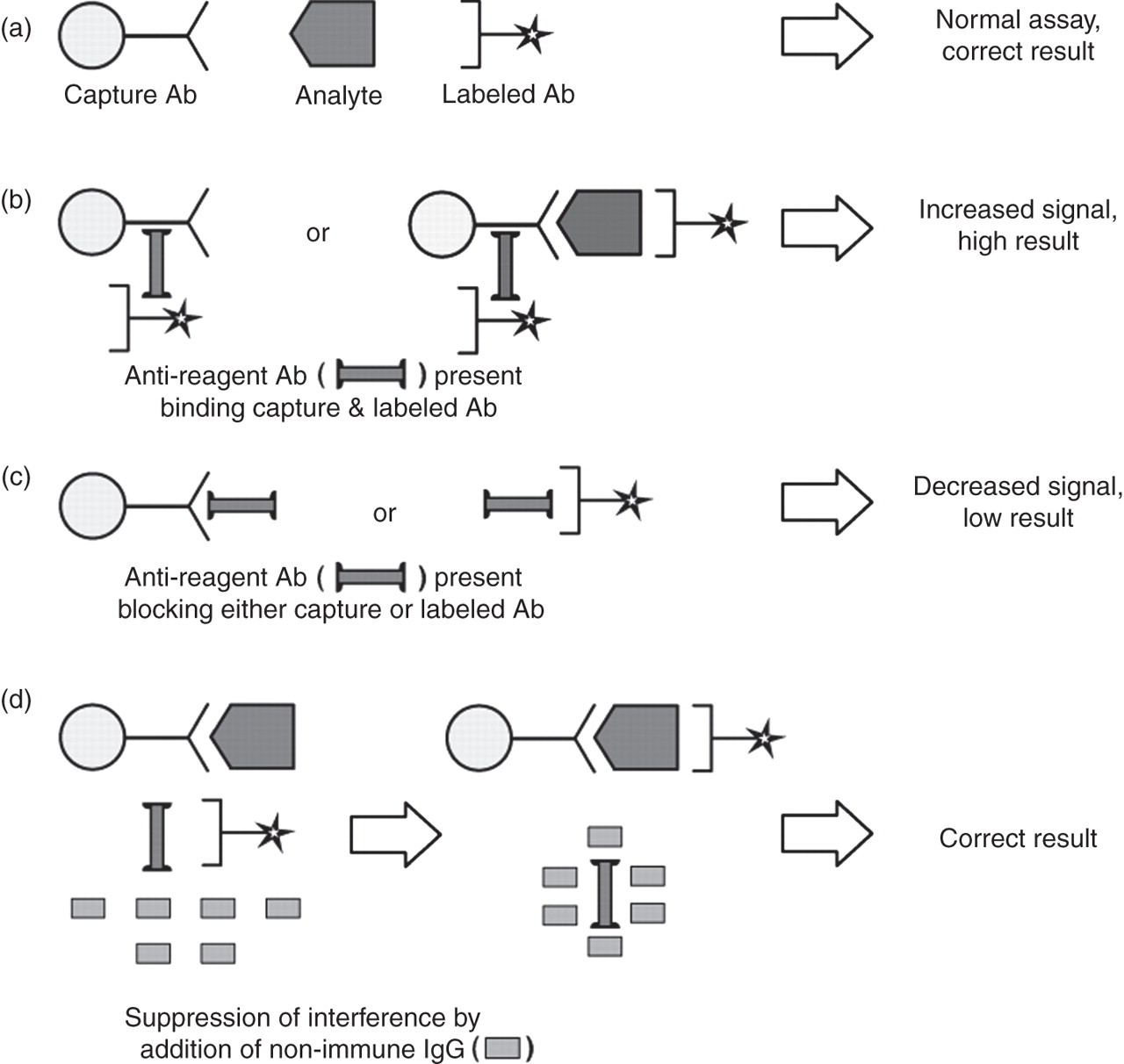
Interference from antireagent antibodies in two-site immunoassays. (a) Interfering antibodies absent – correct result obtained; (b) cross-linking of capture and labelled antibody in the presence of anti-reagent antibody – incorrectly high result (analyte may or may not be bound as well, depending on steric factors, but in either case the result will be falsely high); (c) interfering antibodies binding to either capture or labelled antibody only, reducing sandwich formation – incorrectly low result obtained; (d) effect of adding non-immune animal IgG to reduce interference – correct result obtained. (Reprinted with permission from Seth J, Sturgeon CM. Pitfalls in immunoassays in endocrinology. Endocrinology and Metabolism In-Service Training and Continuing Education. 1993;
The majority of published reports relate to interference from antireagent antibodies 3,8,31 although other complex formations can occur, as described below.
Interference due to antireagent antibodies
Some patient serum samples contain antibodies capable of binding to animal immunoglobulins such as those used in immunoassays, although estimates of the frequency of such antibodies vary widely (<1% to 80% is suggested in one review, 31 0.4–4% in another 2 ), reflecting differences in detection methods and populations studied. 31 However, the mechanism of interference and its severity depend both on assay design and on the nature of the interfering antibody (Table 3). In general, interferences caused by human anti-mouse antibody (HAMA) are likely to be more pronounced than those due to heterophilic antibody, while antibodies in serum samples from patients with rheumatoid disease may not always be of sufficiently high affinity to cause the bridging observed with HAMA or heterophilic antibodies (Figure 2).
Interference due to antianalyte antibodies and other complex formation
Patients with autoimmune disease
Patients with autoimmune disease are likely to have circulating antibodies that may disturb the binding of analytes to reagent antibody. Such antibodies may sometimes be predictable – e.g. circulating insulin antibodies in insulin-treated patients with diabetes – but may also be unexpected and can cause major difficulties. These are perhaps most likely to be observed for thyroid function tests, at least in part because of the frequency of testing. Unusual combinations of results such as concomitantly raised or suppressed TSH and free hormone concentrations or a significantly high or low free hormone result accompanied by inappropriately ‘normal’ or ‘unresponsive’ TSH should raise suspicion of interference. It is essential to consider other explanations not associated with interference. These may include (most frequently) poor compliance or (more rarely) clinical conditions relating to hypothalamic–pituitary pathology (e.g. TSH-secreting tumour), hereditary binding protein abnormalities (e.g. familial dysalbuminaemic hyperthyroxinaemia [FDH]) and end organ resistance to either TSH or free hormone. 13
Free hormone assays that rely on competition of a thyroxine analogue with unbound thyroxine in the sample can give spuriously high results, as albumin binding of the analog is enhanced by the FDH mutation. 32 This is less likely to cause problems in two-step procedures incorporating a wash step, but thyroid hormone results can still be misleading. In patients with suspected FDH, molecular genetic testing is comparatively simple and provides an unambiguous diagnosis. 33 Similarly, although their prevalence is difficult to assess, the presence of antibodies to thyroxine or tri-iodothyronine may affect some thyroid hormone methods, giving high or low results, while apparently not altering results in others. 34 Antibodies to insulin may cause very high insulin results. 35
Subjects with macroprolactinaemia and other macrocomplexes
In some patients’ serum samples, high molecular mass complexes of prolactin, termed macroprolactin, form a substantial component of the total immunoreactive prolactin measured by immunoassays.
36
Macroprolactins may be present in the serum of patients with significantly elevated monomeric prolactin. They have minimal bioactivity
Recent reports suggest that in some patients, similar complexes may be observed for other analytes including cardiac troponin (after acute myocardial infarction), 12,38 follicle stimulating hormone, 39 TSH 33,40 and vitamin B12, 41 but further study is required.
Subjects with differentiated thyroid carcinoma
In patients with treated differentiated thyroid carcinoma, undetectable serum thyroglobulin concentration following TSH stimulation is considered the most reliable marker of cure. 11,42 However, the presence of thyroglobulin antibodies or heterophilic antibodies, respectively, may lead to false decreases and increases in measured concentration, as has recently been described in detail. 11,42,43 Thyroglobulin antibodies are often present in patients with autoimmune thyroid disease, with 10% of healthy individuals having measurable levels. 5 Quantification of thyroglobulin by tandem mass spectrometry may ultimately circumvent the problem of antibody interference. 44 In the meantime, effective multidisciplinary collaboration is essential to minimize the risk of error, as has recently been comprehensively described. 43
Identifying results requiring investigation for possible interference
While considerable information is now available about the nature of possible endogenous interferences that may be encountered in clinical specimens, identifying such interference still relies almost entirely on a high index of clinical suspicion, i.e. the perception that a result does not fit the clinical picture. The random and sporadic nature of most interferences means that unless a cohort of patients at particular risk can be identified (as is the case for subjects with confirmed hyperprolactinaemia whose serum may contain macroprolactin 45 ), screening for possible interference is unlikely to be either feasible or cost-effective.
Interference should always be suspected if results do not correlate with the clinical picture. This is facilitated by a good understanding of the limitations of immunoassays in general as well as of the specific analytical characteristics of the method used. Laboratory staff are well placed to contribute to this process, which is aided considerably by provision of brief clinical details on request forms. Clinical colleagues should always be actively encouraged to contact the laboratory if they have any doubt about a result. When clinical staff are the first to suspect that results may be incorrect, early dialogue is essential so that relevant specimens can be retrieved before they are discarded and appropriate investigation instituted.
Investigating suspected antibody or antibody-like interference
The extent of investigations undertaken once interference is suspected is likely to be influenced to some extent by how much time and serum is available, but the most critical consideration should be the possible implications for patient management. Establishing whether interference is the cause of a clinically unexpected increase in a tumour marker such as hCG – where erroneous results can and have led to unnecessary chemotherapy or other treatment and also have ongoing implications for serial monitoring – is much more critical than demonstrating whether an apparently high LH result in a woman with perimenopausal symptoms is due to antibody interference. Failure to identify heterophilic antibody interference in serum from a patient with a favourable prognosis germ cell tumour, for example, recently resulted in him receiving several courses of toxic and unnecessary second- and third-line chemotherapy. 46
Even when resources are limited, once interference is suspected, there are several simple steps that should always be taken before the need for more specialized investigations is considered. Possible steps are shown in the flow chart in Figure 3 and discussed below.
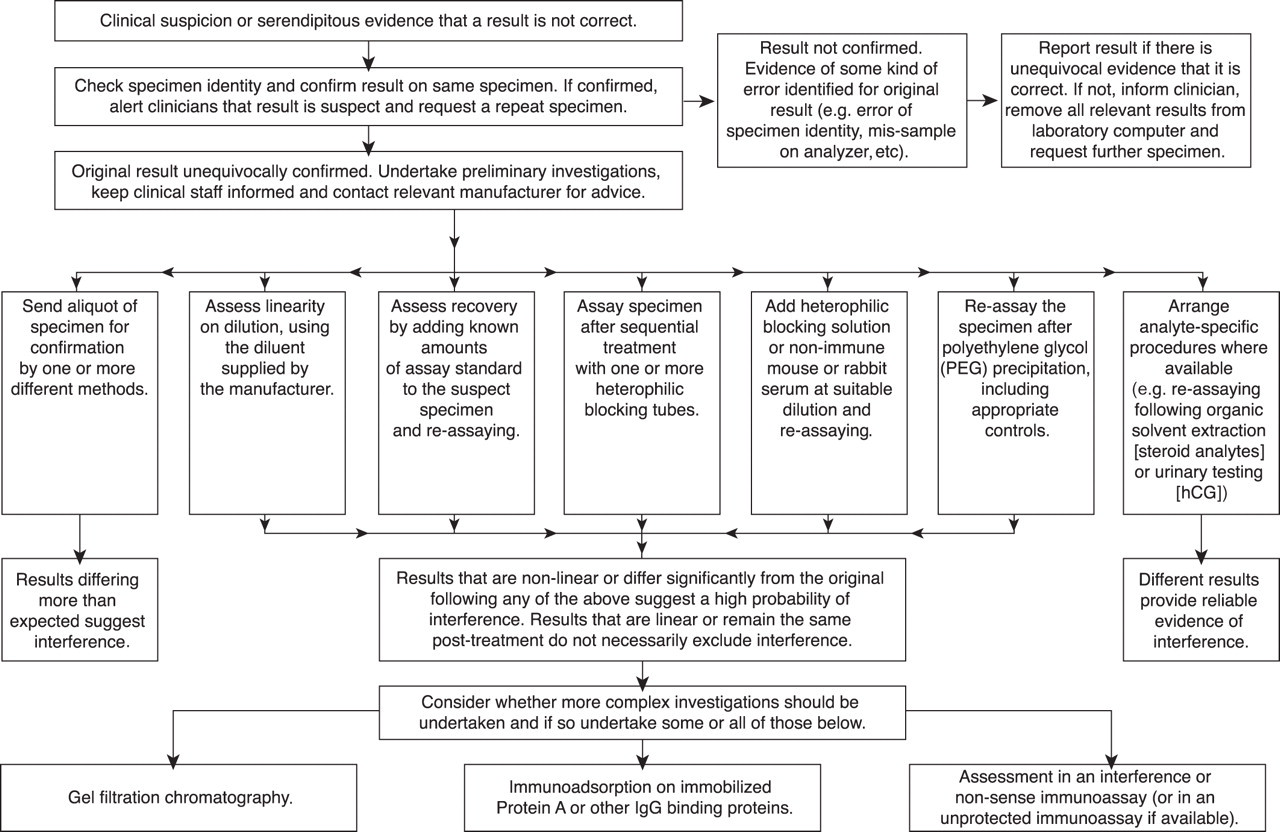
Flow chart showing sequence of investigations that might be undertaken to investigated suspected immunoassay interference
Initial procedures as soon as interference is suspected
As soon as an erroneous result is suspected, the specimen identity should be confirmed before repeating the assay to exclude the possibility of an analytical error. Where samples are subaliquoted (even by automated procedures), it is highly desirable to carry out the repeat assay on serum from the original correctly labelled primary tube. If the result is confirmed on repeat analysis, relevant clinicians should be alerted by telephone, all relevant results relating to the specimen removed from, and the reason flagged in, the laboratory computer with an explanatory note, and a further specimen urgently requested. Depending on the timescale in which this specimen can be acquired, primary investigations for possible interference should be performed on the first or second specimen.
Preliminary investigations for suspected interference
While unfortunately there are no tests that unequivocally exclude the possibility of interference, several relatively simple investigations may confirm it (Figure 3).
Confirmation of results in other immunoassay methods
It is usually desirable for the investigating laboratory to send an aliquot of the specimen to other laboratories for confirmation of the result by one or more different methods. Ideally, at least one of these would depend on an alternative methodology (e.g. radioimmunoassay), but in practice this is usually not feasible, except for hCG and thyroglobulin (see Appendix). Reference methods (e.g. equilibrium dialysis for free hormones) are clearly desirable, but are well-established for relatively few analytes. If, after taking account of method-related differences in bias as reflected in EQA and other available data, results differ significantly, this provides convincing evidence of interference although it will not necessarily identify which result is ‘correct’.
Dilution and recovery studies
Serial dilution and recovery studies can be informative, as lack of linearity of results on dilution of the specimen (in the diluent provided by the manufacturer) or low recovery of known amounts of analyte (e.g. the assay standard) added to the serum being investigated are suggestive of interference. Determining the limits of acceptability for such studies is difficult, however, and apparently satisfactory linearity and/or recovery may be observed even in the presence of an interfering substance. Conversely, some specimens that are apparently free from interfering substances do not dilute out linearly, particularly in some of the more complex tumour marker assays (e.g. CA19-920), making interpretation of results even more difficult.
Treatment with heterophilic blocking reagents
Many laboratories keep a supply of commercially available heterophilic antibody blocking tubes, and treating the specimen with these according to the manufacturers’ instructions is also desirable. The tubes are coated with a heterophilic blocking reagent (HBR) directed against human heterophilic antibody, which is claimed to block interference by steric hindrance. 47 Results that differ from the original result following treatment with these tubes suggest interference, but obtaining the same result does not necessarily exclude it.
HBR is also commercially available in solution and is recommended for use at a concentration of 400
Addition of non-immune animal serum
Adding non-specific immunoglobulins to the reaction mixture may reduce interference if the human antibodies bind to these instead of binding the assay antibodies. 7 Murine and bovine antibodies reduce interference in the highest percentage of patient's samples and also have the highest avidity for heterophilic antibodies. Ingestion of bovine IgG in milk has been suggested to be one of the factors inducing heterophilic antibodies in some subjects, which might explain the effectiveness of bovine IgG as a blocking agent. 7 Polymerized IgG preparations have been shown to be superior to native IgG. 49 Trial and error is required in determining the optimal concentration to be added with appropriate controls included.
Polyethylene glycol precipitation
At neutral pH, immunoglobulins are not particularly soluble and it is relatively easy to precipitate them. 20 When investigating possible antibody interference in immunoassays, reasonably well-controlled precipitation of potentially interfering antibodies and other high molecular weight complexes in serum can be conveniently achieved using polyethylene glycol (PEG), a polymer of ethylene oxide. In principle, PEG precipitation can potentially be applied to any immunoassay in which macro-complex formation is suspected of causing interference, 20,50 but the majority of reports to date describe its use in investigating the presence of macroprolactin (and large molecular weight forms of prolactin) in serum samples with raised total prolactin concentrations. 36 A recommended protocol for PEG precipitation 45 is provided in the Appendix.
While PEG precipitation can be helpful, it is nevertheless a relatively non-specific technique and a proportion of the free analyte is likely to be co-precipitated with any complexes present. 50 The extent to which this occurs has been shown to be both analyte- and method-dependent, 51 and may also be influenced by the characteristics of the individual specimen. It has been shown, for example, that the amount of monomeric prolactin co-precipitated with serum globulins can be increased in the presence of increased globulin concentrations, giving the erroneous impression that macroprolactin is present. 52 It has recently been reported that PEG precipitates almost all IgG and IgM but only 50% of IgA, which could also lead to false-negative results. 37 Confirmation by gel filtration chromatography (see below) may be desirable for such specimens. 52 In addition, PEG can interfere in some immunoassays. 50 It is therefore essential that the PEG procedure used is validated for both analyte and method and that the results are interpreted with caution. It is now also recommended that the absolute post-PEG concentration of prolactin should be reported to the clinician together with an appropriately derived reference interval. 45,53 A similarly rigorous approach is likely to be desirable if using PEG precipitation to investigate interference for other analytes.
Confirmation of serum results by testing in urine (hCG only)
If heterophilic antibody interference is suspected for a serum hCG result, obtaining a urine specimen for qualitative or quantitative analysis can be helpful. 54 As antibodies are not filtered into urine, a negative urine test result confirms interference in the serum immunoassay, provided the serum hCG concentration is ≥50 U/L. 54 Misleading results are most often seen with values <1000 U/L. (It is of course essential to ensure that the urine tested is from the same patient as the serum.)
Sample extraction
When investigating suspected interference in steroid immunoassays, particularly for testosterone, extracting the specimen with diethyl ether, re-suspending in appropriate analyte-free diluent and re-assaying may be desirable. 55 The extraction step should separate the steroid from any binding proteins as well as remove water-soluble steroid conjugates. Cortisol measurements in saliva can also be useful to detect binding protein problems and/or to confirm serum results. 56
Confirmation by mass spectrometry
Mass spectrometry potentially provides an additional means of investigating possible interference for those analytes for which methods are sufficiently well-validated and not reliant on antibody immunoadsorption (e.g. testosterone 57 and 25-hydroxy metabolites of vitamins D2 and D3 58 ). Such techniques have also successfully been applied to assays of plasma steroids in newborn infants where significant interference may occur due to steroids such as dehydroepiandrosterone sulphate and pregnenolone sulphate secreted by the fetal adrenal zone. 59 It is, however, essential to be aware of the possible influence of matrix effects and to appreciate that lower results obtained by mass spectrometry cannot be assumed to be due to increased specificity. 60
More complex investigations to characterize suspected interference
Once preliminary investigations have confirmed probable interference, further studies may be desirable for some specimens (Figure 3). Several relevant procedures are described briefly here. Depending on local arrangements, it may be most efficient where possible to refer such specimens for further investigation coordinated by a specialist referral laboratory or EQA centre (see Appendix).
Gel filtration chromatography
In the context of assay interference, the use of size exclusion gel filtration chromatography to investigate interference has probably been most widely used to confirm the presence of macroprolactin. 45 Considered the gold standard procedure for macroprolactin, the method can in principle be readily set up in any laboratory. However, it is rather slow, labour-intensive and costly and is not well-suited to routine use. 45
Three peaks of immunoreactive prolactin may be obtained on gel chromatography of normal serum samples on Superdex™ SD-75 (GE Healthcare Life Sciences, Buckinghamshire, UK): macroprolactin elutes first, followed by ‘big’ prolactin and finally monomeric prolactin. 45 In serum samples containing macroprolactin, the first (highest molecular weight) peak is significantly more prominent than in normal serum samples. The procedure described for macroprolactin (see Appendix) can be readily adapted for other analytes, if necessary selecting a gel with different size exclusion properties.
Immunoadsorption chromatography on immobilized IgG binding proteins
Protein A, Protein G, Protein A/G and Protein L are native and recombinant proteins of microbial origin that bind to IgGs from a number of species including human, mouse, rabbit, goat and bovine. 61 Commercially available coupled to cross-linked beaded agarose and other resins, they provide an attractive means of investigating potential interference from IgG antibodies, particularly as elution of bound antibodies can be achieved at physiological pH. Protein A and Protein G vary in the strength with which they bind to different IgG antibodies, both between species and between different antibody subclasses from the same species, so using both (Protein A/G) is attractive. Neither Protein A nor Protein G reacts with other classes of immunoglobulin (IgA, IgM, etc.) so they are not helpful in identifying interfering antibodies of these classes.
Protein A/G is a gene fusion product that contains four Fc binding domains from Protein A and two from Protein G. 61 As it binds to all human IgG subclasses, it can be particularly helpful in purifying IgG antibodies where the subclass identity is not known. Chromatography on immobilized Protein A/G has recently been used successfully as a pretreatment procedure to eliminate non-specific assay interferences in individual serum samples from rheumatoid arthritis patients 62 (see Appendix for sample protocol).
Assessment in an unprotected immunoassay
Commercially available immunoassays are protected against potential interference by addition of blocking agents. Consequently, access to pairs of immunoassays, one of which is protected and one of which is not, is now limited. However, paired immunoassays are still available in some specialist laboratories. 7 Different results in such assays provide convincing evidence of interference, but as with most other approaches, obtaining the same result in both methods does not necessarily exclude interference.
Assessment in an ‘interference’ or ‘non-sense’ assay
An early ‘interference’ assay for the detection of multivalent antibody-binding substances in serum used the same antibody as both capture and signal antibody and was used to investigate interference in a two-site immunoradiometric assay for hCG.
63
The method relies on the principle that a signal will only be obtained in the presence of a substance that could bind two or more molecules of the one antibody, which would not be the case for an analyte with only one antibody-binding site. The same principle can be refined using a combination of two unrelated assays, such as solid phase hCG and tracer anti-
Double radial immunodiffusion (Ouchterlony) studies
Referral to a specialist immunology laboratory for investigation using these techniques to identify anti-animal immunoreactivity in patient serum may be helpful.
High-dose hook or pro-zone effect
Two-site immunometric assays, particularly those in which signal and capture antibody are added simultaneously, are vulnerable to the high-dose hook effect at very high analyte concentration. 64,65 Free analyte and bound analyte compete for the limited number of solid-phase antibody-binding sites, yielding inappropriately low results which may even hook back to within the reference interval (Figure 4). The risk of reporting an incorrectly low result is greatest for analytes with wide physiological concentration ranges, e.g. AFP, CA125, CEA, hCG, PSA, prolactin and thyroglobulin. Although manufacturers usually specify a concentration below which hooking is unlikely to occur in a particular immunoassay, the only means of completely eliminating this risk is dilution of all specimens. This is likely to be both costly and over-cautious, particularly in the absence of good data as to the frequency with which such high concentrations may occur.
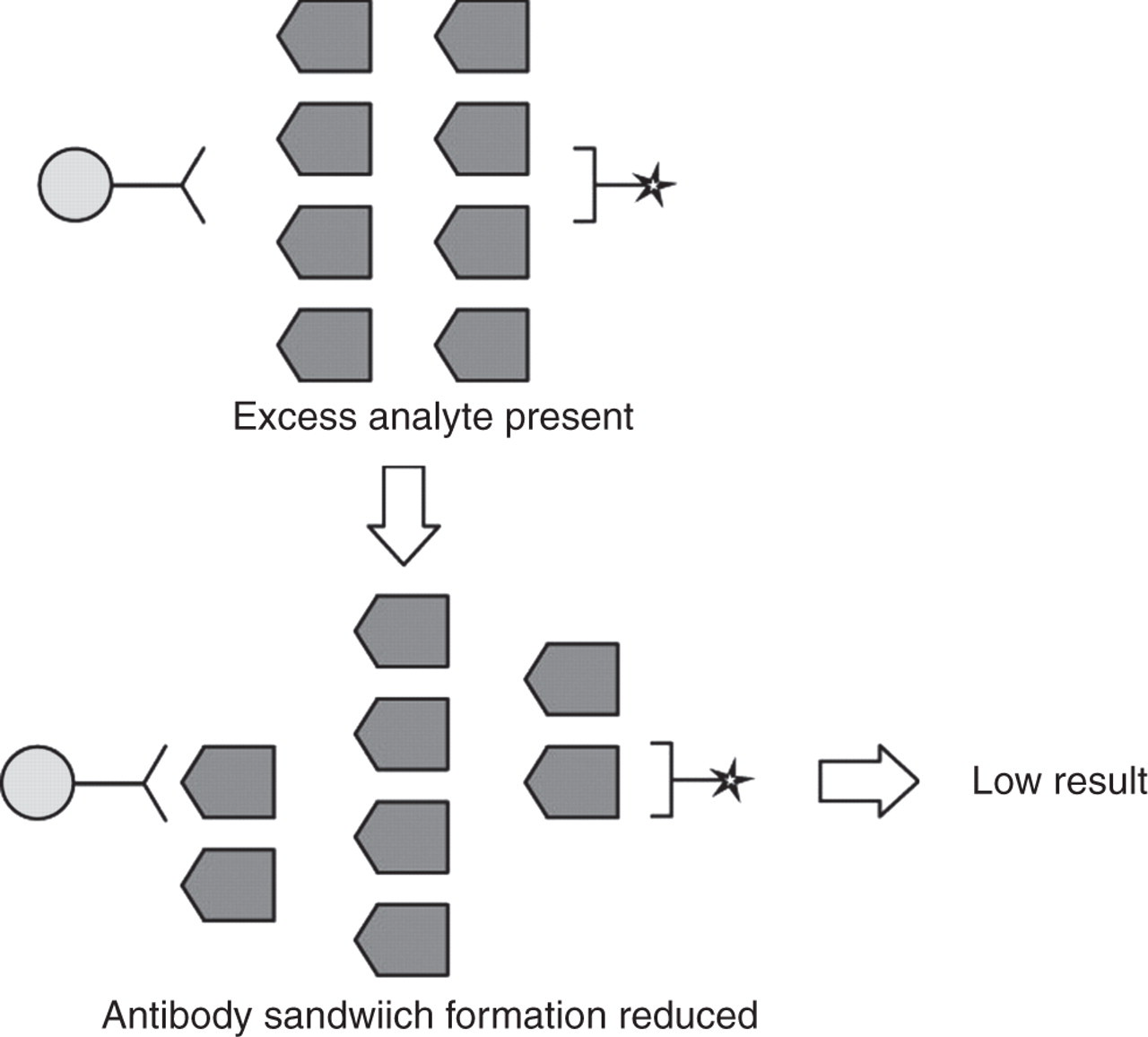
Mechanism of high-dose hook effect in two-site immunometric assay. As analyte concentrations increase to very high concentrations, both capture and signal antibodies become saturated with analyte, decreasing antibody sandwich formation. (Reprinted with permission from Seth J, Sturgeon CM. Pitfalls in immunoassays in endocrinology. Endocrinology and Metabolism In-Service Training and Continuing Education. 1993;
While it is recommended that laboratories have in place a defined procedure for minimizing the risk of an unidentified hook, 66 some hooked specimens are probably never identified. 64 In practice, failure to recognize extremely high AFP and hCG concentrations in conditions that are potentially fatal but curable (e.g. AFP in childhood hepatoblastoma and hCG in gestational trophoblastic neoplasia) constitutes a critical clinical error 64 which, at best, is likely to delay treatment. 67 Ensuring that such specimens are always checked at dilution would therefore seem highly desirable. In some laboratories, all tumour marker results above a certain threshold (usually somewhat lower than the highest standard concentration) are checked at dilution. This requires a rather empirical assessment of the risk the laboratory is willing to take of missing an unexpectedly high result.
As always, provision of relevant clinical information and good communication with clinical staff facilitates identification of specimens that are likely to be problematic and for which additional dilutions would be desirable, for example, when prolactin is measured for the first time in a patient presenting with a visual field defect. 64
Minimizing risk from interference – a shared responsibility
Although much progress has undoubtedly been made, it is clear that the often sporadic nature of many errors and interferences in immunoassay continues to present a number of challenges – in identifying erroneous results that may cause patient harm, in understanding their aetiology and in reducing their risk of recurrence. It is worth considering briefly what is required from whom in order to meet the shared responsibility of ensuring the correct result at the correct time for the correct patient.
Communication between clinical and laboratory staff
Ultimately, as has been emphasized throughout this review and elsewhere, 6,50,64 excellent communication and exchange of information between clinical and laboratory staff is absolutely essential to minimize the risk of clinical errors arising from erroneous analytical results. This should ideally include active laboratory participation in multidisciplinary team meetings. Provision of relevant clinical information with specimen requests enables laboratory staff to focus on possible sample-dependent analytical problems. Clinical staff need to be well-informed about when immunoassay results may be particularly vulnerable to interference and always encouraged to question results that do not seem in accord with the clinical picture, i.e. to expect the unexpected. 6,64 Raising awareness of the possibility of false-positive or false-negative laboratory results should be mandatory in post-graduate medical training (e.g. at Specialist Registrar level in the United Kingdom), particularly for doctors in specialties which regularly rely on immunoassay results (e.g. oncology and endocrinology). It is salutary to note that, in relation to one of the worst reported cases of antibody interference leading to unwarranted treatment (which included an unnecessary hysterectomy in a 22-year-old), doctors involved had complete confidence in the misleadingly high hCG test results, having used the same method without apparent incident for many years. 68 During the subsequent court case it was noted that although the relevant test insert advised that such consistently elevated hCG concentrations should be confirmed by an alternate method, this was not appreciated by the doctors involved as the inserts typically go to the laboratory and not the doctor. 68
Communication with patients and recording of results
Patients for whom there is evidence of endogenous assay interference should be informed that they are at risk of future false-positive results and should be encouraged to explain this whenever they have a blood specimen taken. This information should also be noted prominently in patients’ medical records and ideally in the laboratory information system. In some cases it may simply not be possible to report a result and it is necessary to attribute this to technical difficulties. Recording this is essential.
Communication with diagnostics manufacturers
Manufacturers have significantly reduced the risk of interference in many immunoassays by careful selection of antibodies, by use of better blocking reagents and through generally improved understanding of the complex immunological mechanisms involved. 10 Use of humanized antibodies in which amino acids of the assay antibodies have been exchanged by antibody engineering, for example, may alter antigenic sites and reduce interference. 5 Since most interfering antibodies bind to the Fc part of immunoglobulins, removing or modifying the Fc fragment can also significantly decrease the likelihood of interference. 5,7 Further improvement will become even more important as increasingly complex therapies involving monoclonal antibodies or drug targeting receptors (e.g. those for epidermal growth factors) enter clinical practice. In this context it is encouraging to note that the pharmaceutical sector is already expending considerable effort in validating methods to detect and characterize anti-drug antibody responses in patients. 69
Nevertheless, interferences will inevitably continue to cause problems in occasional specimens. When preliminary investigations suggest this is likely, results should be discussed at an early stage with the relevant diagnostics manufacturer. Information about the assay configuration and the antibodies used can be very helpful in designing further studies. It would be highly desirable if all manufacturers agreed to specify the measures taken to reduce interference in the information supplied with their kits, as has previously been recommended. 49 Such openness could enable much more focused and effective investigation of specimens subject to interference, would undoubtedly benefit patient care and should be strongly encouraged.
Communication with EQA providers and specialist laboratories
Managed service contracts and other changes in laboratory organization, often involving consolidation on platforms from a single manufacturer, mean that even simple checking of suspicious results by a different method is becoming increasingly complex and time-consuming to arrange. Providers of national EQA schemes are well-placed to coordinate such specimen exchange as part of their educational and collaborative remit and may in the future wish to promote this actively as part of their services to participants.
Related to this, whether investigations of suspected interference are best undertaken in-house or are more efficiently referred to a specialist referral laboratory or EQA centre is debatable. A major benefit of retaining at least some in-house capability, perhaps encompassing the preliminary investigations described above, is that awareness of how to identify problem specimens as well as the required practical skills are then actively maintained at local level. For more complex investigations, referral to a specialist laboratory may be desirable (see Appendix).
Conclusion
There have been major advances in understanding the mechanisms by which substances present in the occasional routine clinical specimen can cause false-positive or false-negative results in immunoassays, and this understanding has guided the development of in-house and commercially available assays with improved reliability. Nevertheless, clinically misleading results due to endogenous interferents in the test specimen do continue to occur sporadically. They present a unique and serious threat to patient care because they are not detectable by normal laboratory quality control procedures, are reproducible within the test system, may be clinically plausible and are relatively rare. While the first line of defence in countering such threats is the inclusion of assay robustness in the criteria for choice of test system, the importance of maintaining a high index of suspicion in inspecting assay results cannot be overstated. This is a responsibility of both laboratory and clinical staff, and requires critical review of the test result in relation to other clinical features, especially where clinical interventions are planned on the basis of the test result alone. Procedures are available for the laboratory to check whether a suspicious result might be due to endogenous interferents.
DECLARATIONS