Abstract
Background
The squamous cell carcinoma antigen (SCCA) is widely used as a serological biomarker for various cancers. There are two known SCCA molecules, SCCA1 and SCCA2. We previously found that interleukin-4 or interleukin-13, two related Th2-type cytokines that play an important role in allergic diseases, induce expression of SCCA1 and SCCA2. In this study, we examined whether combined measurements of SCCA1 and SCCA2 are useful for diagnosing atopic dermatitis (AD).
Methods
We established new enzyme-linked immunosorbent assays (ELISAs) to specifically detect SCCA1 or SCCA2. We applied serum samples from AD patients with food allergies and from cervical cancer patients to these ELISAs. We performed receiver operating characteristic analyses to diagnose AD and to distinguish AD from cervical cancer.
Results
Serum concentrations of both SCCA1 and SCCA2 were elevated in AD patients. The serum concentrations of SCCA1 and SCCA2 positively correlated with the clinical severity of AD, showing high specificity (0.86–0.88) and sensitivity (0.86) against control donors. The serum concentrations of SCCA1 and SCCA2 were elevated in cervical cancer patients; however, the SCCA2/SCCA1 ratios clearly distinguished AD patients from cervical cancer patients with high specificity (0.87) and sensitivity (0.87). Expression of SCCA2 was predominant in AD patients, whereas cervical cancer patients showed a predominance of SCCA1.
Conclusions
Combined measurements of SCCA1 and SCCA2 are very useful in estimating the severity of allergic diseases, making it possible to distinguish allergic diseases from cancers.
Introduction
The squamous cell carcinoma antigen (SCCA) was originally purified from the TA-4 complex of squamous cell carcinoma of the uterine cervix. 1 Numerous studies have established that SCCA serves as a serological biomarker for cervical cancer associated with the clinical course. 2–4 The usefulness of measuring serum SCCA concentrations has been extended to other squamous cell carcinomas including laryngeal, head and neck, vulvar, lung and oesophageal tumours. 5–8
Two SCCA molecules, SCCA1/Serpin B3 and SCCA2/Serpin B4, belong to the ovalbumin-serpin/clade B family. 9 SCCA1 and SCCA2 are homologous, with 91% identity at the amino acid concentration. However, SCCA1 and SCCA2 have distinct properties and substrate specificities: 9 SCCA1 inhibits papain-like cysteine proteases (papain, cathepsin-S, -K and -L), whereas SCCA2 inhibits both serine proteases (cathepsin G and mast cell chymase) and cysteine proteases (Der p 1 and Der f 1, major mite antigens). These behaviours suggest that SCCA1 and SCCA2 have acquired diverse and different activities during their evolution.
Interleukin (IL)-4 and IL-13, two related Th2-type cytokines, play important roles in the pathogenesis of allergic diseases. 10 To identify comprehensively IL-4- or IL-13-inducible genes, correlated with the pathogenesis of allergic diseases, we previously used a microarray approach and found that SCCA1 and SCCA2 were the most strongly induced gene products. 11 We then analysed the whole amount of SCCA in the serum samples of allergic patients, because the commercially available kit for SCCA (ARCHITECT® SCC Kit; Abbott, Abbott Park, IL, USA) does not distinguish SCCA1 from SCCA2. We found that SCCA concentrations were elevated in patients with bronchial asthma, atopic dermatitis (AD) and allergic rhinitis. 11–14 These results indicate that SCCA could be a relevant biomarker for allergic diseases downstream of IL-4 or IL-13 signals. However, a problem remains in using SCCA measurements to diagnose allergic diseases because we cannot be sure whether high serum concentrations of SCCA are caused by allergic diseases or cancers. Furthermore, it is unclear whether serum concentrations of SCCA1 and SCCA2 are differentially regulated in patients with allergic diseases or reflect the clinical severity of allergic diseases.
In this study, we established specific detection systems for SCCA1 or SCCA2 and for the first time investigated the serum concentrations of SCCA1 and SCCA2 in allergic patients. We found that combined measurements of SCCA1 and SCCA2 are very useful in estimating the severity of allergic diseases and in distinguishing allergic diseases from cancers.
Methods
Study subjects
The characteristics of AD patients with food allergies enrolled in this study are shown in Supplementary Table 1 (please see
One hundred and seventy-four patients were diagnosed as having squamous cell carcinoma of cervical cancer by histological examination. All of these cervical cancer patients had been consecutively monitored from 1984 to 1995 at Saga Medical School Hospital by gynaecologists. We obtained 176 serum samples from 174 patients at various times of their courses, and 41 serum samples from control donors who had not had cervical cancer.
Recombinant proteins
Glutathione S-transferase (GST)-fused SCCA1 or SCCA2 proteins were prepared as previously described. 15,16 GST-fused proteins were cleaved with thrombin (GE Healthcare, Little Chalfont, UK). SCCA1 or SCCA2 proteins were purified using Glutathione Sepharose 4B and Benzamidine Sepharose 6B (GE Healthcare).
Establishment of anti-SCCA1 or SCCA2 monoclonal antibodies
The footpads of six- to ten-week-old Crj:Wistar rats (Charles River Japan Inc, Kanagawa, Japan) were injected two or more times with 20 μg of recombinant human SCCA1 or SCCA2 emulsified in TiterMax Gold adjuvant (TiterMax USA, Norcross, GA, USA). Three days after the last injection, lymphocytes from popliteal, inguinal and iliac lymph nodes were fused with Sp2/O myeloma cells using polyethylene glycol treatment. Hybridomas were screened for SCCA-recognizing monoclonal antibodies (mAbs) by antigen-immobilized enzyme-linked immunosorbent assay (ELISA) with culture supernatant and a horseradish peroxidase (HRP)-conjugated anti-rat IgG antibody (GE Healthcare). Rat anti-SCCA IgGs were purified from the culture supernatant of hybridomas by protein G affinity chromatography (Protein G Sepharose, GE Healthcare). Among the antibodies obtained, clones SS8G (IgG2b/κ) and SS11G (IgG2a/κ) specifically recognized SCCA2 and SCCA1, respectively. Clone SS14B (IgG2a/κ) recognized both SCCA1 and SCCA2.
Immunoprecipitation and Western blotting
Immunoprecipitation and Western blotting for SCCA1 and SCCA2 were performed as previously described. 15–18 Briefly, 10 μg of recombinant SCCAs was incubated with 5 μg of anti-SCCA mAbs conjugated to 20 μL protein G resin (GE Healthcare) at 4°C overnight. The resin was washed three times with wash buffer (0.1 mol/L Tris-buffered saline [TBS] pH 8.6, 0.05% Tween-20), and then boiled for five minutes in 20 μL sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) sample buffer (0.1 mmol/L Tris pH 6.8, 4% SDS, 40 mmol/L dithiothreitol). The samples were separated by SDS-PAGE and were transferred onto a polyvinyldifluoride membrane. After blocking of the membrane with 5% skimmed milk, SCCAs were detected using a rabbit polyclonal anti-SCCA antibody, which recognized both SCCA1 and SCCA2. 17 This was followed by incubation with an HRP-conjugated rabbit anti-IgG antibody (GE Healthcare) and ECL Western Blotting Detection Reagents (GE Healthcare).
Measurements of SCCA1 and SCCA2 by ELISA
We established new sandwich ELISAs to specifically detect SCCA1 or SCCA2 using three rat anti-human SCCA mAbs (clones SS8G, SS11G and SS14B). SS14B (2 μg/mL) was incubated overnight at 25°C on ELISA plates (MaxiSorp; Thermo Fisher Scientific, Rochester, NY, USA). The ELISA plates were blocked with a blocking buffer (0.5% casein, TBS pH 8.0) overnight at 4°C and incubated with 50- to 1000-fold-diluted samples for 18 h at 25°C. Biotin-labelled SS11G or SS8G (50 ng/mL) for SCCA1 or SCCA2, respectively, were added, followed by incubation for two hours at 25°C. After incubation with 15,000-fold-diluted peroxidase-conjugated streptavidin (Stereospecific Detection Technologies, Baesweiler, Germany) for 90 min at 25°C, substrate solution (0.64 mmol/L 3,3′,5,5′-tetramethylbenzidine, 2.5 mmol/L H2O2) was added, followed by an incubation for 10 min at 25°C. The reaction was halted by adding stop solution (0.7 N HCl). SCCA1 or SCCA2 concentrations in the serum were calculated from the measurements of recombinant SCCA standards. We performed ELISA assays on duplicate samples.
In vitro expression analyses
Normal human epidermal keratinocytes (NHEKs; Kurabo, Osaka, Japan) or HaCaT (a spontaneously immortalized human keratinocyte cell line) cells were cultured for 72 h in KGM-2 medium (Lonza, Basel, Switzerland) at a density of 2 × 104 cells/cm2 with or without 50 ng/mL recombinant IL-13 (PeproTech, Rocky Hill, NJ, USA). The cDNAs were synthesized with a Quantitect Reverse Transcription Kit (QIAGEN, Hilden, Germany) from the total RNA extracted from cells with ISOGEN reagent (Nippon Gene, Tokyo, Japan). Quantitative analyses of SCCA mRNA expression were performed on the cDNAs using a StepOne Real-Time PCR System (Applied Biosystems, Carlsbad, CA, USA). The primers and TaqMan probes used for the analyses have been previously described. 11 Gene copy numbers were calculated using standard curves generated from SCCA plasmids with known copy numbers. The copy numbers of glyceraldehyde-3-phosphate dehydrogenase in the cells were also quantified for normalization.
Statistical analyses
All of the data were statistically analysed by the two-sided Mann–Whitney U test. For multiple comparisons, we additionally applied the Bonferroni correction with the Mann–Whitney U test. Receiver operating characteristic (ROC) analyses were performed using GraphPad Prism (GraphPad Software, San Diego, CA, USA). Statistical significance was inferred when the P value was less than 0.05.
Results
Clinical findings of AD patients with food allergies participating in this study
The correlation of the severity of eruption and eosinophil counts, total IgE concentrations or TARC (thymus and activation regulated chemokine) concentrations in AD patients in this study are shown in Supplementary Figure 1 (please see
Establishment of assay systems to specifically detect SCCA1 or SCCA2
We used three rat anti-human SCCA mAbs (clones SS8G, SS11G and SS14B) for our new sandwich ELISA to specifically detect SCCA1 or SCCA2. SS14B recognized both SCCA1 and SCCA2, whereas SS11G and SS8G recognized SCCA1 and SCCA2, respectively (Figure 1a). We used SS14B as the coating mAb and SS11G or SS8G as primary mAbs for SCCA1 or SCCA2 detection systems, respectively. We confirmed that the SCCA1 and SCCA2 assay systems detected SCCA1 or SCCA2 recombinant proteins specifically in a dose-dependent manner (Figure 1b). In these systems, the total measurement ranges for SCCA1 and SCCA2 were 300–10,000 and 150–10,000 pg/mL, respectively. The respective detection limits were 150 and 80 pg/mL, and the coefficient of variations (CVs) for intra-assay imprecision within the measuring ranges were 0.81–5.20% and 0.64–5.32% (n = 10 each), respectively.
Establishment of specific detection systems for SCCA1 and SCCA2. (a) SCCA1 or SCCA2 was immunoprecipitated with the indicated antibodies. Western blotting of the immunoprecipitates by a polyclonal antiperiostin antibody is depicted. (b) The indicated concentrations of recombinant SCCA1 (closed circles) or SCCA2 (closed squares) are as measured by enzyme-linked immunosorbent assays. SS14B is used as the coating antibody, and SS11G or SS8G are used as the primary antibodies for the specific detection of SCCA1 or SCCA2, respectively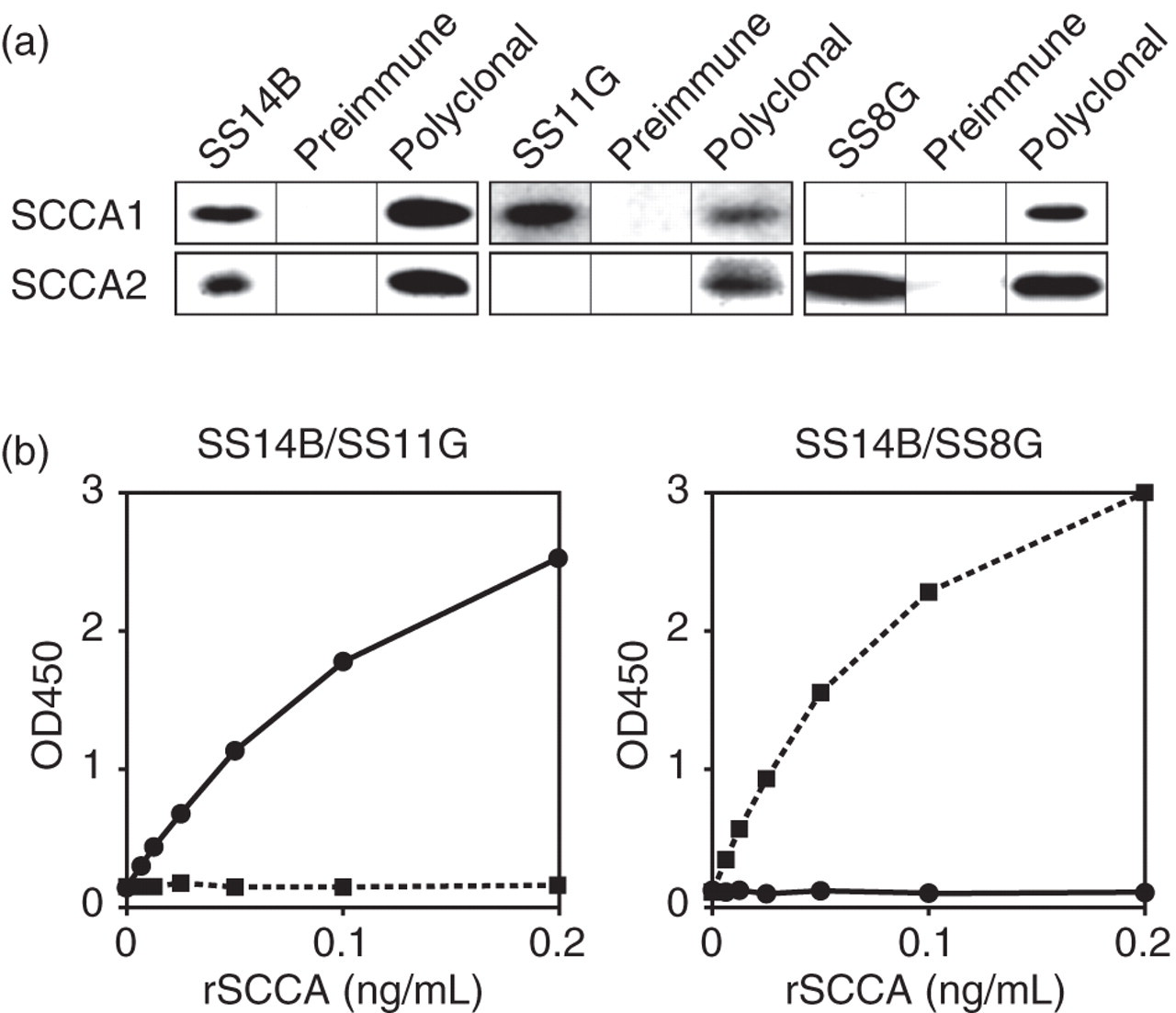
Serum SCCA1 and SCCA2 concentrations in AD patients and their correlation with severity of eruption
We applied serum samples from AD patients and control subjects to the SCCA1 and SCCA2 ELISA systems (Figure 2a). Serum SCCA1 and SCCA2 concentrations were significantly elevated in AD patients (n = 188; 2.9 ± 2.4 ng/mL for SCCA1; 3.2 ± 5.6 ng/mL for SCCA2) compared with control subjects (n = 49; 0.8 ± 0.2 ng/mL for SCCA1; 0.6 ± 0.2 ng/mL for SCCA2). Serum concentrations of TARC, a biomarker widely used for evaluation of AD,
22,23
were also upregulated in AD patients (n = 178; 1.3 ± 0.9 ng/mL) compared with control subjects (n = 40; 0.6 ± 0.2 ng/mL). Furthermore, serum SCCA1 and SCCA2 concentrations were positively correlated with the severity of skin eczema (Figures 2b and c). These results show that serum SCCA1 and SCCA2 concentrations are significantly elevated in AD patients, reflecting the severity of skin eruption.
Serum SCCA1 and SCCA2 concentrations in atopic dermatitis (AD) patients. (a) Serum concentrations of SCCA1 (left), SCCA2 (middle) or thymus and activation regulated chemokine (TARC; right) in AD patients and control donors are depicted. (b, c) Correlation of serum concentrations of SCCA1 (b) or SCCA2 (c) with eczema grades 0–4 is depicted. The bottoms and tops of the boxes, and the bottom ends of the lower whiskers, and the top ends of the upper whiskers represent the 25th, 75th, 10th and 90th percentiles, respectively. The transverse lines in the boxes are the median values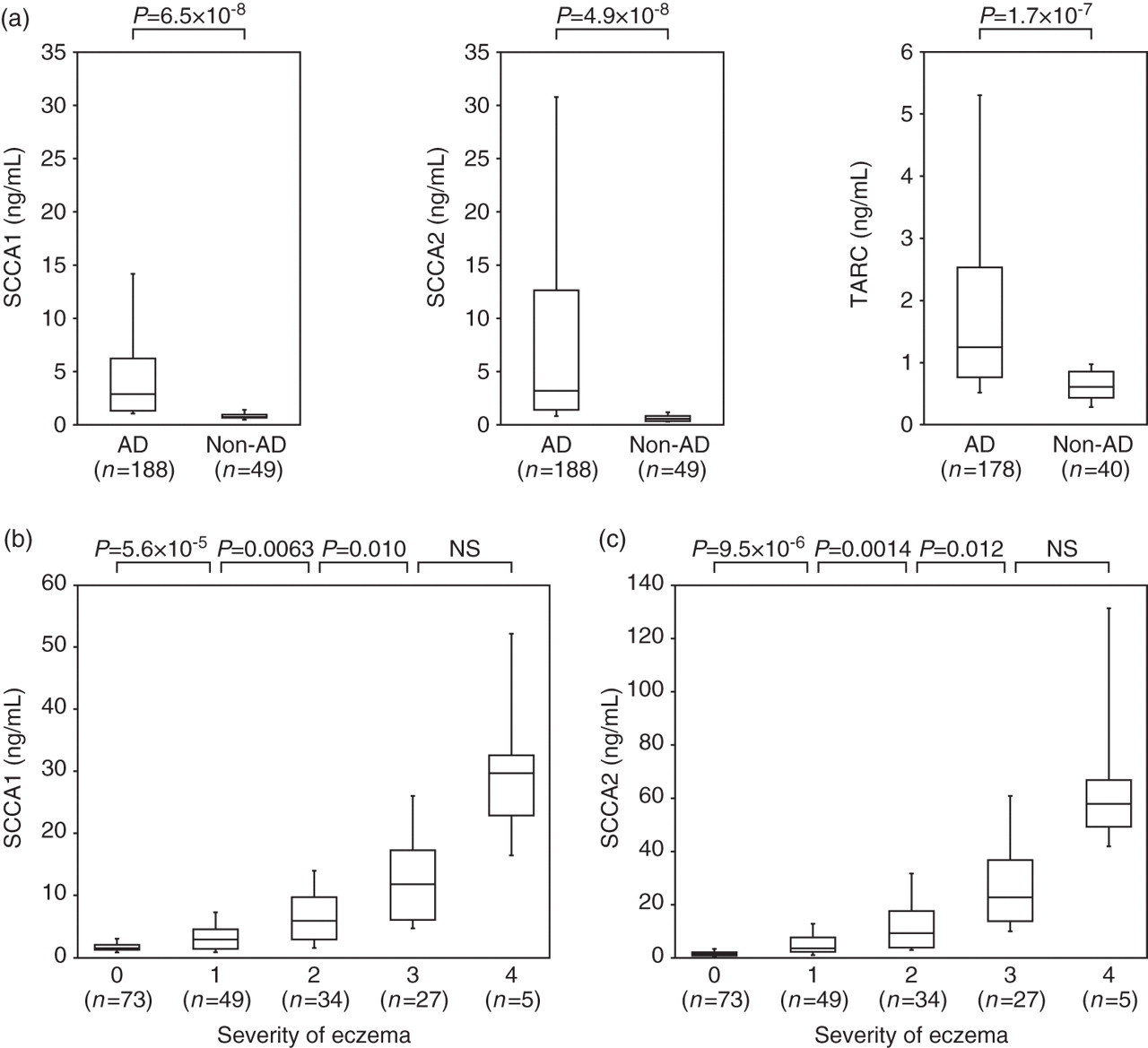
Production of SCCA2 compared with SCCA1 in AD patients
Overall, the SCCA2 concentrations were higher than the SCCA1 concentrations in AD patients and in all grades of skin eczema (Figure 2). This raised the possibility that SCCA2 is predominantly produced compared with SCCA1 in these AD patients. To explore this, we evaluated the SCCA2/SCCA1 ratios of each individual and analysed the association with the severity of skin eruption. Approximately 66% (125/188) of these AD patients demonstrated SCCA2/SCCA1 ratios greater than 1.0 (1.4 ± 0.5), whereas most of the control subjects (43/49) had ratios less than 1.0 (0.8 ± 0.1; Figure 3a). Furthermore, the SCCA2/SCCA1 ratios were elevated as the eczema grade increased (Figure 3b). Although serum concentrations of SCCA1 and SCCA2 were slightly elevated in patients with chronic urticaria (n = 19; 2.5 ± 1.1 year old), the SCCA2/SCCA1 ratios were less than 1.0 (0.86 ± 0.23, data not shown). These results demonstrate that although both serum SCCA1 and SCCA2 concentrations were elevated, serum SCCA2 concentrations were more highly elevated than SCCA1 concentrations in AD patients.
SCCA2/SCCA1 ratios in atopic dermatitis (AD) patients and cervical cancer (CC) patients. (a) SCCA2/SCCA1 ratios in AD patients, CC patients, and control donors versus AD patients or CC patients are depicted. (b) Correlation of SCCA2/SCCA1 ratios with eczema grades 0–4 is depicted. The bottoms and tops of the boxes, and the bottom ends of the lower whiskers, and the top ends of the upper whiskers represent the 25th, 75th, 10th and 90th percentiles, respectively. The transverse lines in the boxes are the median values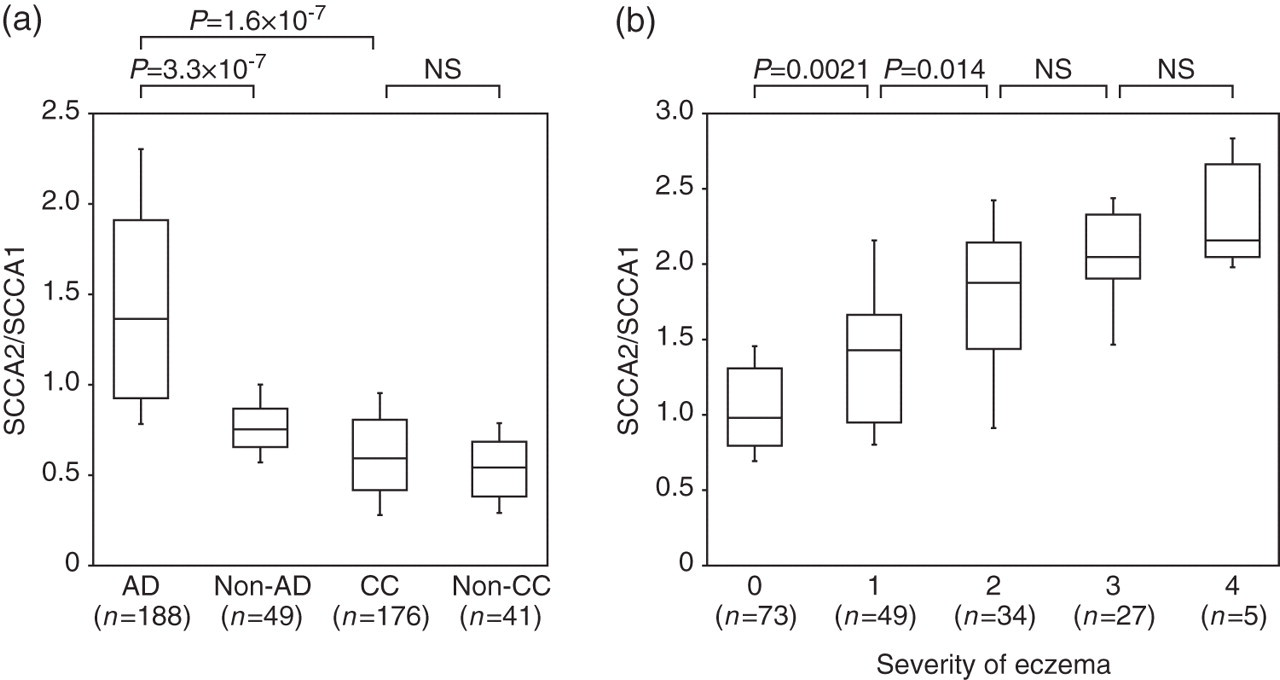
Induction of SCCA1 and SCCA2 by IL-13 in keratinocytes
The high SCCA2/SCCA1 ratios in AD patients raised the possibility that IL-13 predominantly induces expression of SCCA2 rather than SCCA1 in keratinocytes. To investigate, we stimulated NHEK and HaCaT cells with IL-13 and analysed the expression of SCCA1 and SCCA2. IL-13 induced expression of both SCCA1 and SCCA2 in NHEK and HaCaT cells; however, IL-13 significantly elevated the SCCA2/SCCA1 ratios at the mRNA and protein concentrations (Figure 4). These results suggest that SCCA2 is induced to a much greater extent than SCCA1 by IL-13 in keratinocytes.
Induction of SCCA1 and SCCA2 expression by interleukin (IL)-13 in keratinocytes. NHEK or HaCaT cells were stimulated by 50 ng/mL of IL-13 for 72 h, and mRNA or the supernatants were applied to quantitative polymerase chain reaction (a) or enzyme-linked immunosorbent assay (b), respectively, for SCCA1 and SCCA2. The open and filled bars represent the absence or presence of IL-13, respectively. Gene copies (a), protein concentrations (b) of SCCA1 and SCCA2, and the SCCA2/SCCA1 ratios of the gene copies and the protein concentrations are depicted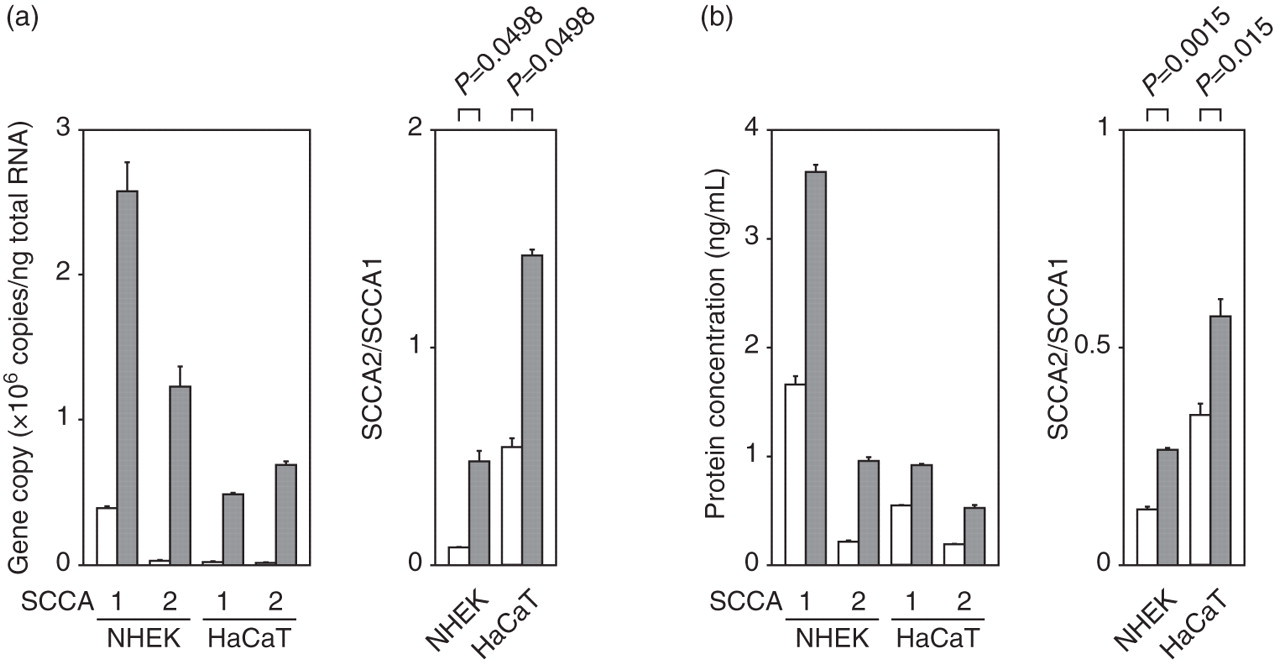
Distinction of cervical cancer from AD using SCCA2/SCCA1 ratios
We applied serum samples from cervical cancer patients to the SCCA1 and SCCA2 detection systems (Figure 5). Serum SCCA1 or SCCA2 concentrations were significantly elevated in cervical cancer patients (n = 176; 1.0 ± 1.3 ng/mL for SCCA1; 0.7 ± 0.7 ng/mL for SCCA2) compared with control subjects (n = 41; 0.7 ± 0.2 ng/mL for SCCA1; 0.4 ± 0.2 ng/mL for SCCA2). In contrast with the AD patients, most cervical cancer patients (162/176) had SCCA2/SCCA1 ratios less than 1.0, which were significantly lower than those of the AD patients (Figure 3a), particularly the active AD patients (grades 1–4, data not shown, P = 3.8 × 10−8). These results demonstrate that although serum SCCA1 and SCCA2 concentrations were elevated in both AD patients and cervical cancer patients, the relative production of SCCA2 and SCCA1 is distinct in these patients.
Serum SCCA1 and SCCA2 concentrations in cervical cancer (CC) patients. Serum concentrations of SCCA1 (left) and SCCA2 (right) in CC patients and control donors are depicted. The bottoms and tops of the boxes, and the bottom ends of the lower whiskers, and the top ends of the upper whiskers represent the 25th, 75th, 10th and 90th percentiles, respectively. The transverse lines in the boxes are the median values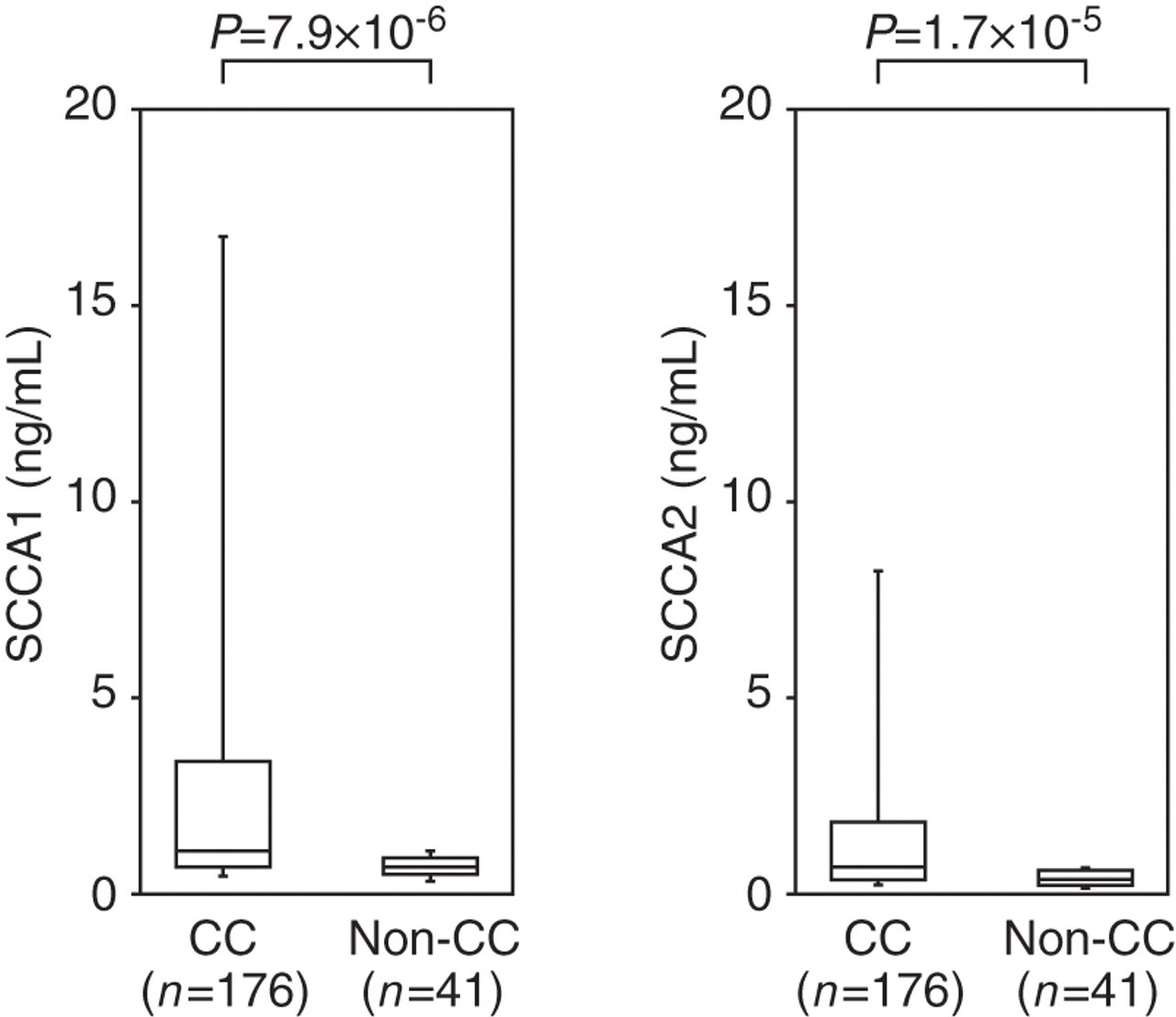
ROC analyses of SCCA1 and SCCA2
Finally, we performed ROC analyses to determine the cut-off of SCCA1 and SCCA2 absolute values, and of the SCCA2/SCCA1 ratio to diagnose AD, to distinguish AD from cervical cancer. The areas under the ROC curves (AUC) comparing overall AD patients and control donors showed statistically significant values (Figure 6a; 0.92 [95% CI, 0.88–0.96, P
< 0.0001] and 0.94 [95% CI, 0.91–0.97, P < 0.0001] for SCCA1 and SCCA2, respectively). Optimal cut-off values of serum SCCA1 and SCCA2 concentrations were 1.1 and 1.0 ng/mL, respectively, based on the Youden index. These cut-off values yielded specificities of 0.86 (SCCA1) and 0.88 (SCCA2) and a sensitivity of 0.86 (SCCA1 and SCCA2). In contrast, the AUC of TARC was 0.81, lower than those of SCCA1 and SCCA2 (95% CI, 0.75–0.87, P < 0.0001). When the cut-off value of TARC was set at 0.93 ng/mL, this resulted in a specificity of 0.93 and a sensitivity of 0.61. These results demonstrate that the SCCA1 and SCCA2 detection systems have comparable specificity and superior sensitivity to TARC.
Sensitivity and specificity measurements of SCCA1 and SCCA2 concentrations and the SCCA2/SCCA1 ratio. (a) Receiver operating characteristic (ROC) curves for serum SCCA1 (solid line), SCCA2 (dotted line) or thymus and activation regulated chemokine (TARC) (grey line) concentrations to distinguish atopic dermatitis (AD) patients from control donors are depicted. The areas under the ROC curves (AUCs) are 0.92, 0.94 and 0.81 for SCCA1, SCCA2 and TARC, respectively. The optimal cut-off values between AD patients and control donors are 1.2, 1.0 and 1.0 ng/mL based on the Youden index. (b) The ROC curve for the SCCA2/SCCA1 ratio to distinguish active AD patients (grades 1–4) from cervical cancer patients is depicted. The AUC is 0.94. The optimal cut-off value between active AD patients and cervical cancer patients is 0.9 based on the Youden index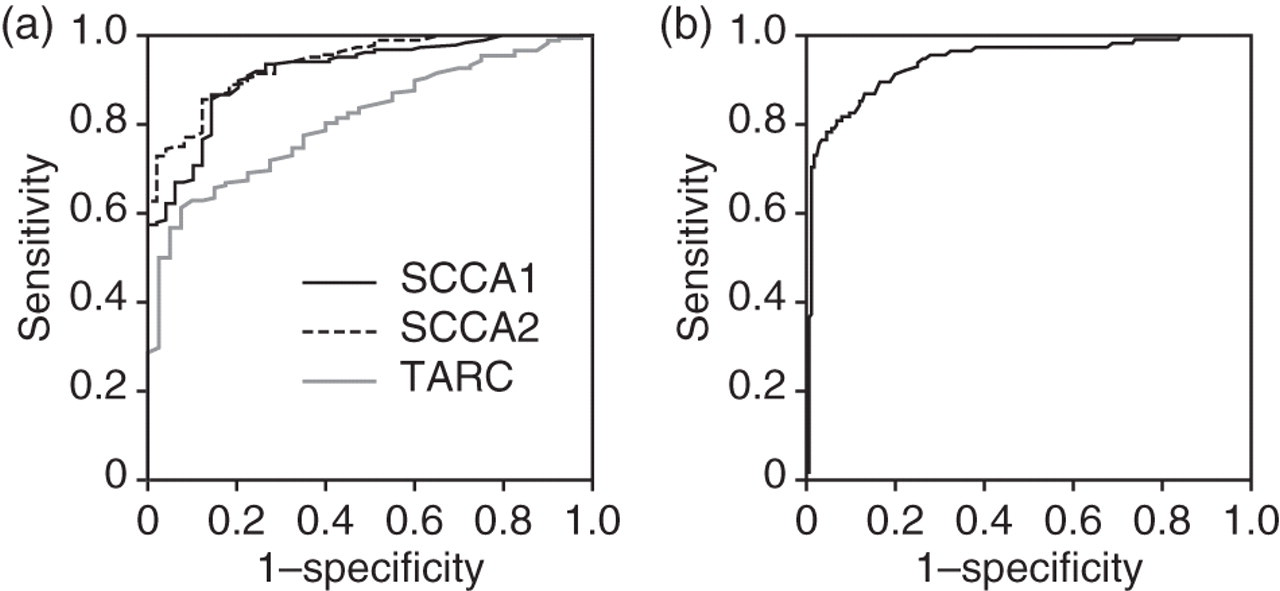
The AUC comparing active AD patients (grades 1–4) and cervical cancer patients showed a statistically significant value (Figure 6b; 0.94 [95% CI, 0.91–0.97, P < 0.0001]). The optimal cut-off value of the SCCA2/SCCA1 ratio was 0.9 based on the Youden index. This cut-off value gave a specificity of 0.87 and a sensitivity of 0.87. These results demonstrate that the SCCA2/SCCA1 ratio calculated from the SCCA1 and SCCA2 detection systems can clearly distinguish high SCCA conditions in AD patients and cervical cancer patients.
Discussion
Most laboratory tests for AD patients (measurement of specific IgE, skin pricks, atopy patches and oral challenges) aim to diagnose the existence of sensitization to allergens or to identify the allergens triggering AD, not to estimate the severity of the disease. 24,25 It has been reported that peripheral eosinophil counts and the quantity of its product, eosinophil cationic protein, are correlated with the clinical severity of AD. 19–21 However, the correlation between total IgE and disease severity is controversial. 21,26 In this study, we showed that both SCCA1 and SCCA2 serum concentrations are elevated in AD patients and that SCCA2 concentrations are higher than those of SCCA1 concentrations as clinical severity proceeds. This suggests that combined measurements of both SCCA1 and SCCA2 concentrations can be used to diagnose AD by estimating its clinical severity.
Because serum SCCA concentrations are elevated in many cancer patients with squamous cell carcinoma, 5–8 it remains an unresolved problem whether individuals with high serum concentrations of SCCA can be diagnosed as having allergies or cancers. We found that a commercially available kit for detection of SCCA proteins (ARCHITECT® SCC Kit, Abbott) can detect SCCA1 but not SCCA2 (data not shown). Therefore, we cannot estimate the serum concentrations of SCCA2 using this commercially available kit. In this study, we investigated the serum concentrations of SCCA1 and SCCA2 in allergic patients for the first time. We found that AD patients predominantly express SCCA2, whereas cervical cancer patients predominantly express SCCA1 (Figure 3a), thereby permitting the differential diagnosis of allergic patients and cancer patients. In the basal state, production of SCCA1 was predominant in normal keratinocytes and cervical cancer cells, as previously reported. 27–29 In contrast, upon stimulation with IL-4 or IL-13, keratinocytes enhance production of both SCCA1 and SCCA2, although production of SCCA2 becomes dominant, which may be one mechanism underlying the reason why SCCA2/SCCA1 ratios are high in the serum of AD patients (Figure 4). Predominant expression of SCCA1 in the patients with chronic urticaria supports the usefulness of the SCCA2/SCCA1 ratios in the specific diagnosis of AD. The precise mechanism of this shift from SCCA1 expression to SCCA2 expression is unclear thus far. No binding site for STAT6, a critical transcription factor for IL-4 or IL-13 signals, has been observed in the promoter regions of the SERPINB3 and SERPINB4 genes encoding SCCA1 and SCCA2, respectively. This would indicate that expression of SCCA1 and SCCA2 via IL-4 or IL-13 induction may be caused via trans-acting factors or de novo protein synthesis.
Although there are some analyses of SCCA1:SCCA2 ratios in cancer patients, 27,28 there is no report showing predominant elevation of SCCA2 in patients with diseases other than cancers. Çataltepe et al. 29 demonstrated that SCCA1 was elevated in amniotic fluids compared with SCCA2. To the best of our knowledge, this is the first report showing predominant elevation of SCCA2 in patients with diseases other than cancers. It is known that SCCA1 and SCCA2 have distinct properties and substrate specificities, 9 although the physiological roles of SCCA1 and SCCA2 in vivo are still obscure. Expression of SCCA2 may be induced by stimulation of IL-4 or IL-13, because SCCA2 rather than SCCA1 has some pathophysiological roles downstream of IL-4 or IL-13 signals. Further studies aimed at clarifying this point are needed.
Given that IL-4 and IL-13 are important for the pathogenesis of allergic diseases, several antagonists against IL-4 and/or IL-13 are now under development as therapeutic agents for allergic diseases. 10 It has been reported that an IL-4 mutant (pitrakinra, Aerovant™; Aerovance, Berkeley, CA, USA), an antagonist against both IL-4 and IL-13, improved lung function in asthma patients, 30 whereas a neutralizing antibody against the IL-4 receptor α chain (AG317) showed only limited efficacy in asthma patients. 31 These results may be due to the heterogeneous backgrounds of allergic patients, in that IL-4 and/or IL-13 would be deeply involved in the pathogenesis of some patients, but not of others. Co-development of molecular target drugs and diagnostics to select patients in whom the drugs would be effective, so-called companion diagnostics, are now crucial for drug development in stratified medicine. 32,33 We identified SCCA1 and SCCA2 as IL-4- or IL-13-inducible gene products. 11 It is possible that serum SCCA1 and SCCA2 concentrations reflect the involvement of IL-4 and/or IL-13 in patients. We hope to improve the efficacy of the therapeutic agents targeting IL-4 and/or IL-13 signals by applying combined measurements of the serum SCCA1 and SCCA2 concentrations to their companion diagnostics.
DECLARATIONS
