Abstract
Background
Calcium exists in human blood in a free form and in a form bound to plasma proteins, principally albumin. Since it is the ionized form that is biologically active, it has long been common practice to present calcium adjusted on the basis of serum albumin concentration. The concept of adjusted calcium has only been evaluated in adults. In this study, we evaluated the use of the adult-adjusted equation to report calcium in children.
Methods
We searched the laboratory information system over three teaching hospitals for young patients aged between newborn and 18 years old with a request for calcium and albumin analysis but with no evidence of disturbances of calcium homeostasis. These data were organized on the basis of age and was separated into four age groups (birth to 1 month old, 1 month to 1 year old, 1 to 5 years old and 5 to 18 years old). These data were subjected to regression analysis to derive the calcium-adjusted equation for each age group.
Results
There is an inverse relationship between the bias value and the age. The younger the age, the higher the difference between the adjusted calcium calculated by the adult equation and that calculated by the age-specific equation. This pattern was maintained on all sites.
Conclusion
For all sites, the adult-adjusted calcium equation may be used to calculate the adjusted calcium for children aged one year old and above.
Introduction
It is widely accepted that total serum calcium concentrations are unreliable markers of the physiologically important ionized calcium (iCa) fraction in serum. Approximately 45% of total serum calcium is protein-bound, principally to albumin, 10% is bound to small anions and 45% is ionized. 1 Calcium–albumin binding is affected by abnormalities in the plasma protein concentration, acid–base status and by the presence of molecules such as drugs or fatty acids that shift the balance between the bound and the free fraction of calcium. 2,3 In order to compensate for these effects, calcium concentrations are adjusted to allow for variations in serum albumin as an aid to improve clinical interpretation. However, to date, this equation has only been validated in adults. 4 In the absence of a valid equation to report calcium in neonates and children, clinicians and laboratories either use total serum calcium or use the adult regression equation to obtain an adjusted calcium value. This may not be valid because the factors that influence calcium binding to albumin are frequent in the sick neonates and children and, moreover, in neonates, other proteins may be more important. 5 Indeed, at present, the use of iCa measurements in neonates and children may represent the most valid option, although it is not the most practical one.
The aim of this study was to evaluate whether the adult calcium adjustment equation could be used to report adjusted calcium concentrations in children. For this purpose, we derived age-dependent adjustment equations for neonates, children and adults. Additionally, we validated the derived equations for neonates by comparing their derived calcium values with concurrent direct measurement of iCa.
Methods
Data collection
Data were collected from our Laboratory Information System which serves three Teaching Hospitals in West Yorkshire, UK: Bradford Royal Infirmary (BRI), Leeds General Infirmary (LGI) and St James’ University Hospital (SJUH). Data were extracted for all patients fulfilling the criteria detailed below who had calcium and albumin measurements performed in 2007 and 2008. The data were then subdivided into two files: patients less than 18 years old (paediatric data) and patients greater than 18 years old (adult data). In addition to total calcium and albumin, alkaline phosphatase (ALP), creatinine and urea results were also collected.
Adult data
The selection of adult patients was based on previously published criteria. 6 All patients attending Endocrinology, Haematology, Oncology or Nephrology outpatient clinics were excluded. Laboratory exclusion criteria were serum urea concentration >15 mmol/L (or 2 × upper reference limit [URL]), serum creatinine concentration >200 μmol/L (1.4 × URL), potassium concentration <3.5 mmol/L or alanine transaminase activity >URL. Only a single result per patient was collected and in all cases this was the first result for the patient.
Due to the large size of the adult groups on each site, the calcium and albumin measurements used to derive the equation were only collected for September and October of each collection year. This time period gave 14,812 and 17,613 data points for calcium and albumin for 2007 and 2008, respectively. The adult data were sorted into three groups by hospital location and used to derive an adult equation for each site.
Paediatric data
The selection of patients under 18 years was based on the same criteria that were applied for the adults except that the upper reference limit for creatinine, urea and ALP data were defined by age and gender. 7–9 Again, only one data-set was collected for each patient. The total number of calcium and albumin measurements from all sites was 17,429 and 18,000 for 2007 and 2008, respectively. The data were sorted by site into four age groups: neonates (birth to 1 month old), 1–12 months old, 1–5 years old and 5–18 years old.
Derivation of calcium-adjusting equation
The regression coefficient of calcium on albumin and the calcium intercept at zero albumin was determined using Deming regression. The intercept value was taken to be the mean of the non-protein-bound calcium concentration of the population. This value was subtracted from the population mean calcium to give the average protein-bound calcium. The values for the slope, intercept and the mean total calcium are placed in the adjustment equation as previously described:
6
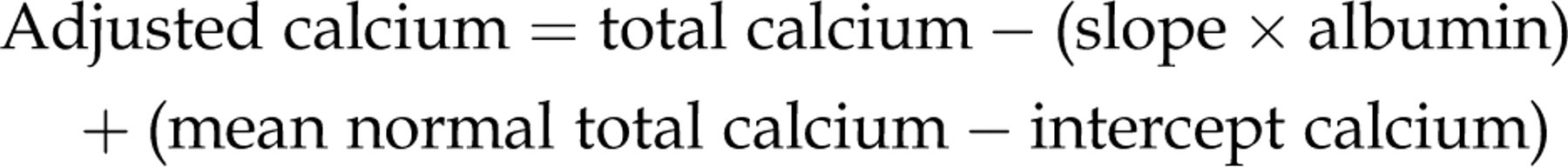
For example, if the regression equation was:
Slope = 0.0216 and intercept = 1.47 and the mean value for total calcium mean was 2.37 mmol/L, then:
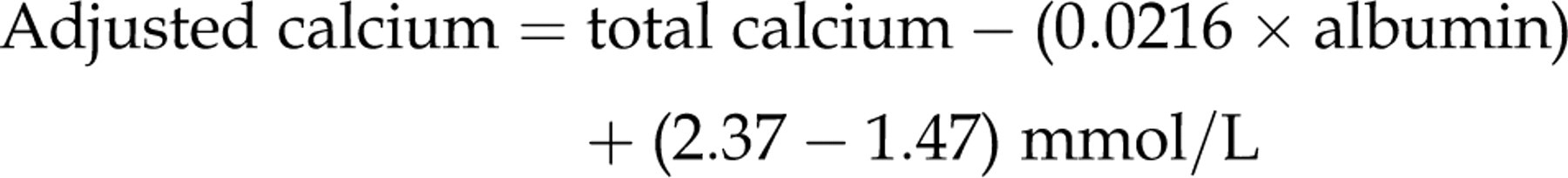
The equation is rearranged to give the final adjustment equation:
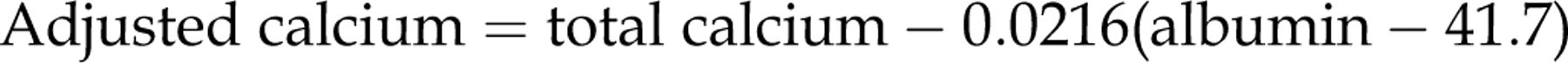
Validation of the regression equations
The calcium-adjusted equations for all age groups were validated by comparing the two consecutive periods of 12 months (2007 versus 2008).
Comparison of total, adjusted and iCa in neonates
Paired samples were taken into serum tubes for measurement of total calcium and albumin and heparinized blood gas syringes for iCa (Pulsator; Smiths Medical International Ltd, Kent, UK). Samples were taken from 61 neonates who were being mechanically ventilated in the neonatal medical unit in SJUH. Venous and arterial samples were taken as per normal practice. This study used those routine samples so no extra puncture or blood volume was required. The same exclusion criteria that were used to filter out a healthy population for the derivation of the adjusted calcium equation were applied to select this group of patients.
Routine adult equation
A single calcium-adjusted equation is routinely used to calculate adjusted calcium from all Leeds and Bradford hospital sites. This equation is:
Adjusted calcium = total calcium − 0.020 (albumin − 40).
This equation has been locally derived as per the Payne et al. method.
Analytical methods
Imprecision data for total calcium and albumin methods over the three laboratories
The data are shown as a median and range of % coefficient of variation over the 12-month period. n= 11,520 per site and test
NA, not applicable; QC, quality control; LGI, Leeds General Infirmary; SJUH, St James’ University Hospital; BRI, Bradford Royal Infirmary
Statistical analysis
A normal probability plot and Deming regression were constructed to derive the slope and the mean of the non-protein-bound calcium (intercept) and to calculate the mean calcium in each population. Altman–Bland analyses were performed to assess the difference in results using the newly derived paediatric equations against the adult site-specific equation. The one-way analysis of variance (ANOVA) was used to test the statistical significance between the albumin mean from the different age groups and the adult albumin mean. Regression of iCa on adjusted calcium for neonate was performed according to the linear regression method. All statistical analysis was performed using the Analyse-it add-in package (version 2.10) for Microsoft Excel (
Results
Regression equation for each hospital over the 2007 period for each children group
Bias is defined as the percentage difference between Adj Ca calculated by the adult equation (considered as a reference method) and child age-specific equation. N, number of points; M, month; T.Ca, total calcium; Adj Ca, adjusted calcium; LGI, Leeds General Infirmary Hospital; SJUH, St James' University Hospital; BRI, Bradford Royal Infirmary Hospital
Comparing albumin mean for different age groups
Albumin mean concentration for all age groups
LGI, Leeds General Infirmary; SJUH, St James’ University Hospital; BRI, Bradford Royal Infirmary
*Statistical significance at 95% confidence level
Validation of the regression equations
Mean difference between adjusted calcium values derived from 2007 and 2008 equations, P< 0.0001 for all age and site groups
LGI, Leeds General Infirmary Hospital; SJUH, St James' University Hospital; BRI, Bradford Royal Infirmary Hospital; CI, confidence interval
Validation of adult site-specific equations to routine calcium-adjusted equation in use
LGI, Leeds General Infirmary; SJUH, St James’ University Hospital; BRI, Bradford Royal Infirmary
Validation of regression equations with iCa for neonates
Sixty-one paired samples for iCa and adjusted calcium were collected from neonates at SJUH. The comparison between the iCa and adjusted calcium that was calculated from the neonates SJUH site-specific equation is as follows (Figure 1a):
Comparison of pH-adjusted measured ionized calcium and adjusted calcium in neonates (n= 61). The regression equations are: (a) adjusted calcium (neonate SJUH equation) = 1.381 + 1.105iCa mmol/L R
2= 0.33. (b) adjusted calcium (adult equation) = 0.8847 + 1.282iCa mmol/L R
2= 0.61 SJUH, St James' University Hospital
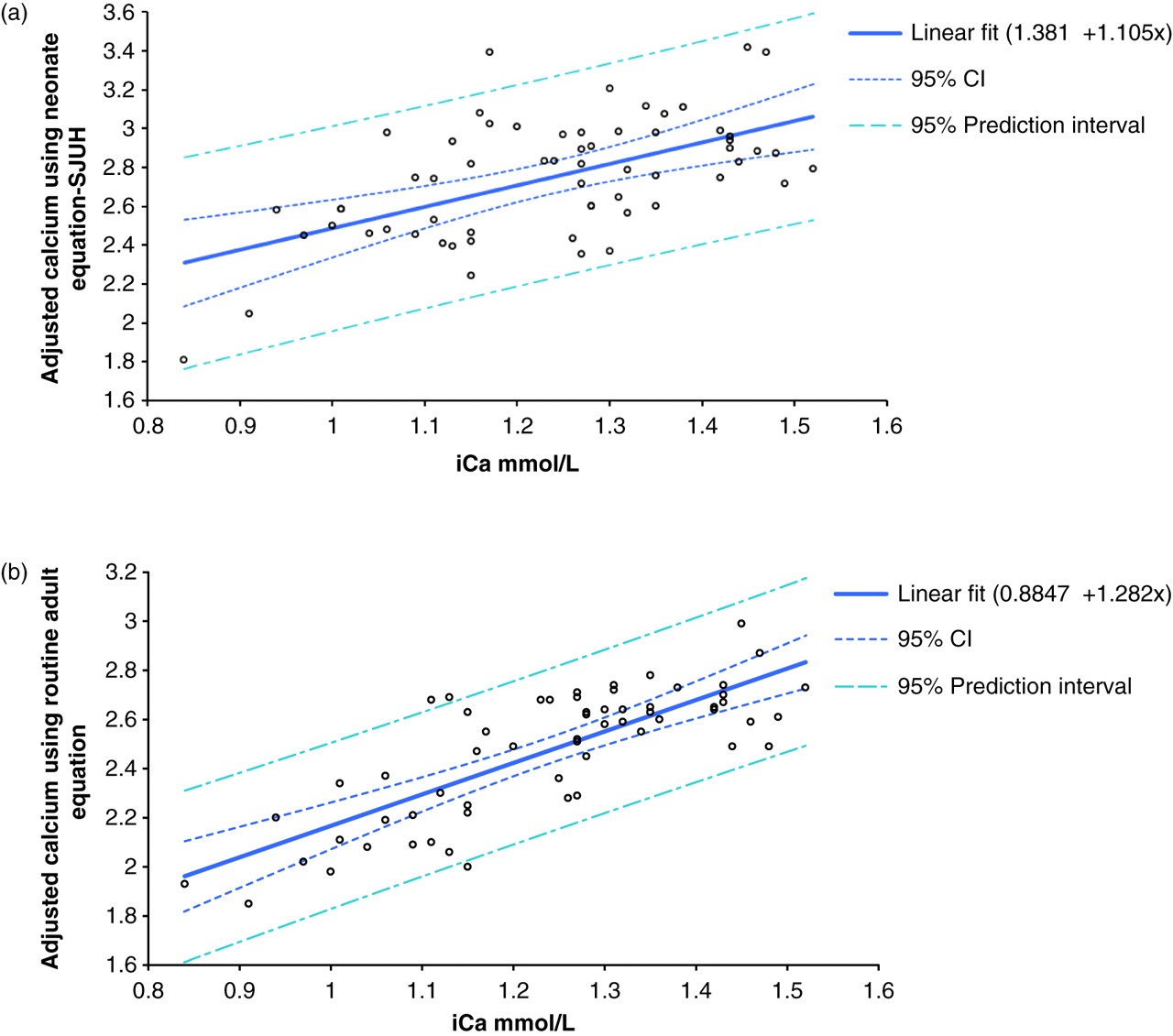
The comparison between the iCa and the routine adult adjusted calcium for the 61 patients included in this study is as follows (Figure 1b):

Using iCa as a gold standard (reference range 1.11–1.32 mmol/L), the number of patients in whom calcium status was correctly predicted using the routine adult-adjusted calcium equation was 40/61 (66%), by SJUH site-specific neonate equation was 33/61 (54%) and by total calcium was 27/61 (44%). However, both total calcium and neonate site-specific equation over-estimated calcium.
In an attempt to derive a paediatric equation from a larger sample size, data for two age groups, birth to one month and one month to one year old for 2007 and 2008, were merged, and an R
2 value of 0.27 (n= 615) was obtained:

Discussion
In adults, calcium adjustment equations have been widely used for many years to account for variations in serum albumin and allow for more accurate clinical interpretation of calcium results. We have evaluated the use of adjusted calcium in children for the first time and we have shown that the relationship between calcium and albumin is significantly different in children of all ages compared with adults, although the effect is most marked, and clinically important, in neonates. The equations for children over one month of age had different biases that varied between locations, and differed from the adult equations. This effect may be due to case-mix (e.g. the SJUH site is a referral centre and it receives the most complex and severe cases) as identical analytical methods, analytical protocol and calibrating materials (ICPMS assigned value) were in use at all three sites. 6 Furthermore, calcium and albumin methods had consistent, stable performance across sites and over the period of study. During the period of this study, the performance of both albumin and calcium methods were within the allowable limits (in-house derived quality specifications for albumin and calcium were 7% and 10% allowable total error, respectively. This is consistent with a maximum allowable bias of 3% and 4% for albumin and calcium, respectively). In support of this point, data from adult-adjusted calcium equations over a two-year period (2007–2008) showed a difference of no more than 1.7% across sites and time. It is a common practice in our network of laboratories to check the validity of adjusted calcium equation annually. The validity of this equation is checked by comparing the mean of adjusted calcium from this equation versus adult site-specific equations. A mean difference of ≤±2% between sites is rendered acceptable according to our in-house quality specifications. The equation was checked and revalidated if there was a change to albumin or calcium methodologies, or performances. Our data show that the currently in-use adult adjusted equation was valid over years and sites due to a regular and effective quality control check for albumin and calcium assays.
However, our data showed that the newly derived site and population-specific equation was insensitive to predict the correct calcium status in neonates when compared with iCa. The explanation for the discrepancy in the neonates is complex and can be attributed to a number of factors. Our routine adult calcium-adjusted equation was derived using the Payne et al. 6 exclusion criteria and the same criteria have been applied to the children population in this study. Payne et al. criteria included adult patients with creatinine of less <200 μmol/L (or 1.4 URL). However, the inclusion of children with creatinine as high as 1.4 × URL of the age-related reference range may have resulted in a sick population with varying degrees of impaired renal function. Further evidence for this population being unwell was indicated by the low serum albumin which acts as a surrogate for the acute phase effect of inflammation. Indeed, the largest effect was seen on neonates in special care baby units. The effect of such a degree of ill health has led three other groups to suggest that adjustment equations are too insensitive to be of clinical use in critically ill adults and this effect was particularly marked in the false-negative rates for the detection of hypocalcaemia. 11–13
Mimouni et al. 11 added albumin to cord blood in vitro and found an incremental lowering of iCa with added albumin. It is not known if this would only be a problem in children with albumin infusions. A further factor is the acid-base status of the neonate. We explored this hypothesis by comparing adjusted calcium with iCa in a series of 61 neonates. Their mean pH was found to be 7.32 (CI 7.30–7.34) which is another marker of ill health. In support of this concept, other groups of researchers found that critical illness alters the affinity of calcium binding to albumin. 14 This proposes another reason for the poor agreement of children equations and iCa.
Our finding is in agreement with other authors that have explored the relationship between adjusted calcium, total calcium and iCa in critically ill patients, in patients with renal failure, patients with hypercalcaemia of malignancy, very elderly patients or renal failure patients. 12,13,15–17 However, our study differs from most of these studies in that the previous studies have applied published equations for calcium correction rather than a derived equation from the studied population. We believe this type of practice may weaken the validity of the conclusion.
The poor correlation between iCa and adjusted calcium estimation may be partly attributed to the small size of the population used to derive paediatric equations for the under five years age group. We have used large data-sets to derive our results but because of the stringent exclusion criteria to include a healthy population and single sample per individual, the actual final sample size in each group was relatively small even though we searched three large teaching hospitals serving a population of approximately 1.4 million. An attempt to generate a larger sample size has not improved the correlation between the amended equation and iCa. The fact that mixing age groups has worsened the relationship between newly derived equation and iCa suggests that the combined age groups population is no longer mimicking the original population used to measure iCa.
Ideally, the comparison between different forms of calcium measurements such as total calcium and iCa should be evaluated against adjusted equations that have been specifically derived for the laboratory where the study has been performed and the studied population. This concept has been supported by a study published in 1978. 16 In this study, Ladenson et al. assessed the ability of a number of published equations for calcium correction (including the Payne formula) and total calcium to predict the calcium status in a large sample of patients with various disorders. The author found that when algorithms were adjusted to their own population, the adjusted algorithms performed better than all the published formula. Ladenson et al. 16 also found that the Payne formula gave a better prediction for calcium status when the formula was applied to a similar population, to whom the original formula was derived for. The findings of Ladenson et al. were later supported by Barth et al., 6 who showed that adjusted calcium equations differ significantly between laboratories and recommended that laboratories should derive their own equation. This difference was attributed to the use of different methodologies for albumin and calcium measurement, different performance specifications or different population.
This study also shows that the regression between calcium and albumin differed between neonatal units and over time. Therefore, it would have been an impractical suggestion to use local regression equations in view of the difficulty in collecting data to validate age- and site-specific equations. Furthermore, our data show that total calcium was inferior to adjusted calcium in predicting the calcaemic status in this group of patients. We, therefore, propose that iCa is used in this group of patients.
Conclusion
Our data show that adjusted calcium derived from adult data-sets can be used for children aged over one year. The mean difference in adjusted calcium value is probably too great in children less than one year. We would propose that iCa measurements are used for this group in view of the difficulty for most units to collect sufficiently large data-sets and the problems introduced by method differences if data-sets are combined.
DECLARATIONS
