Abstract
Background
Serial troponin measurement is important for the diagnosis of myocardial infarction. As troponin concentrations approach the cut-off for detectable myocardial necrosis, smaller changes in troponin as a result of decreased in vitro stability may be sufficient to generate changes of apparent clinical significance. This is particularly relevant regarding retrospective ‘add-on’ testing on samples several hours after venepuncture. We investigated the stability of troponin I (cTnI) at values close to the 99th percentile limit.
Methods
Serum samples with baseline cTnI concentrations in the range 0.04–0.15 μg/L were analysed routinely using the Siemens TnI-Ultra assay. Follow-up analysis was carried out at 6, 9, 12, 24 and 48 h post-venepuncture after storage at room temperature and in the cold room.
Results
There was a significant decrease in cTnI concentration after 6 h post-venepuncture compared with baseline levels (P < 0.001). The maximum percentage change was −17.1% observed after 48 h storage at room temperature. Of samples with baseline cTnI concentration 0.040 μg/L, 80% had a cTnI concentration below 0.040 μg/L on re-analysis after 6 h.
Conclusions
Requests for retrospective addition of troponin measurement on samples several hours after venepuncture is commonplace in many laboratories. Analysis of samples drawn >6 h previously may produce values below the cut-off for myocardial necrosis (0.040 μg/L) that would have been detectable if measured earlier as a result of decreased in vitro stability. Significant percentage decreases in cTnI concentration following storage may also have implications in lowering the threshold for an apparently clinically significant change to occur.
Introduction
Cardiac troponin is the gold standard biomarker for the detection of myocardial necrosis. Expert panels have defined an increased troponin concentration at >99th percentile of a reference population with an assay coefficient of variation (CV) ≤ 10%. 1 The Siemens ADVIA Centaur XP® TnI-Ultra assay has a 99th percentile value of 0.04 μg/L. An in-house evaluation demonstrated a troponin I (cTnI) cut-off concentration for detection of myocardial necrosis of 0.044 μg/L corresponding to the 99th percentile value at 10% CV.
Serial cTnI testing is important in the diagnosis of myocardial infarction (MI) and risk stratification for future coronary events. 1 For investigation of suspected MI, samples for cTnI measurement should be drawn on first assessment and repeated 6–9 h later. If initial concentrations are not elevated and clinical suspicion remains high, an additional sample for cTnI measurement between 12 and 24 h may be required. In many circumstances, cTnI measurement is often only considered when the patient has been admitted to an acute medical receiving ward several hours after initial presentation at Accident and Emergency. Clinicians often request the addition of cTnI measurement retrospectively on samples drawn in the preceding 24 h, often several hours after venepuncture. At concentrations close to the cut-off for detectable myocardial necrosis, the instability of cTnI in vitro may result in concentrations falling below the cut-off. A previous recommendation for a rise or fall of 20% in cTnI concentration on successive samples being clinically suggestive of MI has been generally adopted. 2 As cTnI concentrations approach the cut-off for myocardial necrosis, smaller absolute changes in cTnI may be sufficient to generate a 20% change. Therefore the stability of cTnI at these low concentrations may become clinically significant.
Methods
The study was carried out at Aberdeen Royal Infirmary from January to August 2010. The aim was to determine the in vitro stability of cTnI at concentrations close to the 99th percentile concentration for myocardial necrosis. All blood samples (4–5 mL) were collected into BD Vacutainer® SST™ II Advance gel tubes (Becton Dickinson, Franklin Lakes, NJ, USA). Baseline serum cTnI concentrations in the range 0.04–0.15 μg/L (n = 76) were analysed routinely on arrival at the laboratory (median time from venepuncture 2 h 15 min) using the Siemens ADVIA Centaur XP® TnI-Ultra assay (Siemens Healthcare Diagnostics, Deerfield, IL, USA) and allowed to stand at room temperature. Performance characteristics of the assay are shown in Table 1. At 6 h post-venepuncture, the samples were re-analysed and split, with the primary tube continuing to be stored at room temperature (mean temperature 22°C) and an equal aliquot transferred to a second plain tube for storage in the cold room (mean temperature 6°C). Samples at both storage conditions were re-analysed at 9, 12, 24 and 48 h post-venepuncture. All temperature data were obtained using the Labguard® II (AES Laboratoire Group, Rennes, France) temperature monitoring system. Data were transformed using the equation y = Loge (0.04 + [cTnI]) and analysed by repeated measures analysis of variance and Student's paired t-test.
Analytical performance of the Siemens ADVIA TnI-Ultra assay
RCV, reference change value; CV, coefficient of variation; cTnI, troponin I
Inter- and intra-assay imprecision of the TnI-Ultra assay was determined using ADVIA Centaur XP® on serum sample pools. The reference change value at 99% probability was determined at each concentration according to the method of Fraser and Harris 7 using previously published biological variation data for cTnI 5
Results
The results demonstrated a decreasing trend in cTnI concentration with storage at both temperatures with each successive time point compared with baseline (P < 0.0001; R 2 value = 0.96). Figure 1a illustrates the percentage change in cTnI concentration from baseline at each time point at both storage temperatures. There was a significant decrease in cTnI concentration after 6 h post-venepuncture compared with baseline levels (P < 0.001), equivalent to a −5.7% median decrease (interquartile range [IQR]: −1.0 to −14.1%). The decrease was more marked in samples with baseline cTnI ≤ 0.05 μg/L (n = 26) exhibiting a median −8.8% decrease (IQR: −2.5 to −15.0%) at 6 h (data not shown). The maximum percentage change was −17.1% (IQR: −10.0 to −25.0%) observed after 48 h storage at room temperature. Of samples with baseline cTnI concentration at the cut-off for MI (0.04 μg/L) (n = 17), 80% produced a value that would have been reported as <0.04 μg/L on re-testing after 6 h (Figures 1b and c). The effect of storage temperature on cTnI concentration was apparent after 12 h, with samples stored at room temperature significantly lower than in the cold room (P = 0.03).
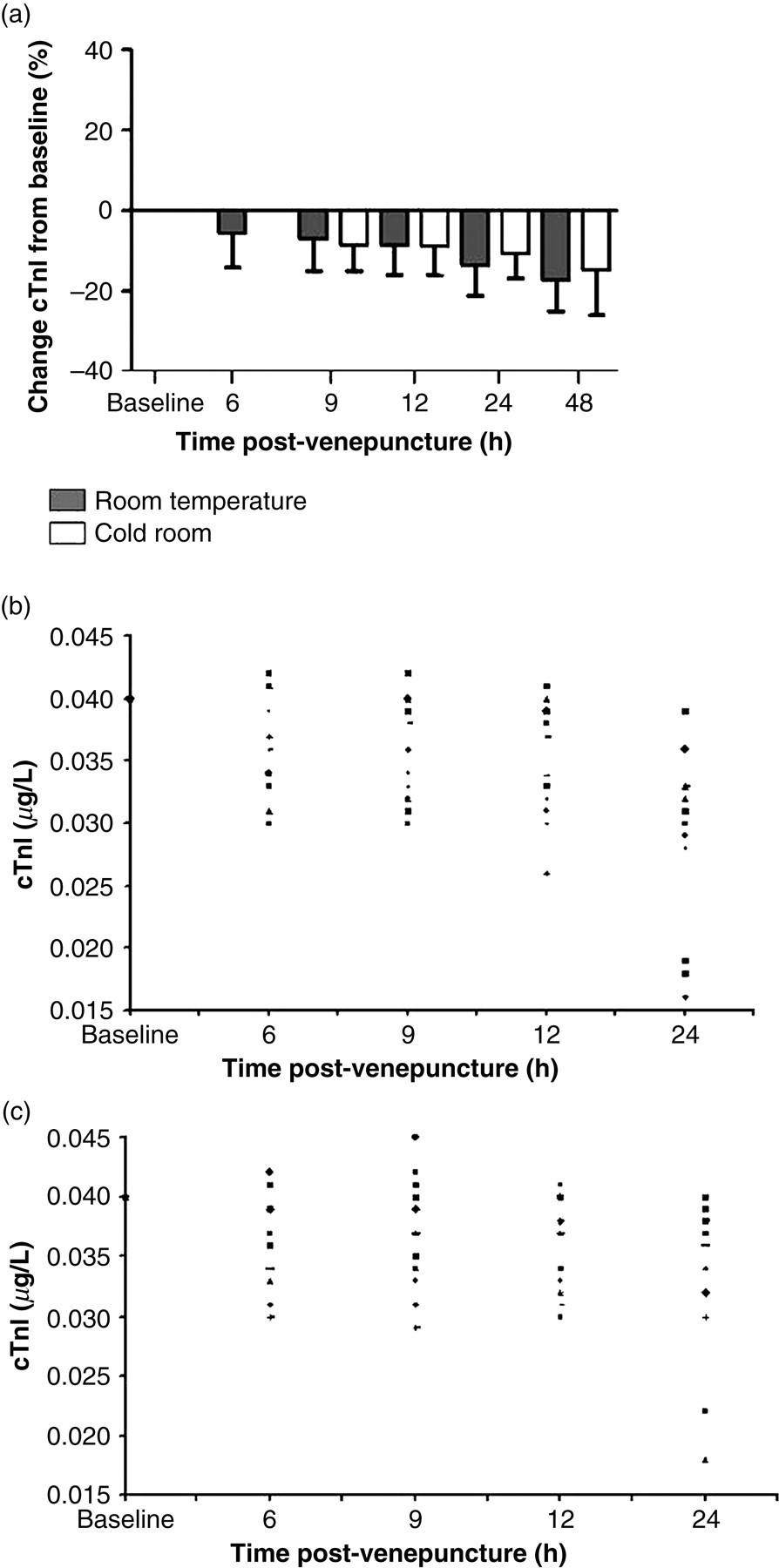
In vitro stability of troponin I (cTnI) using the Siemens TnI-Ultra assay. The stability of cTnI samples was investigated using the TnI-Ultra assay (ADVIA Centaur XP®). Serum cTnI was measured at baseline (median time 2 h 15 min post-venepuncture) and repeated after 6, 9, 12, 24 and 48 h. (a) Represents the median (and interquartile range) percentage decrease in cTnI from baseline concentration at subsequent time points post-venepuncture with storage at room temperature and in the cold room (from 9 h onwards). (b) Individual samples (n = 17) with baseline cTnI concentration at the cut-off level for myocardial necrosis (0.04 μg/L) and their subsequent cTnI levels on re-testing following storage at room temperature. (c) Storage in the cold room (from 9 h onwards)
Discussion
According to National Guidelines, patients presenting with suspected acute coronary syndrome should have serum troponin measured on arrival to guide appropriate management and treatment. 3,4 However, in many UK centres, cTnI measurement is often only considered retrospectively when the patient has been admitted to an acute medical receiving ward several hours later. Departmental audit data over a six-month period showed that approximately 17% of all add-on test requests were for cTnI measurement. A large number of these requests was carried out several hours after venepuncture, the majority of which had values below the cut-off value for detectable myocardial necrosis. Our data have highlighted some potential issues surrounding the use of cTnI. In our study, the median time from venepuncture to result for cTnI analysis was 2 h 15 min. We have observed significant percentage decreases in cTnI concentration in samples re-analysed after this time. Analysis of samples drawn 6 h previously may produce values below the cut-off for myocardial necrosis (0.04 μg/L) that might have been detectable if measured earlier as a result of decreased stability. In the current economic climate, there is significant pressure on acute units to discharge patients and isolated measurement of cTnI for ‘rule-out’ purposes may be encouraged. This practice has potential for false reassurance and possible non-investigation or inappropriate discharge from hospital.
Serial measurement of cTnI for detection of a rising or falling pattern is well established for use in the diagnosis of MI. This is particularly relevant in patients with chronic elevations in cTnI, such as that found in chronic renal failure. There has been general acceptance among clinicians of a 20% change in serial cTnI measurements as being diagnostically significant. 2 Perhaps unsurprisingly, the percentage change in cTnI is more prominent at levels close to the cut-off for myocardial necrosis. This study has demonstrated considerable percentage decreases in cTnI between analyses immediately following specimen receipt versus after just 6 h storage, lowering the threshold for an apparently clinically significant change to occur. There are large percentage decreases observed after 12–24 h storage at room temperature, with a number of samples exhibiting >20% decrease from baseline values. The stability of cTnI in vitro and the practice of retrospective testing may affect clinical decisions on samples taken >12 h previously, where a changing pattern in cTnI may be misinterpreted. The biological variation of cTnI has been reported using a prototype high-sensitivity Troponin I assay (Singulex, Inc, Hayward, CA, USA). 5 This may be used to determine reference change values for use in detecting clinically significant changes for serial cTnI values (Table 1). However, it may be some time before these data have been universally adopted for clinical practice.
Siemens Diagnostics data recommend that the TnI-Ultra assay should not be used on samples >4 h at room temperature. 6 This report has highlighted significant decreases in cTnI with storage at room temperature after 6 h post-venepuncture at concentrations close to the cut-off for detectable myocardial necrosis. These findings are particularly relevant in view of the widespread practice of retrospective ‘add-on’ measurement of cTnI on stored samples, particularly in individuals with borderline cTnI concentrations. Clinicians should be reminded that cTnI should be interpreted within the clinical context and measurement should be carried out on a contemporaneous sample if clinically indicated.
DECLARATIONS
