Abstract
Background
The accurate measurement of cortisol by immunoassay is compromised by the potential for cross-reactivity of reagent antibodies with structurally related steroids present in serum. This susceptibility is potentiated when normal steroid metabolism is altered pharmaceutically by antisteroidogenic drugs utilized in the management of Cushing's syndrome to moderate cortisol production. The clinical implications of falsely elevated cortisol results include over-treatment and unrecognized hypoadrenalism. To investigate the effect of the 11β-hydroxylase inhibitor metyrapone on serum cortisol assay, a comparison of measurement by immunoassay versus liquid chromatography-tandem mass spectrometry (LC-MS/MS) was conducted.
Methods
Cortisol was measured in serum from three patient groups: (1) patients receiving metyrapone (n = 112 samples from 10 patients); (2) control group of patients diagnosed with Cushing's syndrome currently receiving no treatment (n = 31); and (3) control group of patients with no adrenal pathology and not receiving medication known to interfere in cortisol immunoassay (n = 67).
Results
Bland–Altman plots showed agreement between methods for the control group (bias = 1.1% [−4.3 nmol/L]) and Cushing's control group (bias = 1.3% [−3.7 nmol/L]). This was in stark contrast to the metyrapone therapy group (bias = 23% [59 nmol/L]). The difference between LC-MS/MS versus immunoassay in the metyrapone therapy group positively correlated with metyrapone dose and serum 11-deoxycortisol concentration (Pearson's correlation coefficient r = 0.47, 95% CI 0.32–0.61; P < 0.0001).
Conclusions
These data show that liability of immunoassay measurement of serum cortisol to interference in patients receiving metyrapone may lead to erroneous clinical decisions concerning dose titration.
Introduction
Adrenal-directed therapies for the medical management of Cushing's syndrome, including drugs that inhibit adrenal steroidogenesis, are effective in reducing hypercortisolaemia. Metyrapone reduces cortisol and aldosterone production by inhibiting 11β-hydroxylation in the adrenal cortex (Figure 1). Approximately 50–60% of patients with Cushing's syndrome receive metyrapone treatment on a short-term basis prior to surgery, or while awaiting adrenalectomy or radiotherapy if pituitary surgery has been unsuccessful. Previous studies to assess the short-term (1–16 weeks) and long-term (median: 27 months) response to metyrapone therapy (mean dose: 2250 mg/d) showed that adequate control of hypercortisolaemia (mean serum cortisol: <400 nmol/L) was achieved in 75% of patients on short-term therapy compared with effective long-term control in 83% of patients. 1,2 The patients are often started on 250 mg metyrapone thrice daily, with dose titration up to a maximum dose of 6 mg/d. Current clinical guidance stipulates that this dose titration should be against serum cortisol concentration 3 with some centres using cortisol day curves to assess normalization of cortisol production, 4 aiming for a mean serum cortisol value through the day in the range of 150–300 nmol/L. 5
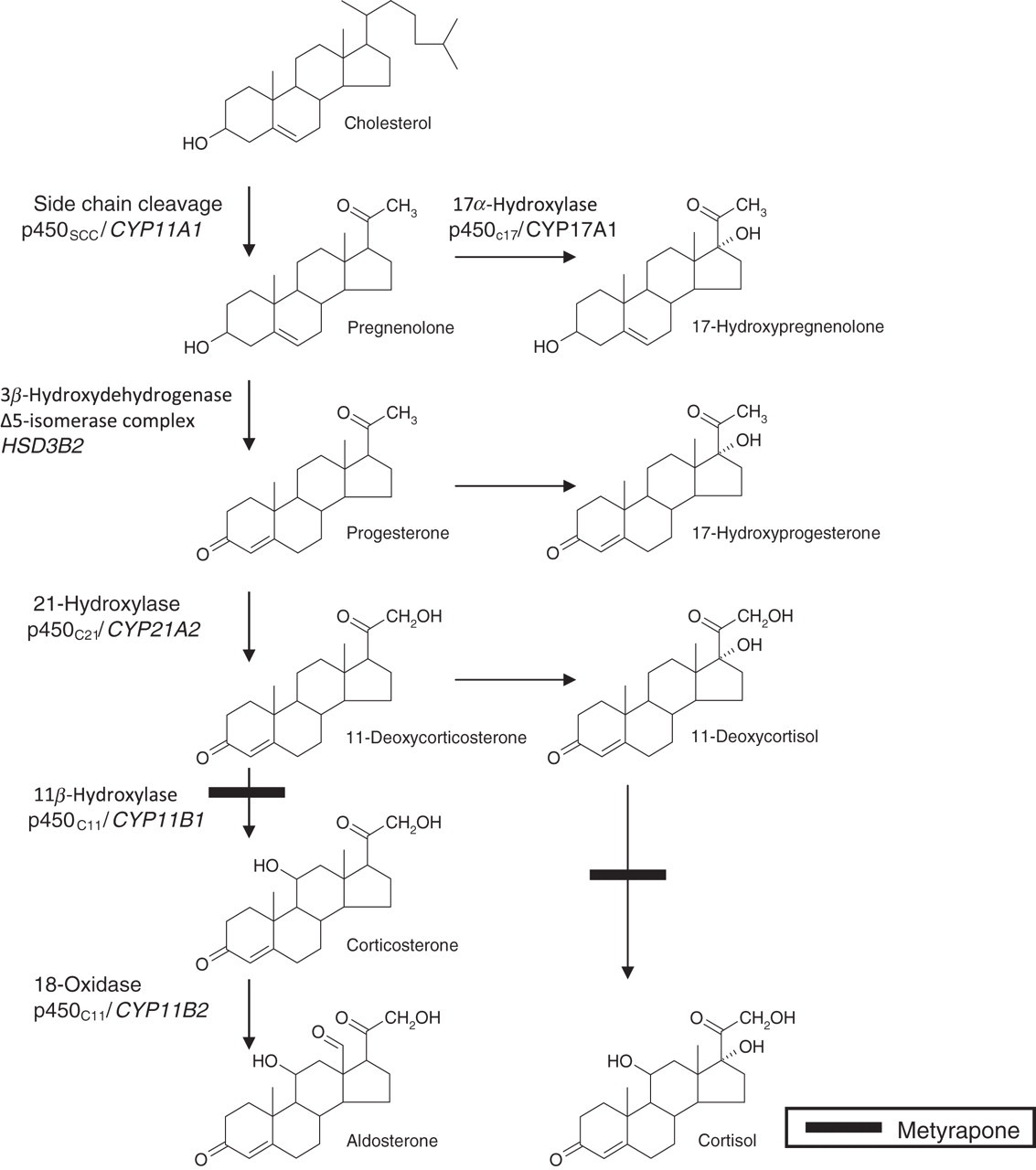
The pathway of adrenal glucocorticoid and mineralocorticoid biosynthesis. Molecular structures of the adrenal steroids are presented to illustrate the close structural homology that elicits cross-reactivity in immunoassay. Inhibition of 11β-hydroxylase by metyrapone causes an increase in serum concentrations of precursor steroids. Gene names are italicized
The measurement of cortisol in clinical biochemistry laboratories is routinely performed by immunoassay and it is well documented that these assays are susceptible to interference, with previous reports alluding to cross-reactivity in immunoassays towards cortisol precursors resulting from metyrapone blockade of the enzyme pathways. 6,7 A recent case report also highlighted the potential shortfalls in the measurement of serum cortisol by immunoassay in patients receiving metyrapone therapy. 8 These previous studies have shown that serum cortisol measurement by immunoassay may result in falsely elevated results in patients receiving metyrapone. However, to date, there has been no report addressing the extent of immunoassay interference resulting from metyrapone therapy. To resolve this question, and to assess if interference from metyrapone is dose-dependent, a comparison of serum cortisol measurement by immunoassay versus liquid chromatography-tandem mass spectrometry (LC-MS/MS) was conducted. LC-MS/MS is widely considered to be a reference method for steroid measurement with superior analytical specificity in comparison to immunoassay. 9,10 Therefore, a comparison of cortisol measurement may permit quantitative information to elucidate the extent of cross-reactivity in immunoassay. 11-Deoxycortisol (11DOC) was also measured simultaneously during LC-MS/MS analysis to assess contribution of cross-reactivity.
Methods
Patient groups
Cortisol measurements were performed on serum taken from three patient groups: (1) patients receiving metyrapone therapy (n = 112); (2) control group of patients diagnosed with Cushing's syndrome receiving no treatment at the time of sampling (n = 31); and (3) control group of patients confirmed to have no known adrenal pathology and not receiving medication known to interfere in cortisol immunoassay (n = 67). The 112 samples from the metyrapone group were day curve samples from 10 patients. Metyrapone dose information was obtained from the biochemistry request form.
Samples
Blood samples for cortisol measurement were collected into Monovette® serum gel Z separator tubes (Sarstedt Ltd, Beaumont Leys, Leicester, UK) and allowed to clot. Samples were then centrifuged at 2400
Serum cortisol and 11-deoxycortisol measurement
LC-MS/MS measurement of cortisol and 11DOC was performed as described previously. 8,11 Immunoassay of cortisol was performed on the Siemens ADVIA Centaur XP analyser (Siemens, Camberley, Surrey, UK).
Statistical methods
Statistical analyses of the data were performed using Analyse-it® statistics software package for Microsoft Excel (Analyse-it Software Ltd, Leeds, UK).
Results
The study included 210 samples. Bland–Altman plots showed agreement between immunoassay and LC-MS/MS for the measurement of serum cortisol in both the control group (bias = 1.1% [−4.3 nmol/L; 95% CI −16.7 to 8.1 nmol/L]) and Cushing's control group (bias = 1.3% [−3.7 nmol/L; 95%CI −14.3 to 6.9 nmol/L]). This was in stark contrast to the metyrapone group (bias = 23% [59.0 nmol/L; 95%CI −45.3 to 72.7 nmol/L]). Bland–Altman plots are shown in Figure 2. Equations calculated by Passing–Bablok linear regression for the control group, Cushing's control group and metyrapone group were: LC-MS/MS = 1.1 (immunoassay) – 30.57 (R 2 = 0.92); LC-MS/MS = 1.08 (immunoassay) – 17.12 (R 2 = 0.86) and LC-MS/MS = 1.01 (immunoassay) – 63.58 (R 2 = 0.74), respectively, showing that bias was predominantly constant in nature. Paired t-tests of each data-set for serum cortisol gave: P = 0.49 (control group), P = 0.48 (Cushing's control group) and P < 0.0001 (metyrapone therapy group). Mean serum 11DOC concentrations in the control group, Cushing's control group and metyrapone therapy group were 3.4, 3.0 and 136.9 nmol/L, respectively.
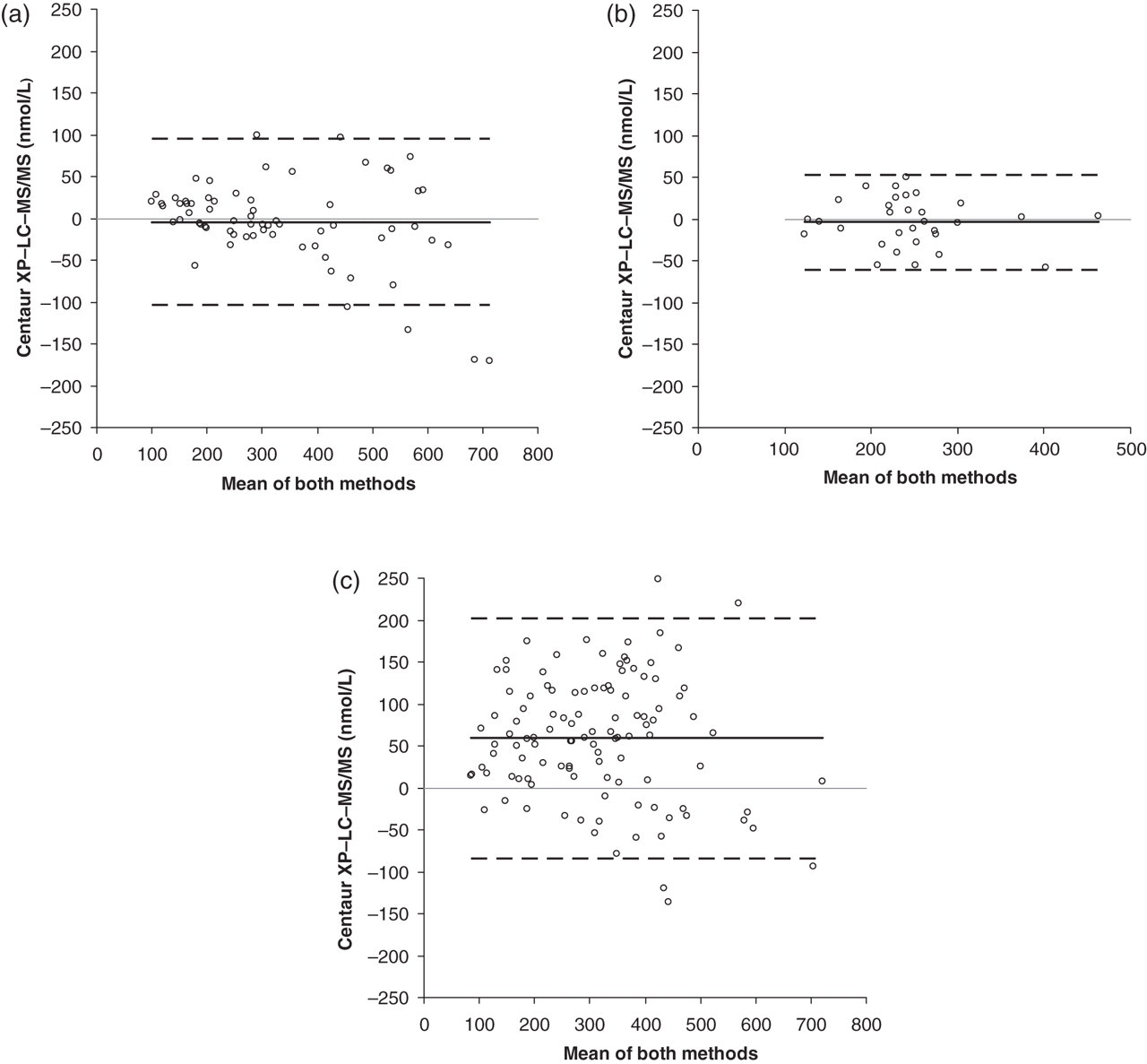
Bland–Altman analysis of serum cortisol measurement by immunoassay (Centaur XP) and LC-MS/MS. (a) Control group (n = 67); (b) Cushing's syndrome control group (n = 31); and (c) metyrapone therapy group (n = 112). The solid black line depicts the bias between methods and the dashed lines represent the 95% limits of agreement. LC-MS/MS, liquid chromatography-tandem mass spectrometry
The metyrapone group data showed that mean difference in serum cortisol results between LC-MS/MS versus immunoassay (delta cortisol) positively correlated with metyrapone dose. The mean serum 11DOC concentration also positively correlated with dose (Figure 3). Analysis of delta cortisol versus 11DOC concentration in the metyrapone group revealed a statistically significant positive linear association; Pearson's correlation coefficient r = 0.47, 95% CI 0.32–0.61 (P < 0.0001; Figure 3c).
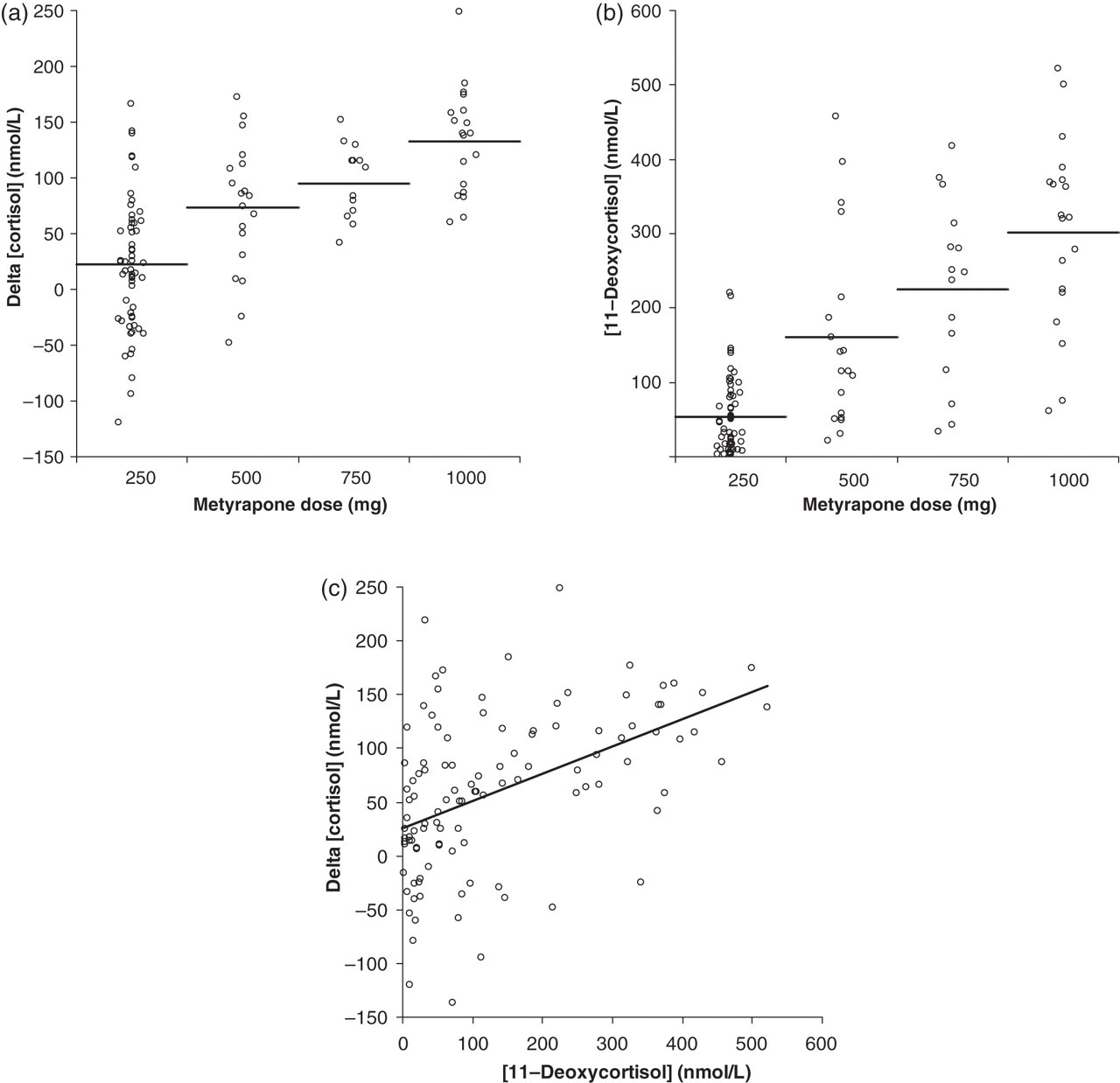
Graphs illustrating relationship between serum cortisol and 11DOC with metyrapone dose. (a) Individual value plot of difference between serum cortisol measurements (immunoassay – LC-MS/MS) with metyrapone dose and (b) serum 11DOC concentration and metyrapone dose. Horizontal lines represent mean value. (c) Scatter graph showing positive linear association between delta cortisol and serum 11DOC concentration. Pearson's correlation coefficient = 0.47. LC-MS/MS, liquid chromatography-tandem mass spectrometry; 11DOC, 11-deoxycortisol
Analysis of an individual patient's cortisol and 11DOC during metyrapone dose titration from 500–1000 mg thrice daily, illustrated a dose-dependent rise in 11DOC and a corresponding decrease in cortisol measured by LC-MS/MS and immunoassay. Critically, however, the decrease in cortisol measured by immunoassay (−52 nmol/L) was less than the corresponding decrease determined by LC-MS/MS measurement (−104 nmol/L; Figure 4).
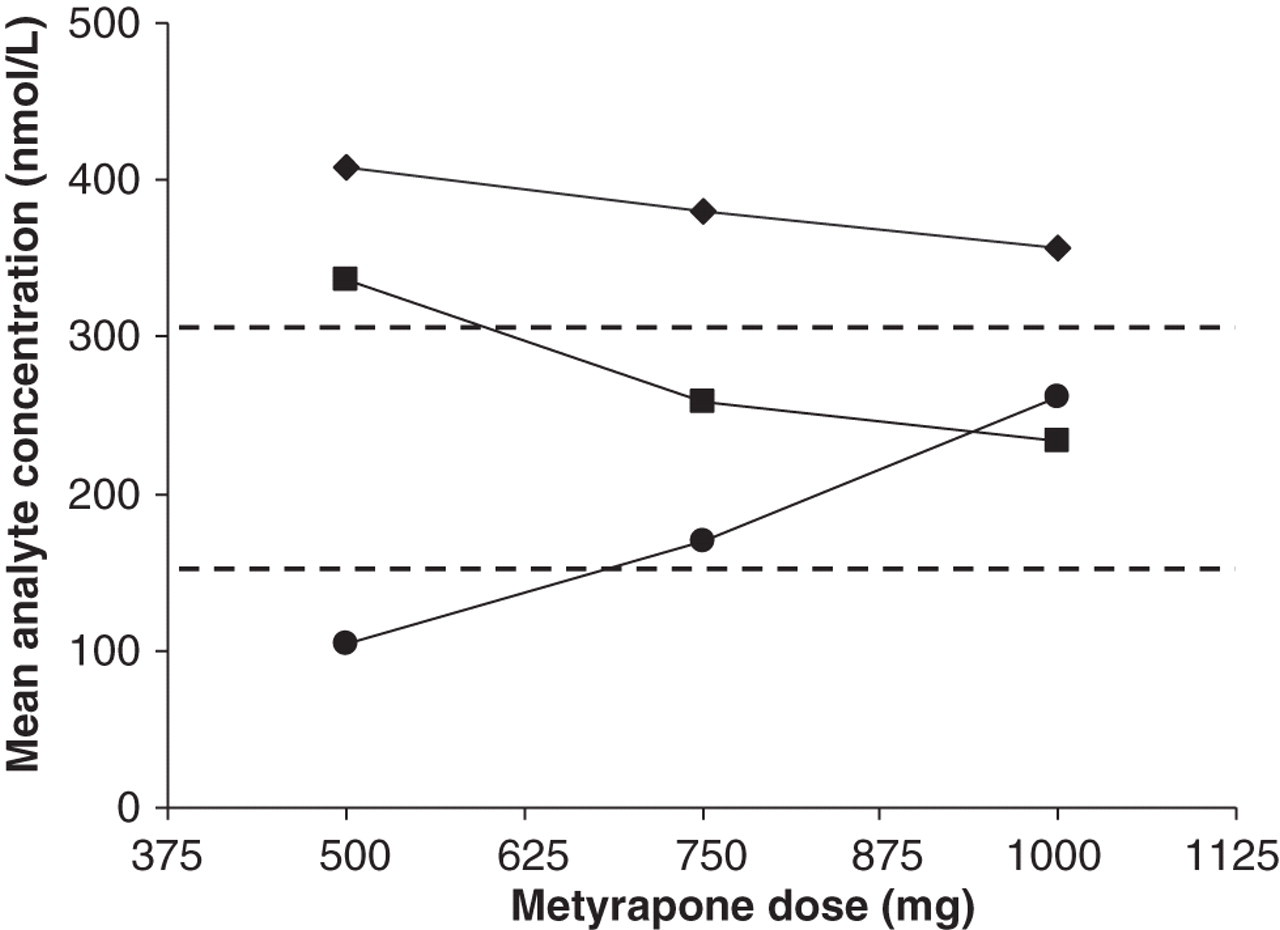
Graph showing the mean day curve concentration of serum cortisol and 11DOC for an individual patient as metyrapone dose is titrated from 500 to 1000 mg thrice daily. Symbols represent serum cortisol measured by the Centaur XP (♦), serum cortisol measured by LC-MS/MS (▪) and serum 11DOC measured by LC-MS/MS (•). Dashed lines represent the target range for mean day curve values of 150–300 nmol/L. LC-MS/MS, liquid chromatography-tandem mass spectrometry; 11DOC, 11-deoxycortisol
Discussion
In this study, we investigated interference from metyrapone treatment on immunoassay of serum cortisol and assessed if interference was dose-dependent. The contribution of 11DOC concentration to cross-reactivity was also investigated. Our results show that metyrapone therapy for the medical management of Cushing's syndrome does impact on the measurement of serum cortisol by immunoassay. Measurement of cortisol using the Centaur XP in patients receiving metyrapone showed a positive bias of 23% in comparison to LC-MS/MS. However, both the control group and Cushing's control group showed excellent agreement between the measurement methods, thus showing that altered adrenal steroid biosynthesis in Cushing's syndrome does not affect cortisol measurement by immunoassay. Secondly, the agreement between methods observed for the two control groups provided excellent comparators for which to interpret the metyrapone group data.
Previous studies comparing the measurement of cortisol by different methods have shown that pharmaceutical blocking of 11β-hydroxylase causes elevations of cross-reacting cortisol precursors resulting in positive interference in immunoassay. Early studies by Canalis et al. 6 showed that cortisol in patients after metyrapone administration when measured by LC and radioimmunoassay (RIA) showed discordance of up to 75%. A poor correlation was also observed for post-metyrapone cortisol concentrations between the two methods. More recently, cross-reactivity elicited by 17-hydroxyprogesterone and 11DOC in cortisol RIA was investigated, with almost half of patients receiving metyrapone showing a relative difference exceeding 40%. 12
These previous studies have alerted clinicians to the potential for cross-reactivity leading to spuriously elevated results when cortisol is measured by immunoassay; however, no study to date has employed LC-MS/MS as a standard measure for comparison of immunoassay results in patients receiving metyrapone. LC-MS/MS allows positive identification by mass-to-charge ratios (m/z) in contrast to immunoassay that lacks positive identification due to potential antibody interference. 13 The ability to measure serum cortisol and 11DOC by LC-MS/MS in patients receiving metyrapone has been previously demonstrated, but the results were not compared with immunoassay. 14 LC-MS/MS has been utilized in a recent method comparison study with immunoassay for serum cortisol measurement in patients during adenocorticotropic hormone (ACTH) stimulation tests, with the authors concluding that LC-MS/MS is the more robust method of analysis. 15
Our finding that 11DOC concentration positively correlates with delta cortisol does infer, albeit indirectly, that 11DOC cross-reacts with reagent antibodies in the Centaur XP immunoassay and corroborates previous findings from interference studies that have shown 11DOC cross-reactivity of 28–33.6%. 8,12 These previous studies assessed interference in the Roche immunoassay and in various RIA kit assays. Unfortunately, our data are limited to the Centaur XP, and due to differences in reagent antibody specificities, our observations cannot be extrapolated to other platforms. Furthermore, our observation of 23% bias is greater than the 11DOC cross-reactivity of 7.3% quoted by the immunoassay manufacturer. Therefore, structural analogues preceding cortisol in the steroid biosynthesis pathway additional to 11DOC, may have a cumulative effect on the observed cross-reactivity in cortisol immunoassays. The differences in degree of reagent antibody cross-reactivity towards these precursors between different immunoassays signifies that comparison studies should be performed for each individual immunoassay platform against LC-MS/MS to determine the level of interference caused by metyrapone. Laboratories with immunoassay platforms for which interference by metyrapone treatment is subsequently established from such comparison studies are strongly advised to refer these samples for LC-MS/MS measurement of serum cortisol.
The correlation in our data for metyrapone dose and mean delta cortisol was pronounced, with values of 22–133 nmol/L (12.6–90.8%) between 250 and 1000 mg thrice daily dose. However, the large variation in the data (Figure 3a) concludes that bias correction factors for metyrapone dose cannot be implemented for serum cortisol immunoassay results. The largest observed delta cortisol in this study was 248.7 nmol/L and given that the target range for mean day curve values during medical therapy is 150–300 nmol/L, there is a significant risk of unrecognized hypoadrenalism, which can result in symptoms, such as nausea and abdominal pain, that can be confused with side-effects of metyrapone or at worst be fatal.
The mean serum cortisol target range of 150–300 nmol/L was established in a previous study on a group of healthy volunteers by RIA methodology. 5 Our current study demonstrates excellent agreement for the measurement of serum cortisol between immunoassay and LC-MS/MS methodology in the control groups studied, further supporting the appropriateness of this target range.
A pertinent issue to note is that 90% of patients receiving metyrapone in this study had ACTH-driven Cushing's syndrome. This factor may augment the interference caused by metyrapone due to accumulation of precursor steroids driven by ACTH. Further studies to address the effects of metyrapone on immunoassay measurement in patients with primary hyperadrenalism are, therefore, warranted. Additionally, data concerning the relative glucocorticoid and mineralocorticoid potency of precursor steroids, including 11DOC, are currently limited. Future studies in this field, applying glucocorticoid and mineralocorticoid bioassays to precursor steroids, are therefore required.
In conclusion, this study provides the first quantitative comparison of serum cortisol in patients receiving metyrapone using LC-MS/MS as the reference method. LC-MS/MS has in recent years become an increasingly available technology in the clinical biochemistry laboratory and has an emerging role in the measurement of steroids. 16 Our data confirm that LC-MS/MS is the method of choice for accurate measurement of serum cortisol in patients receiving metyrapone therapy. This recommendation will mitigate the potential for erroneous clinical decisions concerning dose titration of metyrapone therapy.
DECLARATIONS
