Abstract
Adverse reactions to foods may arise by a variety of mechanisms, both immune (IgE and non-IgE) and non-immune mediated. This article considers those assays useful in the diagnosis of Type 1 hypersensitivity to foods (IgE-based) and, importantly, discusses those assays where evidence is lacking for their use. In all cases of suspected food allergy, a full clinical history is indispensable in facilitating diagnosis. Total serum IgE is not a suitable screen for food allergy. Suspect allergens may be confirmed by either skin prick testing or serological assays for specific IgE. Several studies suggest concentrations of food-specific IgE at which there is a high probability of reaction on food challenge. These cut-off levels are now being used by physicians to direct clinical advice. However, it is important to note that not all studies agree on these limits and the chosen cut-off is dependent on the population studied and the assay used.
Introduction: adverse reactions to foods
Adverse reactions to foods may arise by a variety of mechanisms, both immune (IgE and non-IgE) and non-immune mediated. 1 Classically, immune-mediated hypersensitivity reactions have been described using the Coombes and Gell nomenclature (Table 1). 2 Type 1, IgE-mediated reactions can be life-threatening (anaphylaxis) and may be triggered by exposure to tiny amounts of antigen (allergen). 3 The best example of non-IgE-mediated immune reactivity to food is coeliac disease (reviewed in the Annals). 4
The Coombes and Gell classification of immune hypersensitivity reactions
SLE, systemic lupus erythematosus
As noted by the Royal College of Physicians, 5 the terminology used to describe adverse reactions is confusing and there is a lack of consensus. In this discussion we will reserve the term ‘allergy’ to include only those reactions mediated by IgE in Type 1 or ‘immediate hypersensitivity’. Persons who are predisposed toward developing allergy are said to be atopic (from the Greek meaning ‘out of place’). This is a genetically transmitted (familial) state in which individuals produce increased amounts of IgE. The general public often confuse the terms ‘food allergy’ and ‘intolerance’. Once a person is sensitized (requiring prior exposure), ‘allergy’ to food involves a predictable, reproducible adverse reaction (even on exposure to trace quantities) seen on each and every occasion, typically within a few minutes of exposure (i.e. ‘immediate hypersensitivity’), involving antigen-specific IgE antibody (see later). ‘Intolerance’ (also termed ‘non-allergic food hypersensitivity’ 6 ), by contrast, does not involve any immunological response nor do adverse reactions necessarily occur on each exposure. 1,7 Lastly, variable quantities of food may trigger adverse reactions, so that small amounts of dietary exposure may continue without symptoms.
Examples of intolerance include those where definitive mechanisms are ill-understood (idiosyncratic, psychological, irritable bowel) or other more defined conditions (lactose intolerance due to enzyme deficiency, and high concentrations of sulphites or salicylate linked to exacerbation of asthma).
Provision of services for the investigation of allergy is limited and inadequate in the UK in 2010. There are few dedicated allergy services run entirely by trained allergists in the UK, 5 with many clinics being provided by immunologists 8,9 or other providers (e.g. dermatologists, otolaryngologists, chest physicians).
In all cases of suspected food allergy, a full clinical history (recording, e.g. time interval between food and symptoms) is indispensable in terms of facilitating diagnosis. 1,7
To illustrate the general approach to the investigation of hypersensitivity reactions to foods, consider an episode of vomiting and gastrointestinal discomfort several hours after consuming a fish meal at a restaurant. Because of the time delay, true (IgE mediated) fish allergy is unlikely. Immediate itchy mouth, perhaps with vomiting, collapse, urticarial rash, and or brochospasm would be expected.
One real possibility here is bacterial infection associated with poorly cooked shellfish. In this case we would expect adverse reactions to affect other party members who also ate the under-cooked food. Another explanation might be that the fish contained toxins causing non-immune-mediated adverse reactions. The toxin may either be intrinsic to the fish (e.g. puffer fish poisoning mediated by tetrodotoxin) or produced in the flesh after decay (scombroid poisoning where histamine is released from members of the Scombroidea family [which includes tuna and mackerel] by the action of bacteria).
In this article we will consider those assays useful in the diagnosis of Type 1 hypersensitivity to foods and, importantly, discuss those assays where evidence is lacking for their use.
Epidemiology of allergic disease in the UK
In the UK, over one-third of the population have been diagnosed with one or more of asthma, eczema or hay fever. 5 The incidence of asthma has increased progressively over the last 20 years. Visits to accident and emergency units for acute allergic events are increasing. The consensus view is that Type 1 hypersensitivity is increasing in the UK.
Specific data on food allergy and whether it is truly an increasing problem are harder to obtain. In the USA, the prevalence is up to 6% in children and 4% in adults. 10 In industrialized countries, more than 1% of children have peanut allergy. 11 In the UK prevalence of Type 1 peanut allergy, as with other forms of allergy, seems to be increasing. 12,13
Clinical history and the direction of testing
A good clinical history is key to understanding any hypersensitivity reaction to food. The most important elements are outlined in Table 2.
Overview of symptoms and clinical history associated with Type 1 (IgE mediated) hypersensitivity
*Note other symptoms of Type 1 allergy may also be manifest (rhinitis, asthma) as many patients also have aeroallergen sensitivity
†Failure to thrive may result from restricted diet as a result of parental or carers’ beliefs regarding the child's hypersensitivity 75
The gold standard in terms of proving a suspected allergy remains the double-blind, placebo-controlled food challenge. However, this carries significant risk to the patient and in most circumstances is impractical.
Instead, tests for allergen-specific IgE have been developed. They are designed to supplement, not replace, clinical history and suspected diagnosis. In routine clinical practice two strategies are used. One involves point of care skin prick testing (applying purified test allergen to patient's skin and looking for a local wheal and flare immediate cutaneous reaction); the other uses laboratory-based serological-specific IgE testing. The pros and cons of each of these approaches are summarized in Table 3.
Comparison of skin prick testing and serological testing for specific IgE
*Generally similar for the two modalities
†Author's experience (DJU)
Total IgE quantification
Measurement of total serum IgE is relatively straightforward, but of very little clinical utility in the context of suspected food allergy. Thus a patient with total IgE levels within the normal range may nonetheless have significant peanut allergy, and detectable peanut-specific IgE. The World Health Organization provides a reference for total serum IgE (international reference preparation 75/702). 14 The presence of a standard allows for accurate and precise measurement of serum IgE. There is no consensus in the literature as to what constitutes the adult reference range, which appears to vary by region. The adult reference interval has been quoted variously as <81 kU/L, 15 <114 kU/L, 16 <148 kU/L in female and <169 kU/L in male non-smokers (rising to 194 and 220 kU/L, respectively in smokers) 17 , <183 kU/L18and <150 kU/L. 19
Approximately a third of atopic adults will have a total serum IgE of <120 kU/L, i.e. within the reference interval for non-atopic individuals, as determined by most sources. While the atopic and non-atopic populations show considerable overlap and cannot be absolutely distinguished, it is true that for very high levels of IgE, clinical atopic (allergic) disease is more likely. 18 There is evidence to suggest that in infants (<2 years of age) a raised IgE is predictive of allergy in later life. 20 However, recent data show that total IgE in infants has a low positive predictive value and is a poor screen for atopy. 21
It has been suggested that total IgE may be used as a means of identifying patients with a low probability of the presence of specific IgE, 22 but we detract from this view. The negative predictive value of a normal IgE for atopy is unacceptably low.
Immediate hypersensitivity and skin prick testing
Skin testing is extremely safe, even in high-risk patients with a previous history of anaphylaxis. However it is labour intensive. Historically it is based on the observations of Küstner and Prausnitz. In 1921, Küstner (who had a known reproducible, immediate sensitivity to fish) worked with his colleague Prausnitz to demonstrate a passively transferable serum factor (later identified as IgE) which could transfer sensitivity. 23 Fish extract injected into Küstner's (but not Prausnitz's) skin provoked an immediate wheal and flare urticarial type reaction. Prior injection of Küstner's serum into Prausnitz's skin, however, rendered the latter sensitive to fish extract, but only when injected at the same site as the passively transferred serum.
These experiments paved the way for the diagnostic skin prick point of care testing widely used in clinics today 24 (Figure 1). Unfortunately, skin prick testing is not suitable for use in all patients (e.g. those with severe eczema or dermographism).
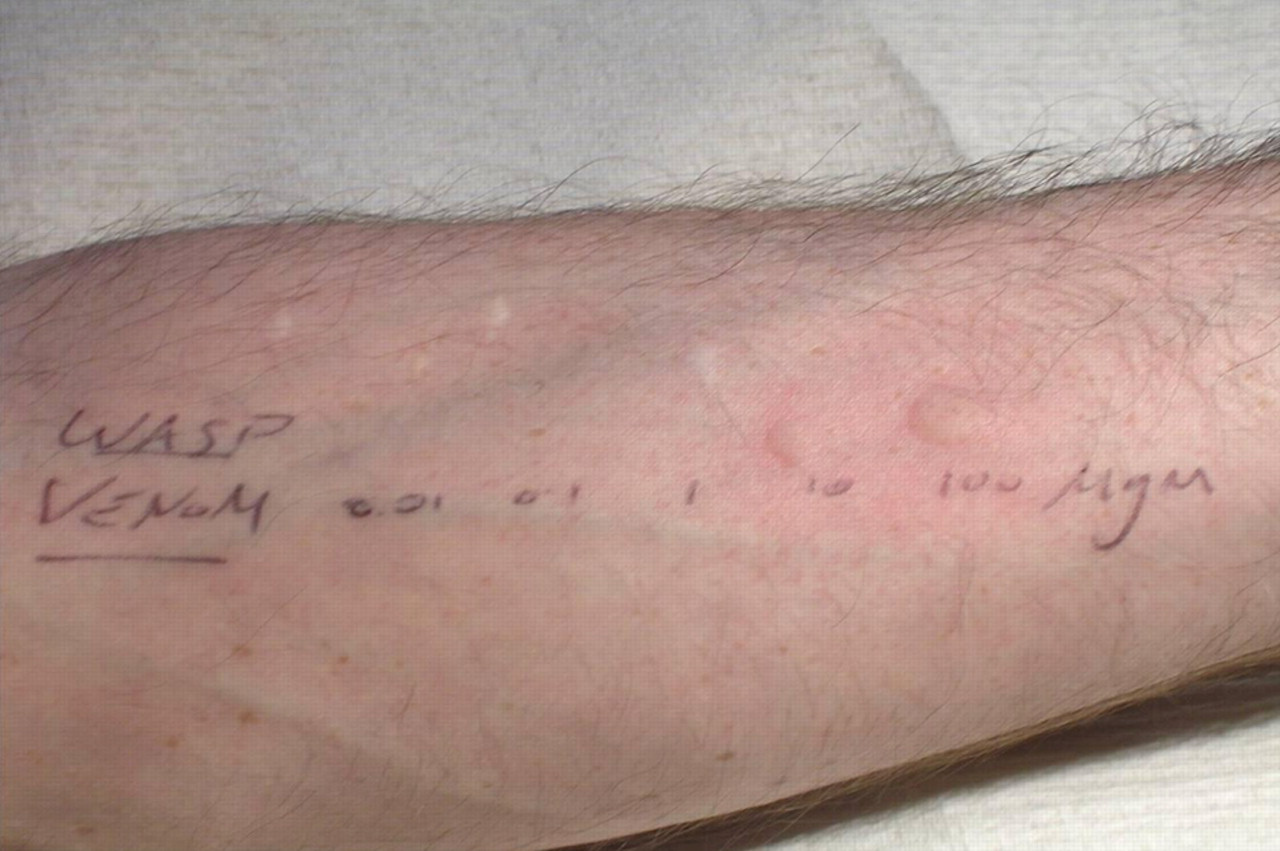
Example of skin prick testing. Wasp venom applied in increasing concentration, resulting in characteristic wheal and flare in response to Type 1 hypersensitivity reaction
In general terms, IgE-specific blood tests and skin prick tests (assuming use of the same or very similar allergen source) give similar results. 25 The highest concordance is achieved in those with allergy to inhalants. (Note that allergy to inhalants such as cat dander, or house dust mite tends to trigger nasal, eye, and respiratory symptoms, but not gastrointestinal symptoms.) However, in patients with suspected food allergy, concordance between skin prick and serological tests is much more variable. There are several reasons for this, not the least that the allergen sources are often very different. For example, one skin-testing assay for wheat tests for reactivity to gluten alone and not other wheat proteins (including, e.g. class 1 chitinases; plant stress proteins induced in response to fungal or bacterial infection). Similarly, there is variability in the source of allergens for serological testing. Allergens in a given formulation may be a mixture of purified allergens supplemented by recombinant proteins.
False-positive results can be a problem in both test modalities. Evidence of sensitization to allergen is not uncommon in subjects in whom there is no evidence of clinical allergy. This is why clinical history should direct confirmatory testing rather than vice versa. It has been demonstrated that false-positive results are more likely than false-negative results. For example, in the Isle of Wight population-based study, of 981 infants tested, 1.2% (15 cases) were test positive against nuts, but only half of these 15 cases were clinically sensitive to nuts. 12 These data must be balanced against other studies that indicate strongly positive reactivity is more indicative of clinically significant responses. There is evidence to suggest that in skin testing, wheal diameters greater than 7 or 8 mm are highly predictive of clinical allergy to peanut, cow's milk or egg allergy (>95%). 20,26–28 Similarly, data are presented later to show that high titre-specific IgE is predictive of significant allergic responses.
Elimination diets
There are two potential uses of elimination diets: diagnostic and therapeutic.
When considering the use of elimination diets for therapy, patients tend to fall into three categories:
Those with a good clinical history, with or without supportive data, either from skin prick testing or specific IgE tests. In this circumstance the identified food(s) should be avoided and eliminated from the diet; Those in whom the history is unconvincing with or without other supporting data. This is a contentious area but consideration could be given to a trial period of elimination. In this circumstance a patient diary of symptoms may be helpful; Those without a history consistent with food allergy and negative for specific IgE or skin prick tests. There is no support for introducing an elimination diet.
Elimination diets are not without risk and may impact on the nutritional balance and hence health of the patient. The residual diet must contain all essential components. For example, if dairy products are eliminated as a result of milk allergy it may be necessary to provide a calcium supplement. The clinician also needs to be wary of making diagnostic conclusions based on the use of dietary manipulation alone as there is a risk of being misled by the placebo effect. For this reason, it is also not advisable to introduce restrictions based solely on test results.
Antigen-specific IgE tests
Limited sensitivity and specificity data are available for peanut and a few other foods (discussed in section ‘Specific foods’ and in Table 4); equivalent reliable data are not, to our knowledge, available for the majority of specific IgE tests available.
The presence of an antibody indicates that the patient has been sensitized (generated an immune response resulting in the production of antibody) but does not guarantee that they will react to the allergen, which is dependent on the degranulation of mast cells. That is, the patient is sensitized but not sensitive to the allergen.
Furthermore, there are significant differences in performance between manufacturers’ kits purportedly reacting with the same antigen, e.g. peanut. 29 Clearly there are many potential differences between assay formulations; the antigens may be native, purified or recombinant and the balance of major and minor epitope expression may be different.
Given these very significant limitations, the authors are surprised that these tests are so widely and inexpertly used. The clinical question may either be to exclude disease (for which a high negative predictive value is needed) or to confirm the diagnosis (for which a high positive predictive value is required). Unfortunately, while a very wide range of test options are available, full evaluation is lacking for the majority.
General methodology
Tests for specific IgE to single or limited screens (e.g. mixed food screen for cow's milk, hen's egg, wheat, soya, cod and peanut) use immobilized allergens in one of four basic formats: paper disc, enzyme-linked immunosorbent assay (ELISA), cellulose matrix or fluid phase polymer. Of these, the paper disc (basis of the old RAST, radioallergosorbent test) and ELISA offer the least binding capacity and thus less antigen is available for binding specific IgE. As such they are more prone to false-negative results. The other formats have advantages in increased capacity for allergen binding which confers benefits of increased sensitivity, the expression of minor epitopes and of alternative configuration of binding.
The use of allergen mixes, such as those described above, confers a small risk of increasing the number of false-negatives. This arises as a result of the finite capacity of the matrix used to bind the allergen. Five allergens in one analysis will of necessity have one fifth of the allergen as a single analyte. In practice this means that some weak reactions will be missed. The clinical importance of this is uncertain. It is argued that, in particular, a false-negative result for peanut could be potentially fatal. However, the authors’ view is that false-negatives occur in all systems and the history will determine the interpretation of the test in any individual.
A further development of the high-capacity assays is the use of individual proteins as allergen sources (component resolved diagnostics), as compared with earlier formats which used a mixture of proteins. The sub-specificity analysis this allows may be important in determining which sensitized patients will be allergic and which tolerant to a particular food. 30
Recently, yet another format, microarrays, has been introduced. 31,32 Microarrays are configured to test for reactivity to many individual proteins simultaneously. The concern here is that the testing is not directed by the history; 90–100 allergens may be tested simultaneously, with the consequent increased chance of false-positivity. While clearly a valuable research tool, more studies are required to determine the value of this approach in routine diagnostics.
Standardization
There is no international standard for specific IgE assays. The original assays and some of those currently in use, employ functional ‘standards’. That is, a single donor serum to a specific allergen (e.g. birch pollen) that has a designated arbitrary value. Use is dependent on the assumption that the ‘standard’ binding to its corresponding antigen behaves in an identical manner to patient serum. This is an oversimplification as different sera will have different avidities and affinities for binding a range of allergens.
More recently, assays for specific IgE calibrate to 75/702 in mass equivalent terms. The international standard was assigned arbitrary mass units (international units, IU). It was later shown that one IU corresponded to 2.42 ng of pure IgE. The assays are configured such that the allergen-bound IgE in the system equates to the total IgE bound in the calibration curve (where IgE is captured by anti-IgE rather than allergen). Although this relationship has been established for some allergens, it has not been unequivocally shown to apply to all allergens; hence the use of allergen-specific units (UA). In serum this extrapolates to the use of kUA/L. These assays have been configured to equate to earlier functional ‘standards’ to facilitate ease of interpretation. Clearly the two approaches are not identical. The measurement of specific IgE requires that the functional part of the molecule (the binding site) reacts with immobilized antigen. This is dependent on the affinity and avidity of binding, which will vary from patient to patient. The resultant reactivity is considerably less predictable than when measuring total IgE.
Screening kits
In order to improve the clinical utility of screening, several multi-allergen assays have been formulated (e.g. Phadiatop®, Phadia Ltd, Milton Keynes, UK). These are based on the knowledge that 95–99% of patients with allergy will respond to one or more of the 15 most common allergens. However, as these are not cheap and further testing for individual allergens is required, they have not become popular in UK laboratories.
Point of care tests (POCTs) for food allergy are available from some supermarkets, high street shops and by post, but of variable quality. The Royal College of Pathologists 33 is emphatic in its advice: ‘These are not recommended’. This is reiterated by a joint statement from the Royal College of Physicians’ and Royal College of Pathologists’ Working Party: ‘Some “point of care” allergy tests are inappropriate’. 9 Concern was particularly expressed over the possibility of misinterpretation of results performed in unskilled POCT settings without evidence of appropriate quality assurance.
One POCT system has been approved by the US Food and Drug Administration for a panel of 10 inhalant allergens (ImmunoCAP Rapid System, Phadia US Inc, Portage, MI, USA). None have yet been approved for food allergy.
Specific foods
In children and adolescents, 85% of all food allergies are accounted for by three allergens: cow's milk, hen's egg and peanut. 34 These are discussed in detail below. Correspondence between different manufacturers’ assays for specific IgE to food allergens is modest. For example, Moneret-Vautrin et al. 35 found only 80% agreement between two leading manufacturers. Furthermore, sensitivity and specificity for the diagnosis of food allergy varies between platforms. 36 Table 4 gives examples of the variability of sensitivity and specificity of specific IgE assays for some common food allergens, as documented. Sensitivity is based on a positive assay in patients who develop unequivocal features of Type 1 hypersensitivity (e.g. rash, abdominal pain) on food challenge. The specificity is determined from those in whom there are no features of Type 1 hypersensitivity on administration of placebo.
Examples of sensitivity and specificity of IgE assays, as reported by different centres, for some common food allergies
DPC, Diagnostic Products Corporation; RAST, radioallergosorbent test
Sensitivity is based on a positive assay in patients who develop unequivocal features of Type 1 hypersensitivity (e.g. rash, abdominal pain) on food challenge. Specificity is determined from those in whom there are no features of Type 1 hypersensitivity on administration of placebo
DPC and Bayer now both Siemens Healthcare Diagnostics, Deerfield, IL, USA
Peanut
In the UK, up to 3.2% of children are sensitized (i.e. have specific IgE) to peanut or tree nuts. 37 Peanut is the most common cause of fatal or near fatal reactions to food. It should be noted, however, that fatal reactions are fortunately extremely rare, with an incidence of around 1 case per 10 million population annually. 38–40 In children, high levels of specific IgE to peanut (>15 kUA/L) have a high predictive value for clinical allergy to peanut (92% positive predictive value, 95% confidence interval 74–99%). 26 These data confirmed the earlier work by Sampson and Ho 41 , where clinical reactivity was predicted with >95% certainty when specific IgE was >15 kUA/L. It is unusual for patients to outgrow peanut allergy but it does happen in a minority of cases, as shown by challenge testing. 42 Interestingly, specific IgE to peanut remains detectable in most of these cases.
Hen's egg white
Similarly to the situation with peanut allergy, in children, predictive levels for clinical reactivity to hen's egg white can be defined but not all sources are in agreement and these vary significantly according to study. Celik-Bilgili et al. 43 found the decision point for 95% probability of positive food challenge to be 12.6 kUA/L. Sampson and Ho 41 found clinical reactivity was predicted with >95% certainty when specific IgE was >6 kUA/L. The commercial source of the assay also has a major bearing on the interpretation. For example, Osterballe and Bindslev-Jensen 44 suggested the diagnostic level with 95% certainty is 10.8 units/mL using Magic Lite (ALK-Abelló, Hørsholm, Denmark) and 1.5 kUA/L using CAP (Pharmacia, Uppsala, Sweden).
Note that these data refer to reactivity with commercial allergen preparations to hen's egg white. Egg white contains several allergenic proteins including ovalbumin, conalbumin (or ovotransferrin) and ovomucoid. Approximately two-thirds of egg-allergic children become tolerant by age five years. Recent studies have shown that those who remain intolerant have specific IgE antibodies to linear epitopes on ovomucoid, whereas those who become tolerant do not. 45
Cow's milk
For cow's milk allergy the data are again mixed. Sampson and Ho 41 found clinical reactivity was predicted with >95% certainty when specific IgE was >32 kUA/L. Garcia-Ara et al. 46 found age-dependent cut-offs of 1.5, 6 and 14 kUA/L gave 90% positive predictive values for clinical reactivity at 13–18, 19–24 and 25–36 months, respectively.
It has been suggested that specific IgE may be more useful in prognosis than diagnosis in infants. Infants who are intolerant of milk protein but specific IgE negative are more likely to become tolerant and at a younger age than those who are specific IgE positive. 47 Similarly, data suggest that lower initial specific IgE concentrations in infants <4 years give a higher probability of developing tolerance in cow's milk and hen's egg allergy. 48 However, persistence of specific IgE in those that are positive is not a good indicator of whether the intolerance will persist or not. 38
Predictive value of tests
Previous work 41,49,36 has suggested concentrations of specific IgE to various common foods at which there is a high probability of reaction to food challenge. These cut-off levels are being used by physicians to direct clinical advice. However, it is important to note that not all studies agree on these limits and the chosen cut-off is dependent on the population studied and the assay used (see Table 5 for egg, milk, peanut and fish). Similar data for other allergens are scarce. Two examples (for hazelnut and celery) suggest that specific IgE can have a high positive predictive value for food allergy at a specific IgE of ≥0.7 kUA/L (92% and 96%, respectively) 50,51 but these studies are based on highly selected populations and need to be confirmed by others.
Quoted values (kUA/L) for a 95% probability of clinical reaction on challenge for some common foods
Also of importance it should be noted that persistence of specific IgE to a food (which may be years, even lifelong) does not predict whether a child will or will not become tolerant after a period of time. 38
Cross-reactivity
A confounder in the interpretation of specific IgE results is the recognition of cross-reacting antibodies. These fall into two major categories. Some antibodies recognize similar epitopes in proteins from different species (‘panallergens’) and may elicit an allergic response. Examples include the major birch pollen protein Bet v 1 (after the Latin for birch Betula verrucosa) and its homologues (e.g. Mal d 1 from Malus x domesticus [apple], Api g1 from Apium graveolens [celery]) or profilins found in many plants (including apple and peach). Invertebrate tropomyosin is another example of a panallergen that may provoke an allergic response to several species being found in crustaceans (e.g. shrimp, crab, crayfish), arachnids (e.g. house dust mite) and molluscs (e.g. oyster, scallop). Others may cross-react in vitro but often fail to elicit an allergic response (e.g. cross-reacting carbohydrate determinants are found in a variety of organisms including peanut but clinical evidence of cross-reactivity is unusual 52,53 ). Again, patients may show reactivity to grasses and grains where cross-sensitization of allergens does not necessarily mean clinical sensitivity to the cross-reactive allergens. 54
Examples of cross-reactions include the oral allergy or pollen/fruit syndrome, latex allergy and some food allergies (Table 6).
Common cross-reactive allergen groups
Of note, specific IgE tests are often negative in the birch pollen (Bet v 1) related oral allergy syndrome (the most frequent form in northern Europe) as the antigens are highly labile and denatured during manufacturing. As a result patients often react to fresh but not cooked fruits. Interestingly, in a Mediterranean variant related to grass or olive pollen and peach sensitivity, specific IgE to peach is often positive. The relevant proteins here appear to be related to Bet v 2 (a profilin). 55
Cross-reactions should not be confused with parallel co-sensitization. This occurs for example in the 30% of peanut allergic patients who develop sensitivity to tree nuts. The two sensitization events are usually separate although some cross-reactions are described.
Anaphylaxis investigation – mast cell tryptase
Where patients present acutely with a severe allergic reaction/anaphylaxis, support for an IgE-mediated mechanism may come from using mast cell tryptase (MCT) to check for mast cell degranulation.
MCT is released into the blood stream after an anaphylactic reaction, reaching peak levels 1–3 h postchallenge and decreasing rapidly thereafter. Concentrations are usually normal after 24 h. The useful analysis window is therefore quite short and the temporal relationship between time of reaction and time the sample is taken is crucial in the interpretation of the result, but when available the assay result may be extremely helpful. 56
Note that specific IgE tests are best deferred until convalescence (>6 weeks post the event), because in the acute setting allergen may consume specific IgE in serum, resulting in a transiently negative serum test.
Tests that are not recommended and/or require further evaluation
IgG class antifood antibodies
There is a large literature on the possible (but unproven) utility of IgG antifood antibody testing. With the single exception of coeliac disease, such tests historically were of no proven value. There are a few coeliac patients who will be positive for IgG antigliadin and negative for IgA antigliadin and hence doing both improves the sensitivity of screening. Nevertheless, IgA antigliadin antibody is more specific than IgG antigliadin in this setting. However, as a general rule, IgG class antifood antibody has no proven value, certainly in the context of Type 1 food hypersensitivity (by definition IgE mediated) nor in ‘food intolerance’ cases.
With IgG food antibody, poor specificity is a major issue. Depending on the assay cut-off, IgG antigliadin is found in up to 15% of healthy blood donors. 57 There is a danger that the presence of these antibodies might therefore be over-interpreted since the co-existence of antibodies and symptoms does not necessarily imply a cause and effect. For example, there is the observation that IgG antigliadin antibodies are found in some patients with ataxia. While some have interpreted this as a new disease ‘gluten ataxia’, 58 others have seen this as no more than a result of the common finding of IgG antigliadin (and other food antibodies) in the background population. 59,60
A recent publication focusing on irritable bowel syndrome has once again brought IgG class antibodies into attention. 61 The authors suggest that these may play a role in food hypersensitivity. It should be remembered, as noted above, that the specificity of such assays is necessarily very low. There is a high prevalence of such antibodies in the normal population 62,63 and hence the positive predictive value is very low. In summary, the authors cannot recommend IgG-based tests in patients suspected of food allergy/intolerance.
Specific IgG4 assays
For many years it has been known that several months after starting subcutaneous allergy desensitization regimens (e.g. for pollen allergy), levels of allergen-specific IgG4 increase during successful treatment. This has been attributed to ‘blocking antibodies’ which interfere with the binding of allergen to specific IgE. Recently, it has been shown that high-dose sublingual regimens, where the allergen is introduced under the tongue, may also induce blocking antibodies. 64 Hence, in the limited context of laboratory support for clinics involved in allergen desensitization, a case may be made for checking allergen-specific IgG4.
These comments, however, are largely irrelevant in the context of food allergy where desensitization treatments are in their infancy. 65 Specific IgG4 has been shown to be elevated in some patients with atopic dermatitis in several studies, but clinical utility is very limited. Specific IgG4 has not otherwise been shown to be helpful in either diagnosis or monitoring of therapy. 52,66–69
Basophil activation and leukotriene release assays
Basophils may be activated in vitro by allergen-specific IgE leading to expression of CD63 and release of leukotriene C4. Assays have been developed to measure these and have been demonstrated to correlate with clinically proven food allergy. 70 The sensitivity of the basophil activation test is in the order of 60–80%. 70,71,39 The sensitivity of the leukotriene release assay is in the order of 65%. 71 There is also some evidence to suggest the leukotriene release assay may also detect activation through non-immune mechanisms. 72 However, both are time-consuming assays, requiring specialist facilities (e.g. flow cytometry), and have not yet found general utility in the routine investigation of food hypersensitivity.
Other (less well understood) tests
There are other tests advocated for allergy investigation but these have, at best, a dubious scientific basis. These include applied kinesiology, the leucocytotoxic test, electrodermal testing and hair analysis. A recent review concluded that ‘to date, no complementary or alternative diagnostic procedure can be recommended as a meaningful element in the diagnostic work-up of allergic diseases’. 73
Recently, multiplex variants for specific IgE analysis have become available that allow the testing for most common allergens in a single analysis. These are becoming increasingly available on the high street, often without proper clinical advice or interpretation. Indiscriminate testing of this nature has insufficient clinical data to justify its place in the armamentarium. Sensitization and sensitivity are not the same things. It is important to remember that false-positive tests are common in patients sensitized to pollens or latex and in those with atopic eczema.
Conclusion
In all cases of suspected food allergy, a full clinical history is indispensable in terms of facilitating diagnosis. Total serum IgE is not suitable as a screen to determine further testing for food allergy. In general terms, IgE-specific blood tests and skin prick tests give similar results. The choice of which to use is driven more by availability (there is a limited repertoire for skin testing), required turnaround time (15 min for skin testing), availability of trained staff for skin testing, and other issues including sensitivity or specificity. Several studies suggest concentrations of specific IgE to various common foods at which there is a high probability of reaction to food challenge. These cut-off levels are being used by physicians to direct clinical advice. However, it is important to note that not all studies agree on these limits and the chosen cut-off is dependent on the population studied and the assay used.
Other assays remain either unproven (e.g. specific IgG to foods) or largely restricted to research laboratories (basophile activation and leukotriene release assays).
DECLARATIONS
