Abstract
Background
Dexamethasone is a synthetic glucocorticoid and is analogous to cortisol. It is used in the low-dose overnight dexamethasone suppression test (LDODST) to diagnose hypercortisolism in patients suspected to be suffering from Cushing's syndrome (CS). Measuring plasma dexamethasone in conjunction with measuring the amount of cortisol following the LDODST may allow clinicians to improve the diagnosis of CS.
Methods
Plasma samples were cleaned up by solid-phase extraction before analysis. Liquid chromatographic separation was carried out under reversed-phase conditions prior to detection by tandem mass spectrometry. The analytes were determined in the presence of deuterated internal standards cortisol-d4 and dexamethasone-d4.
Results
Limit of quantitation (LOQ) was 1.89 nmol/L for dexamethasone and <0.02 μmol/L for cortisol. Recoveries of both analytes ranged from 80.2% to 114.4%. Intra- and interassay coefficients of variation were <15%. The concentration of dexamethasone and cortisol was determined in 62 patients after performing LDODST. Dexamethasone concentrations ranged from 3.0 to 21.5 nmol/L (median 7.4 nmol/L) for 57 of these samples. For five patients the concentration was <LOQ. Cortisol concentrations were <0.08 μmol/L (median 0.037 μmol/L, n = 54) except for eight patients (>0.22 μmol/L).
Conclusions
A method for the simultaneous measurement of dexamethasone and cortisol in human plasma by liquid chromatography/tandem mass spectrometry has been developed and validated. The method is suitable for controlling the compliance to the LDODST and for determining the cortisol plasma concentration after the test. The interpretation of LDODSTs was improved by the simultaneous determination of both analytes.
Introduction
Dexamethasone is a synthetic glucocorticoid that is found to be effective in many inflammatory and autoimmune conditions (e.g. rheumatoid arthritis, multiple sclerosis and allergies). 1,2 Additionally, it is administered in the low-dose overnight dexamethasone suppression test (LDODST). Cortisol is a corticosteroid that is produced by the adrenal cortex following the hypothalamic–pituitary–adrenal axis (HPA axis). Normally, a high cortisol concentration inhibits cortisol production. This biosynthetic pathway is disturbed in patients with endogenous Cushing's syndrome (CS) by excess adrenocorticotropic hormone secretion or by independent adrenal overproduction of cortisol. The LDODST is used in the diagnosis of CS. In this test, a supraphysiological dose of dexamethasone (1 mg) is administered to the patient at 23:00. The next morning, at 8:00, a blood sample is drawn for cortisol measurement. Because of dexamethasone's property to suppress the HPA axis in the same way as cortisol does, this test indicates if the HPA axis functions appropriately. A cortisol concentration <0.050 μmol/L is characteristic for healthy individuals. 3 We can achieve a diagnosis of CS if we apply a cut-off of the sensitivity >95% and a specificity of 80%. 4
Until now, the LDODST has been monitored by routine immunological measurement of plasma cortisol. A cortisol concentration >0.050 μmol/L is not only characteristic for CS but may also be observed in pseudo-CS. Pseudo-CS may be a result of stress or lack of drug compliance. 5 These false-positive results often lead to unnecessary and costly follow-up examinations, such as magnetic resonance imaging.
In addition, considerable variation has been described in the plasma concentrations of dexamethasone following oral doses. 6–8 Morris et al. 7 found that a fixed oral dose of dexamethasone did not produce a constant plasma dexamethasone concentration and that low-degree suppression of cortisol concentrations was associated with low dexamethasone concentrations and vice versa. Several factors are responsible for this variation, including intestinal uptake, inactivation by conversion by CYP3A4 in the liver and renal clearance. Moreover, several drugs have been described to either reduce or accelerate CYP3A4 activity, thereby affecting blood dexamethasone concentrations. Therefore, simultaneous quantification of plasma cortisol and dexamethasone would present more reliable results. As a low dose (1 mg) of dexamethasone is taken in the suppression test, a highly sensitive and accurate test is required. Meikle 6 described a radioimmunoassay (RIA) for the analysis of both components. Because of the cross-reactivity with possible metabolites of dexamethasone, RIAs require a highly selective antibody for accurate quantification of dexamethasone, which is a serious drawback of using RIA methods. 9 Due to the highly selective mass transition pattern for the analytes and simple sample clean-up, liquid chromatography/tandem mass spectrometry (LC-MS/MS) techniques have improved selectivity, analysis speed and throughput. Several methods for the analysis of dexamethasone with LC-MS/MS have been developed. 9 The study conducted by Frerichs and Toernatore 10 allowed for the concomitant analysis of cortisol.
We have developed and validated a rapid and reliable LC-MS/MS method for the simultaneous quantification of dexamethasone and cortisol in plasma. To the best of our knowledge, it is the first time that an LC-MS/MS method has been applied for controlling the LDODST.
Materials and methods
Chemicals
Dexamethasone (99.9%) was purchased from Spruyt Hillen (IJsselstein, The Netherlands). Cortisol (98%) was obtained from Sigma-Aldrich (Zwijndrecht, The Netherlands). The internal standard (IS) dexamethasone-d4 was purchased from CDN Isotopes (Quebec, Canada) and cortisol-d4 from Cambridge Isotope Laboratories (Andover, MA, USA). Methanol, acetonitrile and water (all liquid chromatography-mass spectrometry [LC-MS] grade) were obtained from Biosolve (Valkenswaard, The Netherlands). Acetone (high-performance liquid chromatography [HPLC] grade) was purchased from Lab-Scan (Gliwice, Poland) and formic acid from Fluka (St Louis, MO, USA).
LC-MS measurements
Multiple reaction monitoring (MRM) transitions and optimized compound-dependent parameters of dexamethasone, cortisol and the respective deuterated internal standards dexamethasone-d4 and cortisol-d4
DP, declustering potential; EP, entrance potential; CEP, collision cell entrance potential; CE, collision energy; CXP, collision cell exit potential
Standards, ISs and quality controls
Stock solutions of dexamethasone (2.39 mmol/L), dexamethasone-d4 (897.9 μmol/L), cortisol (2.72 mmol/L) and cortisol-d4 (2.78 mmol/L) were prepared in LC-MS-grade methanol. The stock IS solutions were mixed and diluted with methanol to a concentration of 198 nmol/L for dexamethasone-d4 and 3 μmol/L for cortisol-d4. Different volumes of the cortisol and dexamethasone stock solutions were mixed and diluted with pooled plasma so that a 5-point calibration curve was obtained (standards 1–5). The dexamethasone concentrations obtained were 3, 6, 10, 30 and 60 nmol/L. For calibration of cortisol standard, addition has to be carried out as cortisol is an endogenous plasma compound. The cortisol concentrations added were 0.06, 0.12, 0.20, 0.60 and 1.20 μmol/L. Quality controls (QCs) low and high were also prepared from the dexamethasone–cortisol mixture in pooled plasma and measured within every run. All stock standard solutions, standards, ISs and QCs were stored as aliquots at −20°C.
LDODST
A 1 mg-dexamethasone tablet was orally administered to the patient at 23:00. The next morning at 8:00, a blood sample was drawn from the patient.
Sample preparation
A total of 150 μL of IS solution was added to 1 mL of plasma sample, standard or QC sample. The solution was deproteinized with 3 mL acetonitrile while vortexing. After centrifugation (20 min, 18°C, 3000 rpm), the supernatant was evaporated at 58°C under a constant flow of nitrogen. The components were resolved in 1 mL methanol–water (1:3) by vortexing. The solution was further purified by solid-phase extraction (SPE) using Polarplus Octadecyl C18 columns from JT Baker (Deventer, The Netherlands). The sample was transferred to the conditioned SPE columns, washed with 2 mL water and eluted with 3 mL acetone. The eluate was evaporated, resolved with 100 μL methanol–water (1:3) and transferred to HPLC vials.
Assay performance
Precision
Precision was determined by analysing QC high and low within-runs, between-runs and between-days 10 times during a period of four weeks. The QCs were prepared twice per day and measured in two-fold (two runs per day, two repetitions per run). The data evaluation has been carried out with the statistical program EP Evaluator 9 (Data Innovations, Brussels, Belgium).
Linearity
The linearity of the method was determined by mixing two standards with high and low concentrations of dexamethasone and cortisol in pooled plasma in the ratios 4:0, 3:1, 1:1, 1:3 and 0:4. The five samples were prepared and measured twice.
Limit of quantitation
In order to determine the limits of quantitation (LOQs), pooled plasma spiked with dexamethasone and cortisol to a concentration of 10 nmol/L and 0.20 μmol/L, respectively, was diluted with water by factors 2, 5, 10 and 20. The samples were prepared and measured on five different days. The data were evaluated with EP Evaluator 9.
Recovery
Recoveries were determined by spiking QCs low and high with cortisol/dexamethasone mixtures of two different known concentrations. To QC low, cortisol/dexamethasone mixtures of 0.06 μmol/L/3 nmol/L and, respectively, 0.2 μmol/L/10 nmol/L were added. QC high was spiked with cortisol/dexamethasone mixtures of 0.2 μmol/L/10 nmol/L and, respectively, 0.6 μmol/L/30 nmol/L. The samples were prepared and measured twice and compared with unspiked QCs.
Method comparison
For quantitative comparison, the results of the LC-MS determination of cortisol in 45 patients' samples post-LDODST were compared with a routinely applied electro-chemiluminescence immunoassay (ECLIA) from Roche Diagnostics (Almere, The Netherlands). For dexamethasone, no method comparison was possible.
Patients' samples
Sixty-two samples post-LDODST were analysed for both dexamethasone and cortisol. Forty-six of these samples were derived from patients suspected to have CS and 16 from healthy controls. Dexamethasone and cortisol concentrations in these samples were obtained by means of LC-MS/MS.
Results
The aim of this study was to develop an LC-MS/MS method for simultaneous determination and quantification of dexamethasone and cortisol in human blood plasma for the evaluation of LDODSTs. As dexamethasone concentrations in plasma could be expected in the low nanomolar range, 2,11 the method had to be very sensitive. For sample preparation, several SPE and liquid–liquid extraction (LLE) procedures have been tested. Samples have been deproteinized with perchloric acid, trichloroacetic acid or acetonitrile and, after further SPE purification on Polarplus Octadecyl C18 columns from JT Baker, the results have been compared. LLE using a diethyl ether/dichloromethane mixture has also been investigated. However, the preparation procedure described above yielded the highest peak intensities and the best peak shapes (data not shown).
Applying the above-described HPLC conditions, dexamethasone and cortisol were baseline separated. The two analytes co-eluted with their ISs dexamethasone-d4 and cortisol-d4 at retention times of 5.8 and 5.2 min, respectively. A total ion chromatogram of QC high is shown in Figure 1.
Total ion chromatogram of quality control high (dexamethasone: 28.1 nmol/L; cortisol: 0.84 μmol/L)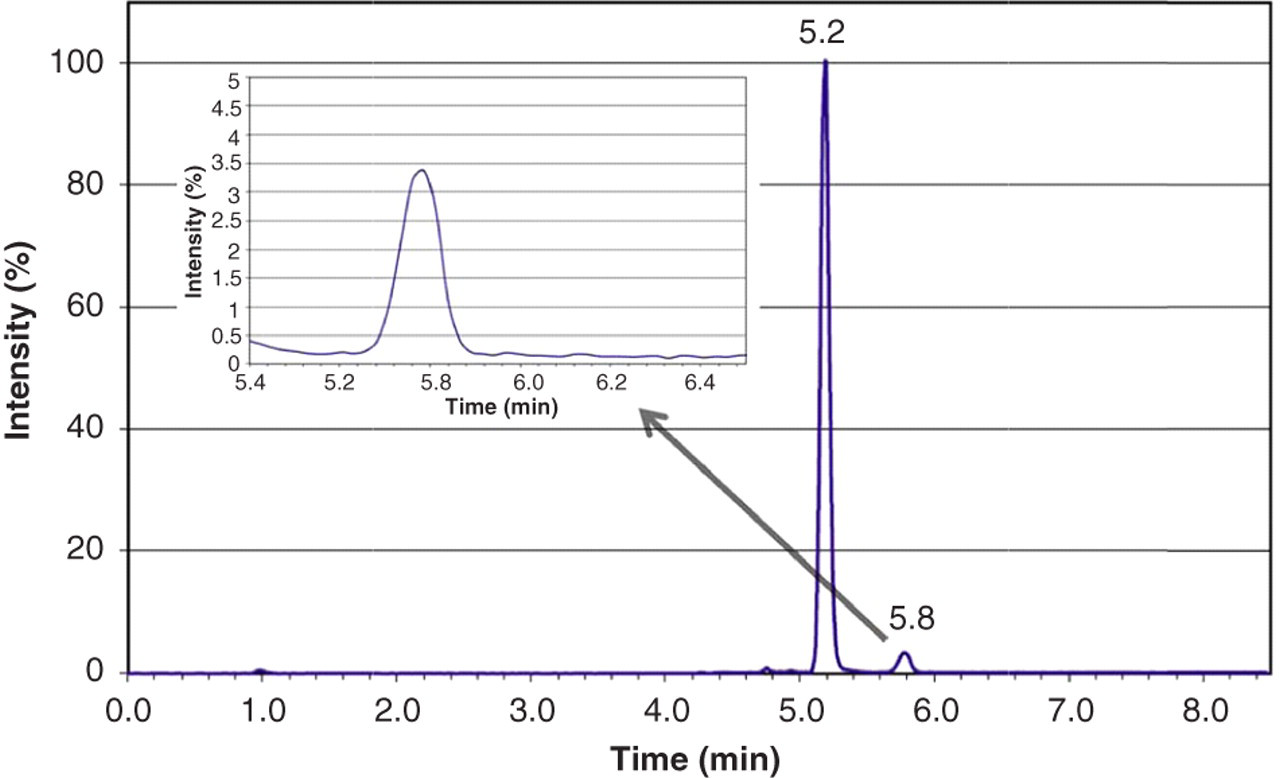
For identification and quantification, several MRM transitions for dexamethasone and cortisol were monitored (Table 1). In the case of dexamethasone, the peak areas of MRM transitions m/z 393.2 → m/z 147.1, m/z 393.2 → m/z 91.0, m/z 393.2 → m/z 77.0 and m/z 393.2 → m/z 121.2 were summed up to increase the signal intensity.
The stability of QCs low and high after sample preparation has been investigated (data not shown). The measured concentrations of samples kept at 4°C for 14, 19 and 27 d have been compared with the results determined prior storage. Although no significant difference was observed, it was found for dexamethasone that noise increases with storage time. After 14 d, an additional peak with a retention time of 5.4 min was detected in the dexamethasone trace. For cortisol, cortisol-d4 and dexamethasone-d4, no significant changes could be observed in the chromatograms.
Within-run (two repetitions per run), between-run (two runs per day) and between-day (n = 10) precision of dexamethasone and cortisol for QCs low and high in pooled plasma
QC, quality control; CV, coefficient of variation
*nmol/L for dexamethasone and μmol/L for cortisol
The linear range of the method was determined by plotting the calculated concentrations of dexamethasone and cortisol, against the measured concentrations in the mixed standards. The plots resulted in a slope of 0.8747, an intercept of 1.1137 and r 2 of 0.9978 for dexamethasone and a slope of 0.8128, an intercept of 0.0726 and r 2 of 0.9996 for cortisol. The experiment illustrated that the determination of dexamethasone is linear up to 100 nmol/L, and the determination of cortisol up to 2.2 μmol/L.
Investigations regarding the limits of quantitation (LOQs) of dexamethasone and cortisol
CV, coefficient of variation
For dexamethasone, a LOQ of 1.89 nmol/L (CV 12.5%) was calculated by means of EP Evaluator 9. The ultimate achievable LOQ for cortisol is <0.02 μmol/L
Recoveries of dexamethasone and cortisol determined in pooled plasma
QC, quality control
*nmol/L for dexamethasone and μmol/L for cortisol
Cortisol concentrations measured by ECLIA were compared with those measured by LC-MS/MS by means of a Passing and Bablok regression. A regression line could be fitted to 27 patients' samples. The rest of the 45 samples could not be included in this comparison since the ECLIA results were <LOQ. The intercept of the regression line was 0.00 (95% confidence interval [CI]: −0.02 to 0.01) and the slope was 1.07 (95% CI: 0.93 to 1.30). The bias was 0.0010 μmol/L (95% CI: −0.0206 to 0.0124 μmol/L) at a concentration level of 0.022 μmol/L, at a concentration level of QC low (0.29 μmol/L) 0.0185 μmol/L (95% CI: −0.0289 to 0.0875 μmol/L) and at a concentration level of QC high (0.84 μmol/L) 0.0545 μmol/L (95% CI: −0.0676 to 0.2633 μmol/L). The data showed a good correlation for measured cortisol values for the two different techniques. Since there was no commercial assay available for the determination of dexamethasone in plasma, no method comparison could be carried out for this analyte.
Both dexamethasone and cortisol concentrations in 62 patients' samples derived after a LDODST were analysed. The dexamethasone concentration ranged from 3.0 to 21.5 nmol/L (median 7.4 nmol/L) for 57 of these samples. For five patients, the concentration was <LOQ. Cortisol concentrations were <0.08 μmol/L (median 0.037 μmol/L, n = 54) except for eight patients (>0.22 μmol/L). The results are depicted in Figure 2.
Cortisol and dexamethasone concentrations of all 62 patients' samples measured by liquid chromatography/tandem mass spectrometry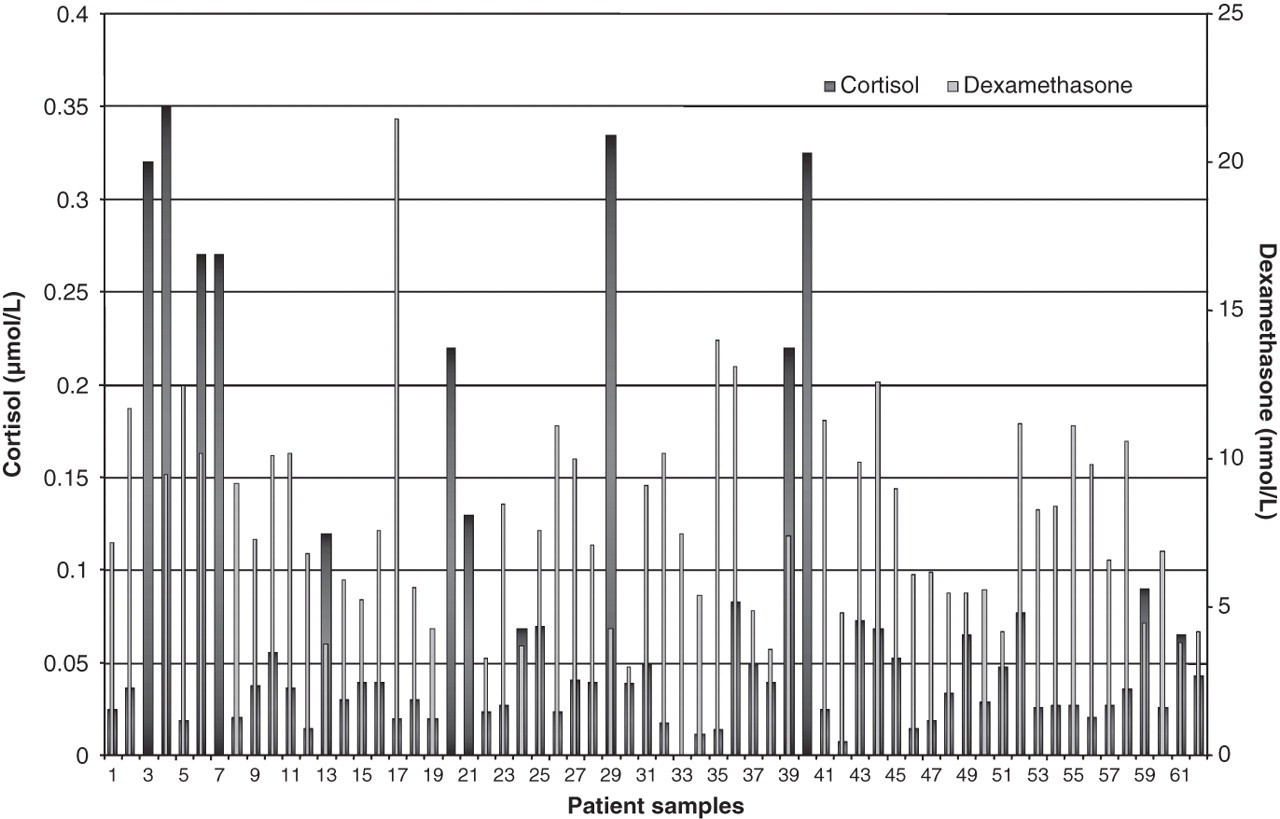
Discussion
We have developed an LC-MS/MS method for the simultaneous determination of dexamethasone and cortisol in human blood plasma to have an entire view on the LDODST. The validation data indicate a sensitive and precise measurement of the two analytes. The analytical range of cortisol is covering the wide range of possible patients' cortisol concentrations. Dexamethasone is quantified with acceptable standard deviations, also in the very low nanomolar range (1.89 nmol/L). We could improve the S/N ratio at very low concentration by summing up peak areas of several ion transitions. Investigations with respect to recoveries and linear range also yielded satisfying results for both analytes.
Frerichs and Toernatore 10 have also developed a method for the simultaneous LC-MS determination of dexamethasone and cortisol next to prednisone and prednisolone in human serum. However, as they determined the LOQ for dexamethasone at 10.7 ng/mL (27.3 nmol/L), the sensitivity would not be sufficient to apply the method in the LDODST. The LC-MS/MS method of Taylor et al. 13 allowed the parallel measurement of various synthetic steroids in serum, plasma, urine and tablets. Although a very low LOQ of dexamethasone was observed, the method would also not be suitable for our investigations with respect to the LDODST as cortisol was not determined at all.
Our method was applied to evaluate the dexamethasone and cortisol concentrations in plasma samples of patients who followed the LDODST. For method comparison we measured cortisol in 45 patients' plasma samples post-LDODST by LC-MS/MS as well as by ECLIA. For cortisol, the method comparison with a routinely applied ECLIA was very satisfying. The dexamethasone concentration range was comparable to the results of Meikle 6 who observed concentrations from 3.3 to 19.6 nmol/L. This was also in agreement with calculations on the theoretically expected dexamethasone concentrations which are, for example, 6.5 nmol/L for a person of 60 kg and 4.8 nmol/L for a person of 80 kg. LDODST cut-off levels for cortisol have been described extensively. The current guideline uses a cut-off level of 0.05 μmol/L for cortisol. Morning cortisol concentrations below 0.05 μmol/L after the LDODST exclude CS with a sensitivity of 95%. 4 In the evaluation of false-positive results, Meikle 6 reported a cut-off level for dexamethasone: the concentration after the LDODST should be above 5.6 nmol/L. Future studies including a larger number of CS patients and healthy controls should give more insight into the variation in dexamethasone concentrations and the above-mentioned cut-off level.
Both cortisol and dexamethasone in our patients' samples derived after the LDODST could be quantitatively analysed. There is a clear advantage of analysing both analytes as opposed to analysing cortisol alone. In Figure 2, samples 1–47 were derived from patients suspected of CS and samples 48–62 from healthy controls. The LDODSTs of patients 3, 7, 20, 21 and 40 would have been interpreted as not suppressible, whereas most likely both patients showed a lack of drug compliance. Generally, no information on the dexamethasone concentration is obtained in routinely performed LDODSTs to diagnose CS and to prevent expensive follow-up examinations. Patients 4, 6 and 39 had a sufficient dexamethasone concentration but cortisol concentrations could not be suppressed, which is highly suggestive for CS. Patient 10, for example, yielded a comparable dexamethasone concentration as in patient 6; however, the cortisol concentration is only slightly increased. Additional analysis of the cortisol-binding globulin (CBG) concentration could be supportive for making the correct interpretation, since in the assay, total cortisol concentrations are being measured and increments in CBG concentrations (e.g. induced by estrogens) result in elevated cortisol concentrations. Healthy control samples 49 and 52 showed increased cortisol concentrations in the LDODST despite sufficiently high dexamethasone concentrations. The samples were derived from women using oestrogens. Samples 59 and 61 clearly indicated that sufficient dexamethasone concentrations have to be reached to suppress the cortisol concentration. In these two healthy controls, a variable absorption and metabolism of the dexamethasone may explain the insufficient dexamethasone concentration achieved and, as such, the cortisol concentrations should be interrelated with cause. As we concentrated in this article on method development and validation, we decided to not discuss all of these samples in detail.
O'Sullivan and co-workers have performed investigations on the pharmacokinetic behaviour of dexamethasone. 8 They observed an incomplete and variable dexamethasone absorption yielding a wide individual variability in plasma dexamethasone concentrations. They therefore also recommend measuring the dexamethasone plasma concentration together with cortisol in an LDODST.
Conclusion
We have developed and validated a simple LC-MS/MS method for the simultaneous quantitation of cortisol and dexamethasone in human plasma. The method has proven to be capable of taking the first step towards diagnosis of CS by differentiating whether the cortisol production was suppressed by the dexamethasone dose or not. The analytes are directly determined without the need for a derivatization step. All results of the validation procedure showed that a new reliable and robust LC-MS/MS method has been introduced that can routinely be applied in a clinical chemistry laboratory. For future work, our new method should be applied for further investigations with respect to the variation in dexamethasone concentrations that can be expected in plasma samples of patients who followed the LDODST. The observed interindividual variability in plasma dexamethasone concentrations provides an additional argument for a combined assay of cortisol and dexamethasone.
DECLARATIONS
