Abstract
Background
Branched chain amino acid (BCAA) analysis is needed for the diagnosis and management of patients with maple syrup urine disease (MSUD). We report an improved ultra performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS) method for the determination of BCAAs and Allo-Ile in dried blood spot (DBS) samples.
Methods
BCAAs were extracted from a 3 mm blood spot into methanol/water containing stable isotope internal standards. Eluents were dried and reconstituted in the mobile phase. Gradient elution was performed on an Acquity™ BEH C18 (100 × 2.1 mm, 1.7 μm) column. BCAAs were detected and quantified in the multiple reaction monitoring mode in a five-minute analysis.
Results
The assay was calibrated to give best possible alignment with plasma results. Retrospective analysis of newborn DBSs from six classic MSUD patients showed elevated alloisoleucine (Allo-Ile) in all cases. Two of four patients with mild disease had normal values; the other two had significant elevations in Allo-Ile.
Conclusions
Analysis of BCAA in DBS by UPLC-MS/MS is a useful second tier newborn screening test to identify classical MSUD and for monitoring of remote patients.
Introduction
Maple syrup urine disease (MSUD) is an autosomal recessive inherited disease with a prevalence of 1:185,000 caused by deficiency of branched-chain α-keto acid (BCKA) dehydrogenase multienzyme complex. 1 This results in a toxic accumulation of the branched-chain amino acids (BCAAs), leucine (Leu), isoleucine (Ile) and valine (Val), and their corresponding BCKAs in the blood, urine and the other tissues of the body. Alloisoleucine (Allo-Ile) is elevated in the plasma of classical MSUD patients but is typically below the limit of detection in normal individuals. 2
Ion exchange chromatography, using ninhydrin detection, 2 is the most commonly used quantitative method for BCAA analysis, but it is time consuming, and plasma assays require venipuncture and transport of samples to the reference laboratory requiring specialist packaging and typically courier delivery.
The introduction of electrospray ionisation tandem mass spectrometry (ESI-MS/MS) allowed expanding newborn screening programmes for inborn errors of metabolism, including MSUD, which have been shown to be effective for MSUD patients with the classic form, allowing early treatment. 3,4 Routine MS/MS methods fail to differentiate between isomeric amino acids, Leu, Ile, hydroxyproline (OH-Pro) and Allo-Ile, limiting sensitivity and specificity. 4,5 In 2008, Oglesbee et al. 1 developed a liquid chromatography-tandem mass spectrometry (LC-MS/MS) method for the separation and quantification of isomeric BCAAs in dried blood spot (DBS) and proposed its use as a second tier screen on samples with elevated total leucine/hydroxyproline.
In this paper, we report a modification of the method of Oglesbee et al. using ultra performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS) for the determination of BCAAs in newborn screening DBS as a second tier test. We also highlight the utility of the method for monitoring patients from home using DBS samples. The use of UPLC allowed a significant reduction in runtime, and calibration of the assay to give comparability with plasma results enhanced remote monitoring of patients.
Materials and methods
Chemicals and reagents
Allo-Ile, Ile and Val were supplied from Sigma-Aldrich (Sydney, NSW, Australia), and Leu from Fluka (Sydney, NSW, Australia). Acetonitrile (ACN) and methanol (MeOH) were obtained from BDH chemicals (Minto, NSW, Australia). Formic acid (FA) was from Ajax Finechem (Taren Point, NSW, Australia) and heptafluorobutyric acid (HFBA) from Fluka.
Samples
Sixty-two DBS samples from MSUD patients undergoing routine monitoring were taken at the same time as plasma samples for BCAA analysis by an amino acid analyser (Biochrom 30, Cambridge, UK). For preparation of calibrators, blood samples from healthy individuals were enriched with the target analytes.
Experimental conditions
Analysis was performed using the Waters Acquity UPLC system with TQ detector (Waters Corporation, Rydalmere, NSW, Australia) with an Acquity™ BEH C18 (100 × 2.1 mm, 1.7 μm) column. A gradient elution was used for the separation; with mobile phase A (organic) comprizing ACN with 1 mL/L FA and 0.2 mL/L HFBA; and mobile phase B (aqueous) comprizing water with 1 mL/L FA and 0.2 mL/L HFBA at a flow rate of 400 μL/min. The gradient elution conditions were: 5% organic phase for 0.52 min, then linear gradient to 20% organic over 2.33 min, step to 100% organic held for 1.3 min, and step to 5% organic phase held for 1 min. The injection volume was 5 μL.
The TQ detector was operated in positive ion multiple reaction monitoring mode. The ESI-MS/MS parameters were optimized for the native compounds and their internal standards.
Data analysis was performed using the supplied Quanlynx application (Waters Corporation) using equal weighting of calibration points.
Calibrators
For initial calibration, the working DBS standards were prepared from a healthy control blood sample with added BCAAs. The added amounts were 0, 50, 100 and 200 μmol/L of Allo-Ile; and 0, 250, 500 and 1000 μmol/L of Val, Leu and Ile.
Following initial running of a number of paired plasma and DBS samples, values for the blood spot calibrators were re-assigned to give the best overall agreement with plasma results.
Sample preparation
BCAAs were extracted from a 3 mm DBS punch with 100 μL methanol/water (50/50 v/v) containing the internal standards in a 96-well microtitre plate. The plate was gently agitated for 30 min at room temperature, eluent transferred to an Eppendorf (Hamburg, Germany) microcentrifuge tube and evaporated under air at room temperature. After drying, samples were reconstituted in 80 μL of aqueous mobile phase, mixed, and centrifuged at 2150
Results
The BCAAs were reliably and reproducibly separated along with their internal standards within 3.5 min (Figure 1). The inter-assay precision determined over 90 batches expressed as CV% was 6.7% at 211 μmol/L for Val, 14.2% at 38 for Allo-Ile, 10.3% at 101 for Ile and 11.1% at 228 for Leu.
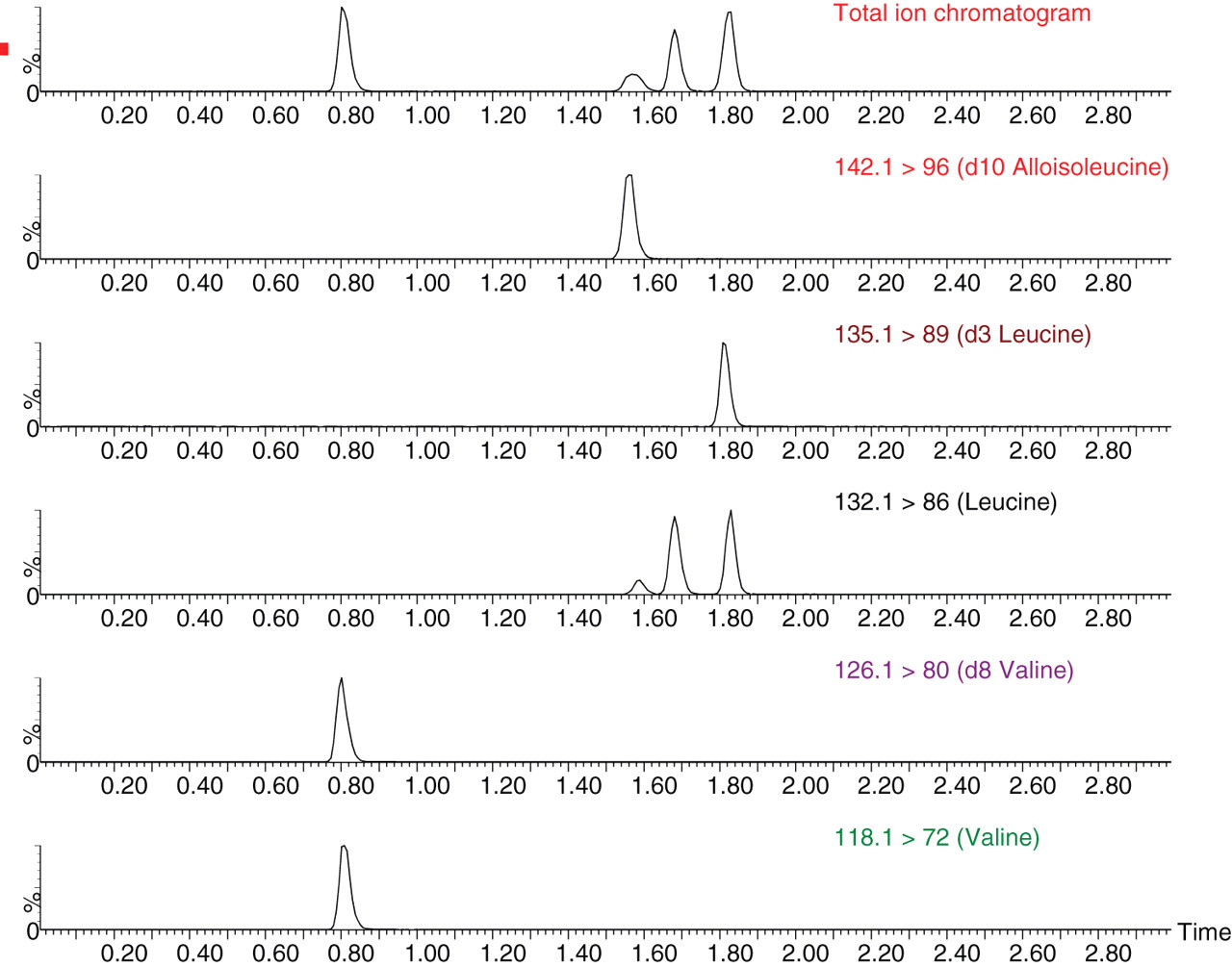
Typical ion chromatograms of branched-chain amino acids from a standard sample
Following re-assignment of calibration values, analysis of the results obtained from plasma samples taken simultaneously analysed by cation exchange chromatography and DBS assays (n = 62) showed excellent comparability with regression equations showing slopes of 1 (±1.5%) and acceptable intercepts for all analytes.
Allo-Ile was undetectable in most normal DBSs, although occasionally a peak of up to 3 μmol/L was found. Retrospective analysis of stored newborn screening samples from classical MSUD patients showed Allo-Ile ranging from 6 to 73 μmol/L. Two patients with mild/intermediate MSUD had borderline elevations of total leucines but clearly elevated Allo-Ile, both 16 μmol/L. Two further patients, previously reported with ‘intermittent’ MSUD and normal newborn screening results, 6 did not show elevated Allo-Ile in the newborn sample. These patients continue to have normal plasma amino acid values with undetectable Allo-Ile except when catabolic.
In the first six months of 2010, among 51,566 babies screened, plasma collection for further testing could be avoided in 23 babies (0.04%). In addition, employing a lower cut-off (350 μmol/L) for second tier testing, a further 182 samples with borderline total leucine levels were tested by this method.
Discussion
This method improves the determination of Allo-Ile and the other BCAAs by taking advantage of improved separations offered by UPLC-MS/MS systems. Our method requires a higher concentration of ion pair reagent but has improved resolution and reduced mobile phase flow rate, running time and injection volume compared with conventional high-performance liquid chromatography. 1
Using Allo-Ile as a second tier marker clearly distinguished between normal and affected samples from individuals with classical MSUD. However, as previously reported, 6 patients with attenuated forms of the condition may have normal newborn screening results and our two previously reported patients also had undetectable Allo-Ile.
We eliminated the need for further sampling in a total of 205 babies with elevated total leucine on primary screening in a six-month period, at the same time reducing the cut-off level of leucine.
By aligning DBS and plasma values, we have been able to extend DBS monitoring of MSUD patients on treatment using this technique. Parents or patients take their own samples at home and in most cases, samples mailed before 17:00 are delivered to the laboratory within one to two days and results are available before noon. Results from plasma samples taken during acute admissions or outpatient clinic visits are seamlessly compared with home monitoring. It has proved useful in identifying deteriorating control and we successfully managed a pregnancy in a woman with classical MSUD living over 300 km from the metabolic centre.
In summary, we have developed an improved method for the reliable quantitative analysis of all of the BCAAs using DBS, and have demonstrated the utility of this method as a second tier for newborn screening programmes and for remote monitoring of patients with MSUD.
DECLARATIONS
