Abstract
Background
We report a liquid chromatography-tandem mass spectrometry (LC-MS/MS) method for the detection of four commonly prescribed steroid drugs (prednisolone, dexamethasone, betamethasone and beclomethasone dipropionate) while simultaneously measuring 24-h urine free cortisol and cortisone in patients.
Methods
Two hundred and fifty microlitre aliquots of urine were spiked with internal standard and extracted with dichloromethane. The MS instrument was operated with positive electrospray and multiple reaction monitoring. Two transitions were monitored for each analyte of interest and the ratio of the intensities of the product ion fragments was established.
Results
The LC-MS/MS method for the measurement of urine free cortisol and cortisone was established. The assay was linear up to 788 nmol/L for cortisol and 777 nmol/L for cortisone, with a limit of quantitation of 5.0 nmol/L for both. Analysis time per sample was seven minutes. Transitions for four synthetic glucocorticoids were included, and they were identified based on the ratio of the intensities of product ion fragments. Analysis of 219 samples collected from 154 patients (55 male and 99 female) revealed the presence of prednisolone in five samples from three patients. Dexamethasone was detected in samples from four patients, and betamethasone was detected in one sample.
Conclusion
This is the first LC-MS/MS method in routine use to combine quantification of urinary cortisol and cortisone and detection of synthetic glucocorticoids in patients being investigated for Cushing's syndrome. Since the most common quoted cause of Cushing's syndrome is steroid treatment, this is a valuable diagnostic tool.
Introduction
Cushing's syndrome is characterized by chronic glucocorticoid excess. It may be adrenocorticotropic hormone (ACTH)-dependent (Cushing's disease or ectopic ACTH production) or ACTH-independent (adrenal adenoma or carcinoma), but the most common quoted cause is steroid treatment, which may be caused by accidental or iatrogenic supraphysiological administration of corticosteroids. 1 Prolonged glucocorticoid therapy may lead to a similar clinical picture as endogenous Cushing's syndrome: truncal obesity, proximal muscle weakness, bone disease, marked hypertension and hirsutism or acne. 2 Endogenous corticosteroid production may also be suppressed. 3
Isoenzymes of 11β-hydroxysteroid dehydrogenase (type 1 and type 2) regulate the tissue availability of cortisol. 4,5 11β-HSD2 is highly expressed in mineralocorticoid-responsive tissue, such as the distal convoluted tubule of the kidney, and catalyses the inactivation of cortisol to cortisone to prevent its binding to the mineralocorticoid receptor (Figure 1). 6 Saturation of the enzyme occurs in patients with Cushing's syndrome. Loss of 11β-HSD2 activity causes the syndrome of apparent mineralocorticoid excess. Over 30 mutations of the 11β-HSD2 gene have been reported, with severe manifestations resulting in childhood hypertension and hypokalaemia, while milder forms have been identified in adulthood. Reduced 11β-HSD2 activity will result in an increased ratio of urinary cortisol to cortisone or their metabolites. 7 Simultaneous measurement of urine free cortisol and cortisone has been recommended to detect variations in 11β-HSD2 activity. 8,9
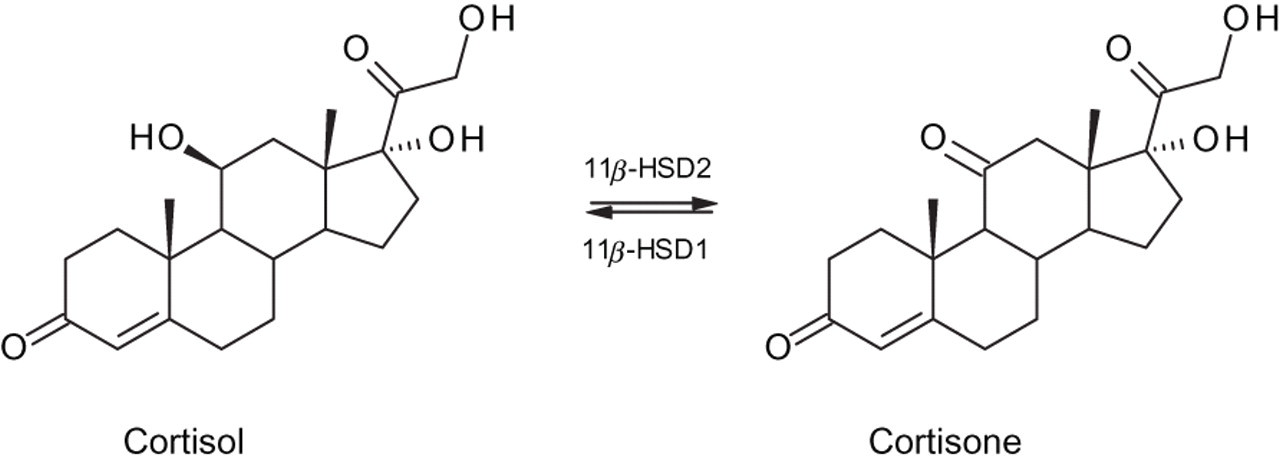
Interconversion of cortisol and cortisone
The recommended screening tests for Cushing's syndrome are either an overnight dexamethasone suppression test or 24-h urinary free cortisol collection, which is a direct assessment of circulating free cortisol. 10 There are a number of published methods for liquid chromatography-tandem mass spectrometry (LC-MS/MS) analysis of urine free cortisol. 11–16 The use of corticosteroids is strictly controlled in livestock production by the European Union and is prohibited in athletes by the World Anti-Doping Agency. As a result, LC-MS/MS methods have also been developed for the detection of synthetic corticosteroids in matrices such as plasma, urine, milk, animal tissues and hair samples. 17–30 Taylor et al. developed a separate screening tool for patients with unexplained cortisol suppression or possible iatrogenic Cushing's syndrome. This LC-MS/MS method quantified 14 frequently used synthetic corticosteroids in the serum, plasma and urine. 31
We have developed a LC-MS/MS method for the measurement of urine free cortisol and cortisone, but we have also incorporated transitions for four common synthetic glucocorticoids. The steroids chosen for this study were prednisolone, dexamethasone, betamethasone and beclomethasone dipropionate. Prednisolone, dexamethasone and betamethasone are frequently prescribed corticosteroids with predominantly glucocorticoid activity, while beclomethasone dipropionate is an inhaled corticosteroid for the management of asthma and hay fever.
Materials and methods
Patient samples
Twenty-four-hour urine samples (with boric acid preservative) were collected from patients under investigation for Cushing's syndrome. Two hundred and nineteen samples were collected from 154 patients (55 male and 99 female) ranging in age from seven to 82 years old. Aliquots were stored at −20°C before analysis. According to the local Ethics Committee, the use of residual material from patients did not require ethical review.
Materials
Hydrocortisone, cortisone, dexamethasone, prednisolone, betamethasone, beclomethasone dipropionate, ammonium acetate and formic acid were purchased from Sigma-Aldrich (Dorset, UK). The internal standard, 9,11,12,12-d 4-cortisol (IS), was obtained from Cambridge Isotope Laboratories Inc (Hampshire, UK). LC-MS grade solvents (methanol and water) were acquired from Fisher Scientific (Leicestershire, UK). Lyophilized internal quality control material (Assayed Urine Control Level 2 and 3) was obtained from Randox (Antrim, UK).
Stock solutions (1 g/L) of d 4-cortisol, cortisol, cortisone, dexamethasone, prednisolone, betamethasone and beclomethasone dipropionate were prepared gravimetrically in methanol. These solutions were diluted in 10% methanol: water (v:v) as required. Working calibrators containing cortisol and cortisone were prepared over the range 5.6–788 nmol/L. The concentration of the IS working solution was 164 nmol/L.
Sample preparation
Urine aliquots were thawed and centrifuged at 3000 rpm. Patient urine, controls or calibrators (250 μL) were pipetted into disposable borosilicate tubes. One hundred microlitres of the working internal standard solution were added to each tube and 1 mL of dichloromethane was added to extract the glucocorticoids. The tubes were vortex-mixed for 30 s and centrifuged for 10 min at 4°C at 3500 rpm. The organic layer was transferred to a clean autosampler vial. The solvent was evaporated to dryness in a vacuum centrifuge (MiVac Duo Concentrator, Genevac Ltd, Suffolk, UK; 12 min at 1000 rpm at 50°C). The residue was reconstituted in 400 μL of 10% methanol:water (v:v). Vials were sealed, mixed and assayed by LC-MS/MS.
Instrumental analysis
Reversed-phase ThermoHypersil high-performance liquid chromatography (HPLC) columns (BDS C8; 50 × 2.1 mm [i.d.]; 3 μm particle size) were obtained from Fisher Scientific. The HPLC system consisted of an Agilent 1100 system with a quaternary pump, a vacuum degasser, a temperature-controlled autosampler and a temperature-controlled column oven. The API 3000 tandem mass spectrometer and TurboIonSpray source were supplied by Applied Biosystems (Warrington, UK) and the system was controlled by Analyst software (Version 1.4.1; Applied Biosystems). Disposable 12 × 75 mm borosilicate tubes and autosampler vials (2 mL) were purchased from Fisher Scientific. Nitrogen gas was supplied by a gas generator (Peak Scientific, Inchinnan, Scotland, UK) and was used as the nebulizing, curtain and collision gas.
The API tandem mass spectrometer was operated in positive mode with a TurboIonSpray electrospray source operating at a voltage of +5 kV and desolvation temperature of 300°C. The conditions for the ion selection and collision-activated fragmentation of the molecular ions were optimized by continuous infusion (20 μL/min; Harvard infusion pump, Harvard Apparatus, Kent, UK) of pure compounds (10 μmol/L, 50% methanol:water [v:v] containing 0.1% formic acid). Following optimization, the instrument was operated in multiple-reaction monitoring mode (MRM). Two transitions were monitored for each compound to allow detection of interferences, and the qualitative ratio of the peak area intensities of the product ion fragments was determined by repeat analysis (n = 5) of solutions of each compound (500 nmol/L in 10% methanol:water [v:v]). The qualitative ratio acceptability limits for the controls and patient samples were set as ±20% of the values established. 32,33 The transitions monitored and the respective ratios for each analyte are shown in Table 1. Calibration was performed using the analyte to internal standard peak area ratios through linear regression with 1/x weighing to give a higher priority to calibration points with a low concentration.
Tandem mass spectrometry parameters for selected compounds: retention time (RT, min), SRM transitions (parent ion, 1° product ion & 2° product ion), collision energy (CE, eV) and ion ratios (±20%)
SRM, selected reaction monitoring
Chromatographic separation was achieved using a BDS C8 column (50 × 2.1 mm, 3 μm) maintained at 40°C. The mobile phase consisted of A (water) and B (methanol), both containing 2 mmol/L ammonium acetate and 0.1% formic acid (v:v). The flow rate was 500 μL/min, and 90 μL of extract was injected per assay. The composition of the mobile phase was 30% B for 1.5 min. The concentration of eluant B was increased to 90% over three minutes and then returned to 30% B over 0.3 min. The column was re-equilibrated with 70% (A):30% (B) for 2.2 min. The analysis time per sample was seven minutes.
Results from patient samples analysed by LC-MS/MS were compared with the results for the MS/MS group reported by the UKNEQAS scheme for steroid hormones.
Results
LC-MS/MS
Two mass transitions were optimized for each analyte (Table 1). Cortisol and cortisone were quantified based on the primary product ion (quantifier transition). The secondary product ion (qualifier transition) was used to assess the specificity of the analysis by calculating the ion ratio. 32,33 This ion ratio was determined for each compound based on the relative intensities of the product ions. It was used to confirm identity and detect the presence of interfering isobaric compounds. Figure 2 shows chromatograms of the primary and secondary transitions selected for each compound.

Liquid chromatography-tandem mass spectrometry (LC-MS/MS) chromatograms showing the primary and secondary transitions for patient samples containing (a) cortisol, (b) cortisone, (c) prednisolone, (d) dexamethasone, (e) betamethasone and (f) a urine sample spiked with beclomethasone dipropionate (500 nmol/L)
In patient samples, monitoring of the transitions corresponding to cortisol, m/z 363.3 → 121.2 and 363.3 → 97.2, revealed the presence of two peaks (4.5 and 4.8 min) preceding the cortisol peak at 5.0 min (Figure 2a). These peaks are clearly resolved by LC and have not been identified, but are likely to be steroid metabolites.
Interferences in this method should be minimized due to the chromatographic separation and high specificity of MRM, but the ion ratio may be used to detect the presence of any possible interfering compound. Dexamethasone, betamethasone and beclomethasone dipropionate do not elute at the same time as cortisol or cortisone (Figure 2). Prednisolone and cortisone, which have the same molecular weight, are clearly resolved by LC, and, although they have the same precursor ion, they have different product ions. Addition of dexamethasone, betamethasone and beclomethasone diproprionate (final concentration 500 nmol/L) to QC material had no effect on the measurement of cortisol and cortisone. Five hundred nmol/L of prednisolone increased the cortisol signal from the m/z 363.3 → 121.2 transition (2.2% interference). Kushnir et al. 12 found that a metabolite of prednisolone, tetrahydroprednisolone and the drug fenofibrate interfered with the m/z 363 → 121 transition for cortisol but not the 363 → 97 transition. We detected prednisolone in samples from three patients. The ratio of the product ions (121.2/97.2) was increased as predicted, and the m/z 363.3 → 97.2 transition was used to quantitate cortisol.
Betamethasone and dexamethasone are epimers, where the only structural difference is the orientation of the methyl group on C16 (Figures 3d and e). Under the chosen chromatographic conditions, the two compounds co-elute at 5.6 min. The product ion spectra for dexamethasone and betamethasone showed identical common fragments (m/z 393.1 → 147.2 and 393.1 → 237.2). However, the ratio of the product ions (mean ± 20%) using the same collision energy was clearly different: 2.88 ± 0.58 for betamethasone and 1.27 ± 0.25 for dexamethasone, enabling us to distinguish between the two synthetic glucocorticoids (Figures 2d and e).
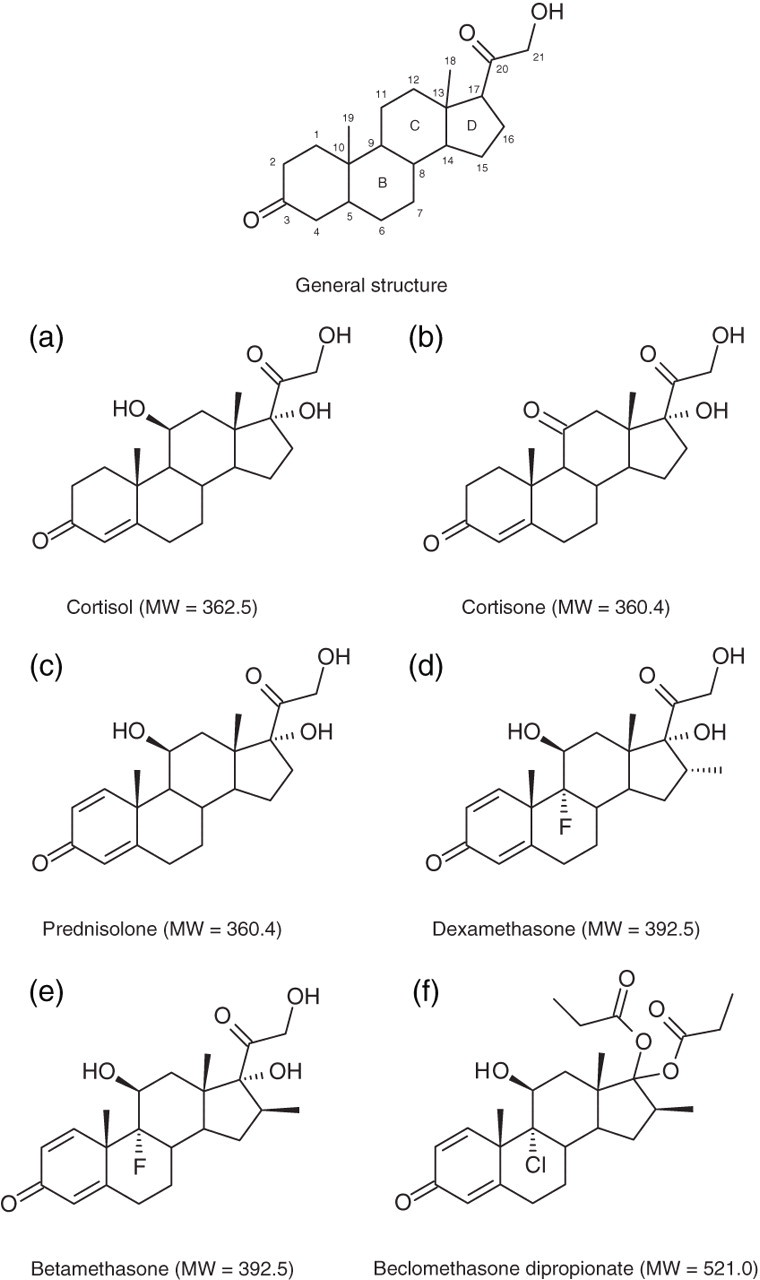
Chemical structures of the selected corticosteroids
Assay performance and validation
d 4-Cortisol was used as internal standard to quantitate cortisol and cortisone. The concentrations ranged from 5.6 to 788 nmol/L for cortisol and from 5.5 to 777 nmol/L for cortisone, and linear responses were achieved over this range.
The limit of detection is defined as a peak with a signal-to-noise (S/N) ratio >3 and the limit of quantitation as a S/N ratio >10. A 10% methanol:water (v:v) solution of 5.6 nmol/L cortisol and 5.5 nmol/L cortisone was measured 10 times, and the limit of quantitation was set as 5.0 nmol/L for cortisol and cortisone (Table 2).
Limit of quantitation established using a 10% methanol:water (v:v) solution of 5.6 nmol/L cortisol and 5.5 nmol/L cortisone
S/N, signal-to-noise ratio
The synthetic glucocorticoids were not quantitated as part of this assay, but their limits of detection were determined. Each corticosteroid was added to urine to give a final concentration of 5.0 nmol/L (n = 5). The S/N ratio for each compound was much higher (>100) than that required for limit of detection (>3).
The intra- and inter-assay precision was calculated for cortisol and cortisone by analysis of the internal quality control material. Two levels of Randox Assayed Urine Control material (levels 2 and 3) are available and were prepared according to instructions. A third level was prepared by 1:2 dilution of level 2. Each level was spiked with an appropriate volume of cortisone (2.78 μmol/L) in 10% methanol:water (v:v) to achieve concentrations equivalent to the cortisol concentrations. Intra- and inter-assay precision data for cortisol and cortisone are shown in Table 3.
Intra- and inter-assay precision data (n = 20) for urine cortisol and cortisone using Randox quality control material spiked with cortisone
Method comparison
External quality assurance samples from UKNEQAS (n = 60) were analysed using LC-MS/MS. The cortisol concentrations from the external quality assurance samples were between 12 and 616 nmol/L. The Deming regression against the reported MS/MS method mean was y = 0.97x–11.0 (r = 0.99), where y is the LC-MS/MS method under evaluation and x is the method mean for the MS/MS group.
Patient samples
Of the 154 patients who provided urine collections, beclomethasone dipropionate was not detected in any of the samples analysed. We identified four patients positive for dexamethasone and one for betamethasone. The ion ratios (based on m/z 147.2/237.2) for the four patients on dexamethasone ranged between 1.22 and 1.46, and the ion ratio for the patient taking betamethasone (based on m/z 147.2/237.2) was 2.59. All these results were within the derived acceptance limits for the relevant compound.
Prednisolone was detected in five samples from three patients. The ion ratios for prednisolone (based on m/z 147.2/279.2) were between 2.49 and 2.81. The chromatogram in Figure 2c from one of the patients shows two peaks at 4.5 and 4.7 min in the m/z 361.2 → 147.2 and 361.2 → 279.2 transitions. These two peaks are similar to the ones seen in the cortisol transitions and attributed to steroid metabolites (Figure 2a). For these patients, the cortisol concentrations were calculated based on the m/z 363.3 → 97.2 transition to avoid interference from tetrahydroprednisolone, as recommended by Kushnir et al. 12 The concentrations of cortisol quantitated based on the m/z 363.3 → 121.2 transition were between three and 15 times higher than the concentrations calculated using the m/z 363.3 → 97.2 transition.
Two further patients had cortisol and cortisone less than 10 nmol/L, but further enquiries could not determine if this was due to administration of synthetic glucocorticoids or other drug therapy.
Discussion
Since many steroids are structurally similar and have identical fragment ions, selectivity was improved by two mass transitions for each analyte. 32,33 Product ion selection was based on its relative abundance, and the extent of the fragmentation was compound-specific, as shown by the ability to successfully differentiate between dexamethasone and betamethasone. 34–37 All the corticosteroids studied have keto groups at C3 and C20, a double bond at C4 and a methyl group at C10 (Figure 3a). The synthetic glucocorticoids also contain a double bond at C1. The primary product ions at m/z 121 and 147 have been attributed to fragmentation of the B and C rings, while the product ion at m/z 97 is thought to be composed of A-ring carbon atoms. The secondary product ion at m/z 237 has been explained by fragmentation of the C13–C17 and C15–C16 bonds. The ion at m/z 105 is also common to all the selected corticosteroids. 28 Determining the ratio of the peak areas of the two chosen product ions served a number of purposes: confirmation of the identity of the analyte detected, e.g. dexamethasone and betamethasone, and detection of interfering compounds, e.g. tetrahydroprednisolone.
This method was designed to detect four different synthetic corticosteroids simultaneously: prednisolone, dexamethasone, betamethasone and beclomethasone dipropionate. Samples from eight of the 154 patients under investigation for Cushing's syndrome (5.2%) were found to have detectable concentrations of synthetic glucocorticoids: four positive for dexamethasone, one for betamethasone and three for prednisolone. Beclomethasone dipropionate was not detected in any of the samples which may be explained by activation by esterases in the lung to the 17-monopropionate form. 38 Six of the eight patients had suppressed cortisol (<10 nmol/L), although cortisone was only suppressed in two patients. We were unable to determine if the suppression was caused by exogenous corticosteroids.
The concentrations of the synthetic steroids were not determined. This method was intended to be qualitative and informative but not quantitative. The effect of corticosteroid administration on cortisol secretion will depend on the half-life, the mode and duration of administration, the dose and the bioavailability of the steroid. 39 The risk of adrenal suppression may last for months for patients who have been on long-term glucocorticoid therapy but is difficult to predict. 3,40 Herbal remedies may contain glucocorticoids, and progestogens can have glucocorticoid effects at high dose. 2 Any agent that interferes with the cytochrome P450 system, which is involved in steroid metabolism, has the potential to interfere with corticosteroid metabolism. 41
By incorporating transitions for four common steroids and enabling their detection, clinicians may make informed decisions about patient management. Our investigation revealed that the patients with dexamethasone present in their urine samples were undergoing overnight dexamethasone suppression tests. This illustrates a need for further education of clinicians and patients, with the physiological effect of synthetic corticosteroids and other drugs being clearly communicated.
In summary, we have developed a sensitive and specific method for the routine measurement of cortisol and cortisone in urine and the simultaneous detection of a number of synthetic glucocorticoids. The method has been in routine use in this laboratory for 12 months and we have identified a significant number of patients on steroids (5.2%). In future, the number of synthetic glucocorticoids may be expanded and altered according to local prescribing practice.
DECLARATIONS
