Abstract
Background
Valid assays measuring free thyroxine (FT4) must perform without bias despite large variations in the concentrations and affinities of serum thyroxine-binding proteins in the population. We developed a new, rapid one-step labelled-antibody time-resolved fluoroimmunoassay (TRFIA) for FT4.
Methods
Based on the heterologous combination of anti-T4 monoclonal antibody and triiodothyronine–immunoglobulin G conjugate, a one-step TRFIA for FT4 detection was established and compared with the two-step DELFIA® Free Thyroxine Assay. Matrix interference caused by endogenous binders and exogenous non-esterified fatty acids (NEFA) was also accessed in the proposed assay.
Results
The developed method generally took only one hour, had a detection limit of 0.6 pmol/L and a large linear range of 2.5–120 pmol/L. The inter- and intra-assay coefficients of variation were 3.5–6.6% and 4.4–9.8%, respectively. Results from 110 specimens showed apparent agreement with that from the DELFIA® FT4 Assay with the square of the correlation coefficient of 0.975. This assay indicated that there was no significant dependence on endogenous binders and displayed potential interference by exogenous NEFA up to 5 mmol/L.
Conclusions
The proposed one-step heterologous TRFIA FT4 assay possesses simplicity, accuracy, high sensitivity and exhibits great potential for FT4 measurement. The combination of heterologous immunoassay with TRFIA may be advantageous for FT4 immunoassay development.
Introduction
Circulating thyroxine (T4) is tightly but reversibly bound to several proteins such as thyroxine-binding globulin (TBG), prealbumin and albumin in blood. About 0.03% of the blood thyroxine, which is not attached to serum proteins but in free form, has been demonstrated to be the metabolically active fraction and a better barometer for the thyroid status of the host. Hence, free thyroxine (FT4) is an important parameter when assessing thyroid functions. 1 Because of the extremely small fraction of T4 in the free form and wide variations in the concentration and affinity of serum thyroxine-binding proteins in human population, the measurement of FT4 is analytically difficult. 2 The golden rule for a valid measurement of FT4 in any assay is that under the assay conditions, serum protein-bound T4 is minimally displaced into the free phase and such displacement causes insignificant disturbance of the endogenous bound–free balance for T4 in the sample. 3 A repertoire of assays has been employed to directly measure FT4 concentration in human serum, including equilibrium dialysis, 4 ultrafiltration, 5 radioimmunoassay (RIA), 6 enzyme immunoassay (EIA), 7 time-resolved fluoroimmunoassay (TRFIA), 8 chemiluminescence immunoassay (CLIA) 9 and electrochemiluminescence immunoassay (ECLIA). 10 Labelled immunoassays, such as RIA, TRFIA, CLIA and ECLIA, are the most popular methods that have currently been used in clinical laboratories for FT4 determination. Based on the assay principle, labelled immunoassays for FT4 detection may be generally divided into three categories, namely, two-step method, 11 one-step labelled analog method 7,12 and one-step labelled-antibody method, 13,14 respectively.
The direct measurement of FT4 via equilibrium dialysis is mainly used as a reference method, but it is lengthy (usually 16–24 h) and technically demanding. The two-step assay (developed in the late 1970s and called back-titration methods), by definition, is independent of the influence of the serum proteins and their bound T4, but its execution is always laborious and inconvenient, especially when conducting the analysis manually. As an example of two-step assay, the DELFIA® FT4 kit (PerkinElmer Life Sciences, Turku, Finland) involves sequential multi-step TRFIA and takes about three hours to finish the whole sequence. 15 Historically, earlier one-step labelled analog methods could avoid the inconvenience of using the two-step approach and be amenable to automation. But these one-step labelled analog methods are highly vulnerable to changes of serum constituents, which leads to spuriously low values in patients with hypoalbuminemia, a common finding in hospitalized patients. 16 Hence, their imperfect performance has been criticized for a decade. 17,18
In the past few years, a new one-step assay design known as one-step labelled-antibody method was developed. One-step labelled-antibody method, also commonly referred to as solid-phase antigen-linked technique, resembles the classic one-step analog assay. 19 Consequently, both the problems and the advantages of this assay type are generally similar to those of one-step labelled analog immunoassays. However, as pointed out by Ekins, 20 it is important to recognize that the attachment of the analog to a solid support creates a ‘macroanalog.’ This ‘macroanalog’ may sufficiently differ in its physicochemical properties from the unattached analog. Physicochemical differences resulting from the binding of analog to the solid support confer kinetic differences, which might lead to decreased analog affinity for endogenous binding proteins and a more reliable measurement of the free hormone. The labelled-antibody approach is currently favoured in free hormone assay on most automatic platforms including Roche® Elecsys Systems (Roche Diagnostics GmbH, Mannheim, Germany) and Vitros® Immunodiagnostics (Ortho-Clinical Diagnostics, Cardiff, UK).
As a non-isotopic immunoassay, TRFIA is a well-established technique in routine immuno-diagnostic work, and it is employed in the DELFIA system. 21 Compared with RIA, EIA and CLIA, TRFIA is safe, highly sensitive, and has a wide dynamic range. Furthermore, the labelling procedure is very easy and produces labelled compounds with high specific activity and good stability with minimal influence on immunoreactivity and other bioactivities.
In comparison with traditional homologous immunoassay systems, heterologous immunoassay systems, benefiting from factors such as a steeper dose–response curve, faster kinetics, higher signal amplitude and higher sensitivity, are widely used to measure free ligands in biological fluids nowadays. 8,22,23 In this study, a one-step labelled-antibody TRFIA was developed for the determination of FT4 in human serum by attaching triiodothyronine (T3) conjugate (heterologous hapten) rather than T4 conjugate (homologous hapten) to a solid phase and using the anti-T4 antibody as a tracer. This new assay displayed desirable characteristics such as no radioactive waste, relatively simple operation, high sensitivity and large linear range. Hence, the new one-step labelled-antibody TRFIA for FT4 might be applied in clinical diagnosis settings for the determination of FT4 in patients.
Materials and methods
Preparation of T4-free human serum
The FT4 human serum was prepared as described by Chard
24
with minor modifications. Briefly, 20 g activated charcoal (Sigma-Aldrich, St Louis, MO, USA) was added to 100 mL pooled human serum. This was incubated at 4°C for 24 h with stirring on a magnetic agitator, followed by centrifugation at 20,000 ×
FT4 calibrations
The thyroxine (Sigma-Aldrich) stock solution of 1000 nmol/L was prepared in 0.04 mol/L NaOH solution. For calibration purposes, serial dilutions of the stock thyroxine solution in T4-free human serum samples were performed. The freshly prepared thyroxine solutions were calibrated using DELFIA® Free Thyroxine Assay in order to obtain a set of standards of known FT4 concentration, at concentrations of 0, 2.5, 6.6, 15, 42 and 120 pmol/L (S0, S1, S2, S3, S4 and S5), respectively, and stored at 4°C.
Synthesis of N-hydroxysuccinimide-activated triiodothyronine derivative
As shown in Figure 1, a solution of suberic acid bis(N-hydroxysuccinimide ester) (Sigma-Aldrich) (100 mg, 0.27 mmol) in dry N,N-dimethylformamide (1.0 mL) was added to a solution of T3 (Sigma-Aldrich) (75 mg, 0.12 mmol) in dry N,N-dimethylformamide (1.0 mL) and dry triethylamine (30 μL). The reaction mixture was stirred overnight at room temperature. After concentration, the activated T3 derivative was purified by preparative thin-layer chromatography (TLC; Merck, KGaA, Darmstadt, Germany) using chloroform/methanol (9/1, v/v) to develop the plates. Yield: 69 mg.
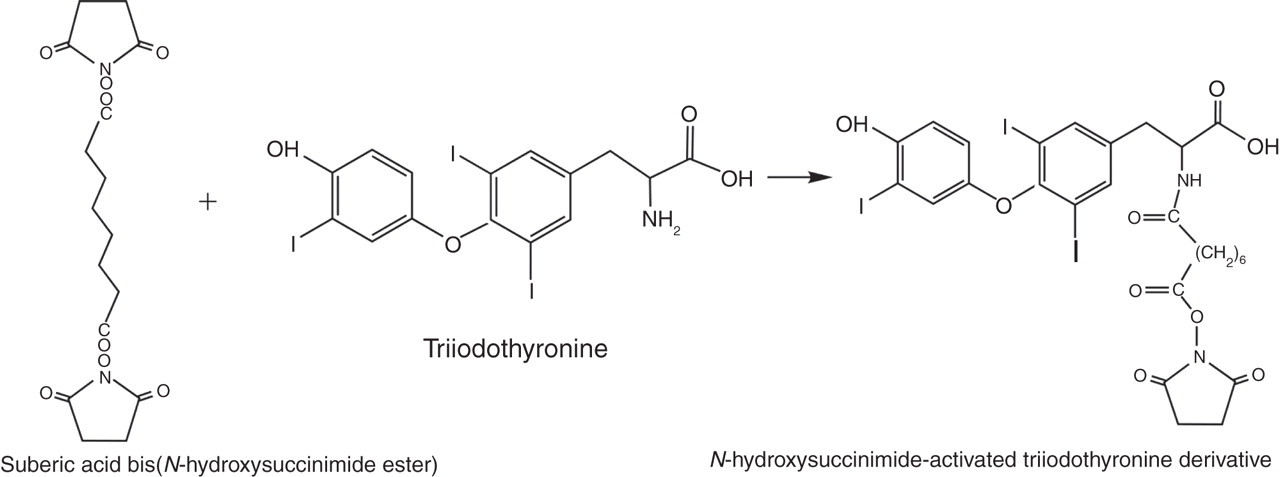
Synthesis of N-hydroxysuccinimide-activated triiodothyronine derivative
Preparation of triiodothyronine–immunoglobulin G conjugate
Rabbit IgG (Sigma-Aldrich) (20 mg) was dissolved in 0.3 mL of NaCl solution (150 mmol/L) containing 0.03 mL of 1 mol/L carbonate buffer (NaHCO3/Na2CO3, pH 9.0). To the resulting protein solution was slowly added 5 mg N-hydroxysuccinimide-activated T3 derivative dissolved in 50 μL 1,4-dioxane. The mixture was stirred for three hours at room temperature and then applied to a (1 × 40 cm) column of Sephadex G-75 (GE Healthcare Bio-Sciences AB, Uppsala, Sweden) that had been equilibrated with 0.05 mol/L carbonate buffer (pH 9.2) containing NaN3 (0.5 g/L). The column was eluted with the same buffer and the presence of protein in the fractions was determined by monitoring the absorbance at 280 nm on a Model EM-1 Econo UV monitor (Bio-Rad, Hercules, CA, USA). Fractions containing the conjugate were pooled, filtered through a 0.22-μm Millipore filter, and protein determination was performed by Bradford's method. 25 Then, the conjugate solution was adjusted to 1 mg/mL and stored at 4°C for further use.
Preparation of the labelled-antibody (anti-T4 antibody-DTTA-Eu3+)
Sodium azide was removed from the anti-T4 antibody (Medix, Kauniainen, Finland) solution by dialysis against 0.9% NaCl solution. Conjugation was performed using a 50-fold molar excess of N 1-(p-isothiocyanato-benzyl)-diethylene-triamine-N 1,N 2,N 3,N 4-tetraacetate-Eu3+(DTTA-Eu3+; Tianjing Radio-Medical Institute, Tianjing City, China) in 0.9% NaCl. The pH of the solution was adjusted to 9.0 with 1 mol/L sodium carbonate solution. After incubation overnight at room temperature, the europium-labelled antibody was separated from excess label by gel filtration on a (1.5 × 40) cm Sepharose-6B column (GE Healthcare Bio-Sciences AB) eluted with 50 mmol/L Tris-HCl buffer (pH 7.7) containing 0.9% NaCl and 0.05% NaN3. The number of europium ions incorporated per immunoglobulin molecule was determined by measuring the fluorescence in comparison to known EuCl3 standards. 26 On average, six europium ions were incorporated per IgG molecule. Bovine serum albumin (BSA; 1 g/L) was added as carrier protein to the labelled-antibody solutions.
Triiodothyronine–immunoglobulin G conjugate coating of the microstrips
The coating of triiodothyronine–immunoglobulin G (T3-IgG) conjugate was performed by physical adsorption. The microstrips (NUNC, Roskilde, Denmark) were coated with 150 μL T3-IgG preparation per well in 0.1 mol/L carbonate solution (pH 9.5) for 20 h at 35°C. After washing three times with wash solution, the coated wells were saturated for 20 h by adding 0.3 mL blocking buffer (50 mmol/L Tris-HCl, pH 7.2, containing 0.1% gelatin, 0.04% NaN3). The coated and saturated strips were aspirated and stored humid at 4°C until used.
Collection of specimens
One hundred and ten clinical serum samples including 18 hypothyroidism, 37 hyperthyroidism, 42 euthyroid and 13 non-thyroidal illness subjects were obtained from the Nanfang Hospital, Guangzhou, China, and stored at −20°C. Samples used to obtain precision data were prepared from volunteers and constituted the routine internal quality-control material for the assay. The Ethical Committee of Science and Technology Department of Southern Medical University approved collection of these samples.
One-step labelled-antibody TRFIA for FT4 determination
The principle of one-step labelled-antibody TRFIA for FT4 detection is shown in Figure 2. It was a solid-phase fluoroimmunoassay based on heterologous competition between solid-phase T3-IgG conjugate and FT4 in the sample for europium-labelled anti-T4 monoclonal antibody. The test sample and labelled anti-T4 antibody were simultaneously incubated in the presence of solid-phase-bound T3-IgG conjugate. After washing, enhancement solution, which dissociates europium ions from the bound labelled anti-T4 monoclonal antibody, was added into solution, where they formed highly fluorescent chelates with components of the enhancement solution. The labelled antibody bound to the solid phase was inversely proportional to the FT4 concentration. Thus, the fluorescence signal produced was inversely proportional to the FT4 concentration. The details are as follows: 25 μL standard or serum samples together with 150 μL of the diluted labelled anti-T4 antibody in assay buffer were added in duplicates in the wells coated with T3-IgG conjugate. The assay buffer was a Tris-HCl-based buffer (pH 7.4) containing 0.5% NaCl, 0.05% NaN3 and 0.1% gelatin. After incubation in the wells with stirring at 37°C for 60 min, the microstrips were washed six times with wash solution (9 g/L NaCl solution containing 0.5 mL Tween 20 and 0.5 g sodium azide per litre), and the enhancement solution was prepared according to Hemmila et al. 26 and added to the microstrips (150 μL/well). The resulting mixture was incubated at room temperature for five minutes with gently shaking. The fluorescence in each well was measured with a DELFIA 1235 analyser (PerkinElmer Life Sciences). The fluorescence counts were inversely proportional to FT4 concentrations in standards (with given FT4) and unknown samples.
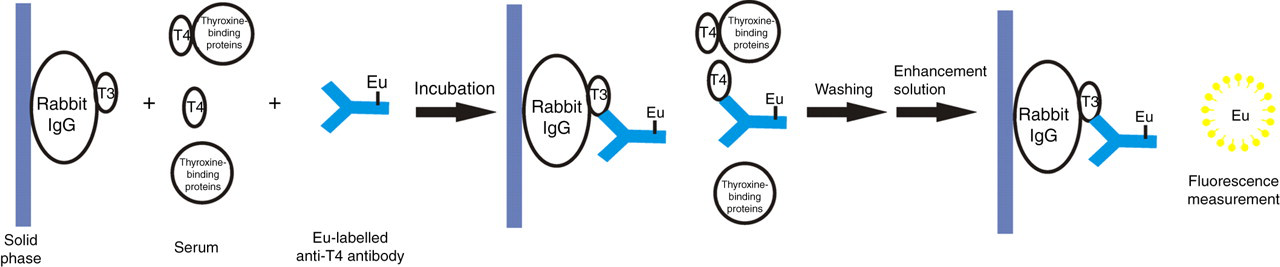
Basic configuration of one-step labelled-antibody TRFIA for FT4 detection. TRFIA, time-resolved fluoroimmunoassay; T3, triiodothyronine; T4, thyroxine; FT4, free thyroxine
The accuracy of one-step TRFIA was investigated by comparison with DELFIA® FT4 Assay (PerkinElmer Life Sciences). One hundred and ten serum samples with FT4 concentrations were analysed by both methods. DELFIA® FT4 Assay is a manual two-step TRFIA that is based on back-titration principle and using second-antibody separation. It takes about three hours to complete the assay. DELFIA® FT4 Assay was performed according to the manufacturer's recommended protocols.
Influence of serum thyroxine-binding proteins and non-esterified fatty acids
The possibility of interference, which might be caused by interactions between the T3-IgG conjugate adsorbed by microstrips and the endogenous thyroxine-binding proteins, was investigated in the one-step labelled-antibody TRFIA system by adding TBG (Sigma-Aldrich), human serum albumin (Sigma-Aldrich) or prealbumin (Sigma-Aldrich) to the zero standard. Various concentrations of TBG (0–100 mg/L), human serum albumin (0–100 g/L) and prealbumin (0–500 mg/L) were added to the zero standard, respectively. Any interactions between the solid-phase T3-IgG conjugate and thyroxine-binding proteins would be reflected by a change of fluorescence counts between the zero standard without added protein and the one with added protein.
Various concentrations (1–5 mmol/L) of aqueous oleic acid (Sigma-Aldrich) were used as non-esterified fatty acid (NEFA) to reconstitute lyophilized-delipidized euthyroid control serum. These samples were determined by the developed assay to evaluate potential interference by exogenous NEFA.
Results
Assay optimization
We obtained calibration curves with varying amounts of T3-IgG conjugate added to the wells during coating. As shown in Table 1, when working with a constant labelled-antibody dilution we observed that the fluorescence counts of the zero standard (B 0) increased significantly, but effective dose (ED50) increased little with the increase of the amount of coating (from 300 to 450 ng/well). Furthermore, with higher but still subsaturating amounts of coating (from 450 to 550 ng/well), B 0 had little change, but ED50 increased remarkably. In other words, the sensitivity of the calibration curve decreased. When reaching saturating amounts of coating (550 ng/well), B 0 began to decrease when the amount of coating continued to increase (550 to 800 ng/well). For optimal results, we chose to coat the microtitration wells with 450 ng/well for the T3-IgG conjugate.
Determination of optimal amount of coating*
Based on eight different amounts of coating, the corresponding B 0 and ED50 were obtained when keeping a constant labelled-antibody dilution of 1:800. B 0 is defined as fluorescence counts of the zero standard. ED stands for effective dose and ED50 is used to define the concentration required to give 50% of the maximum calibration response
*All the calibrators were analyzed by testing two replicates
†Based on an average of two replicates
The effect of dilution of europium-labelled antibody on the shape of the calibration curve was also studied in order to determine the optimal dilution of tracer. As shown in Figure 3, the shape of the calibration curve became increasingly steep with higher dilutions of labelled-antibody in the range from 1:600 to 1:1000, showing that the sensitivity of the calibration curve was enhanced under the circumstances. However, at even higher dilutions of labelled-antibody (1:1200 and 1:1400), the sensitivity decreased and fluorescence counts of the zero standard (B 0) dropped. Furthermore, the curve started to become relatively flat, narrowing the working dynamic range. Hence, 1000-fold tracer dilution was chosen as the optimum.
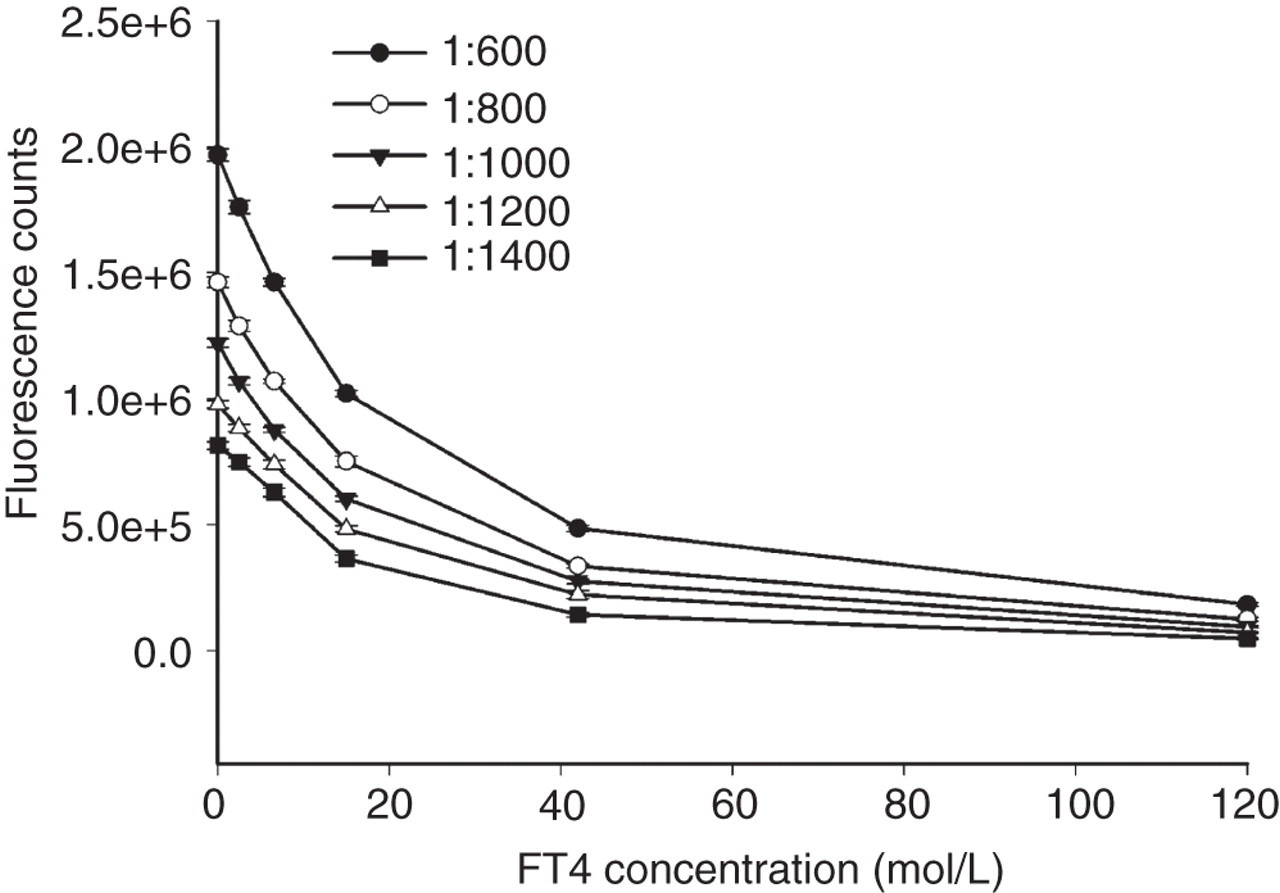
Optimization of the dilution of europium-labelled antibody. The corresponding dose-response curve was obtained at five different labelled-antibody dilutions of 1:600, 1:800, 1:1000, 1:1200 and 1:1400. Vertical bars indicate the ±SD (n = 2) for each concentration; FT4, free thyroxine
We optimized the incubation time of the assay so as to obtain accurate and reproducible results. The kinetics of the approach to equilibrium was showed by plotting tracer binding ratio versus incubation time (Figure 4). As shown in Figure 4, the assay was sufficiently near equilibrium for most standards after 30 min of incubation; hence, the developed assay was not sensitive to accurate timing since binding did not increase significantly after the specified incubation time. However, Figure 4 shows that the intra-assay precision of each standard at 30 or 40 min was poor compared with that at more than 60 min. Therefore, the optimum incubation was chosen to be 60 min with stirring at 37°C.
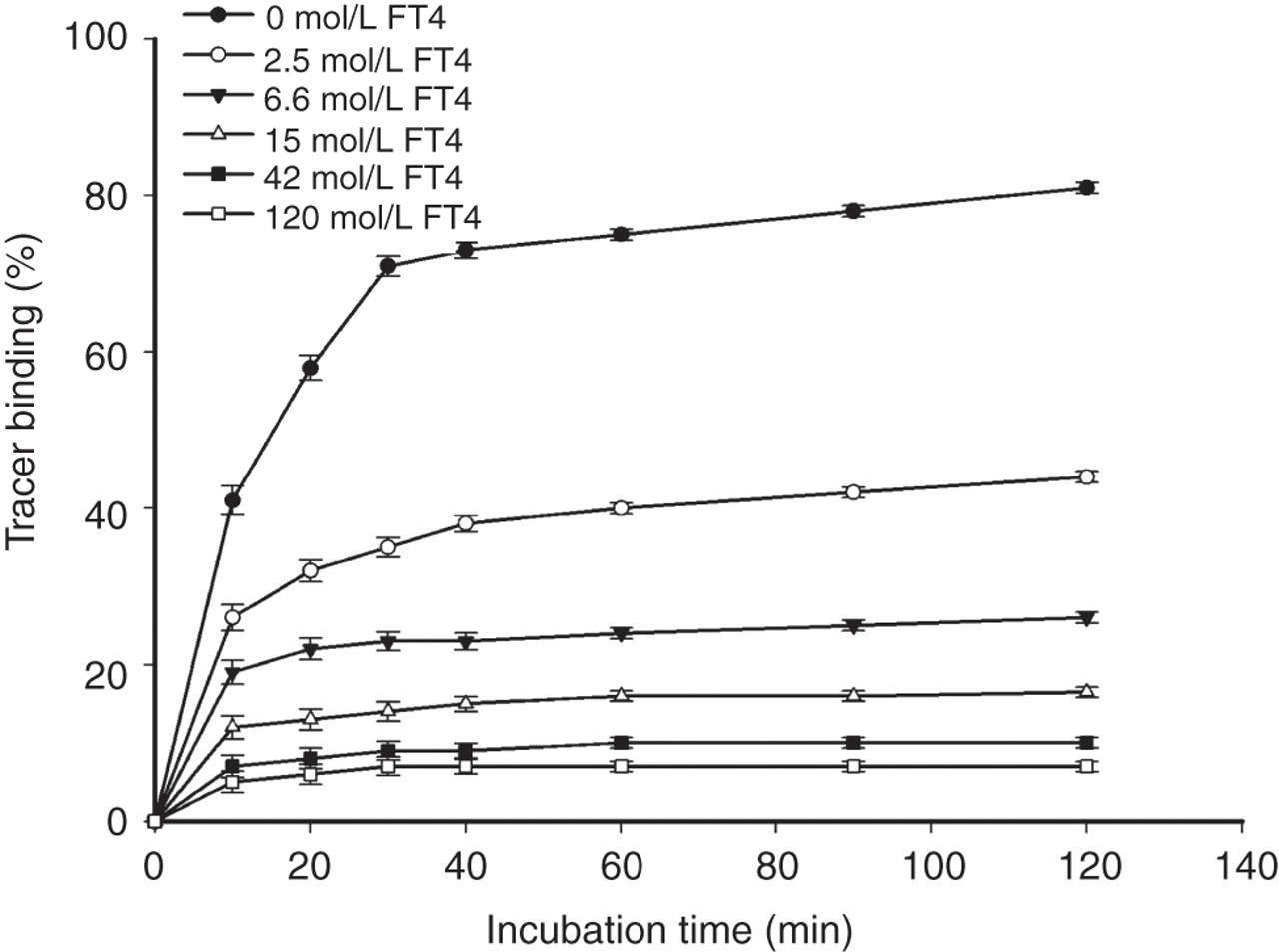
The influence of incubation time at indicated different FT4 concentrations. Y axis: tracer binding (%) = (fluorescence counts of labelled antibody bound/total fluorescence counts) × 100. Vertical bars represent the ±SD (n = 3) for each concentration. FT4, free thyroxine
Calibration and detection limit
Under the optimal conditions, the resulting calibration curve (logit-log plot) for FT4 is shown in Figure 5. A linearized standard curve was obtained by plotting logit (B/B 0) against the decimal log values of the corresponding standard concentrations.
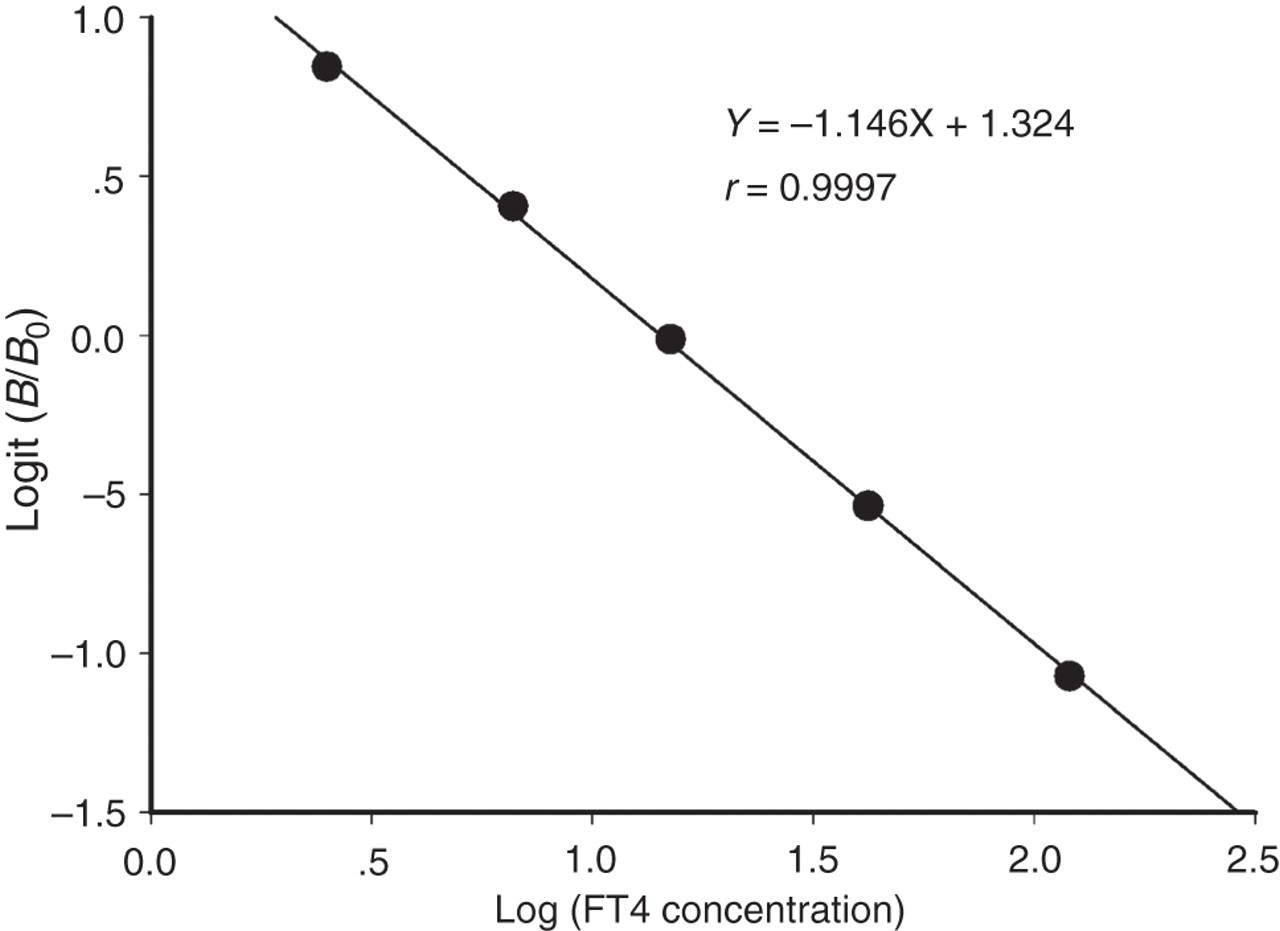
Calibration curve of the one-step labelled-antibody TRFIA for FT4 measurement. B 0 and B are defined as the fluorescence signals from tracer bound to solid phase at the zero FT4 standard and at the other FT4 standards, respectively. B/B 0 is their ratio. Logit (B/B 0) was calculated according to the formula: Logit (B/B 0) = ln[(B/B 0)/(1 − (B/B 0))]. Each data point is the mean of two replicates. TRFIA, time-resolved fluoroimmunoassay; FT4, free thyroxine
The detection limit was the lowest measurable analyte level that can be distinguished from zero. It was calculated as the concentration of FT4 that corresponded to the fluorescence counts that were two standard deviations less than the mean fluorescence counts of 10 replicate determinations of the FT4 zero standard.
Precision assay
Precision was evaluated using three different FT4 concentrations. As shown in Table 2, the coefficients of variation (CVs), calculated from values for three samples, were 3.5–6.6% (intra-assay) and 4.4–9.8% (interassay), respectively.
Data on precision of one-step TRFIA for FT4 determination
TRFIA, time-resolved fluoroimmunoassay; FT4, free thyroxine
Specificity assay
To probe the specificity of the developed one-step TRFIA, some molecules bearing analogous structures to T4, such as 3,3′,5-triiodo-
Cross-reactivity studies with anti-T4 monoclonal antibody
Influence of serum thyroxine-binding proteins and NEFA
The effects of increasing concentrations of TBG, albumin or prealbumin on the binding of solid-phase T3-IgG with the labelled-antibody in the zero standard are shown in Figures 6a–c. Increasing amounts of any thyroxine-binding proteins added to the zero standard had only a slight effect on the B 0 assay, indicating that the T3-IgG conjugate did not interact with the binding proteins. The addition of TBG concentrations up to 100 mg/L, prealbumin up to 500 mg/L or albumin up to 100 g/L, decreased the B 0 value by no more than 4%. Based on these results, these thyroxine-binding proteins have no apparent effect on B 0 determination.
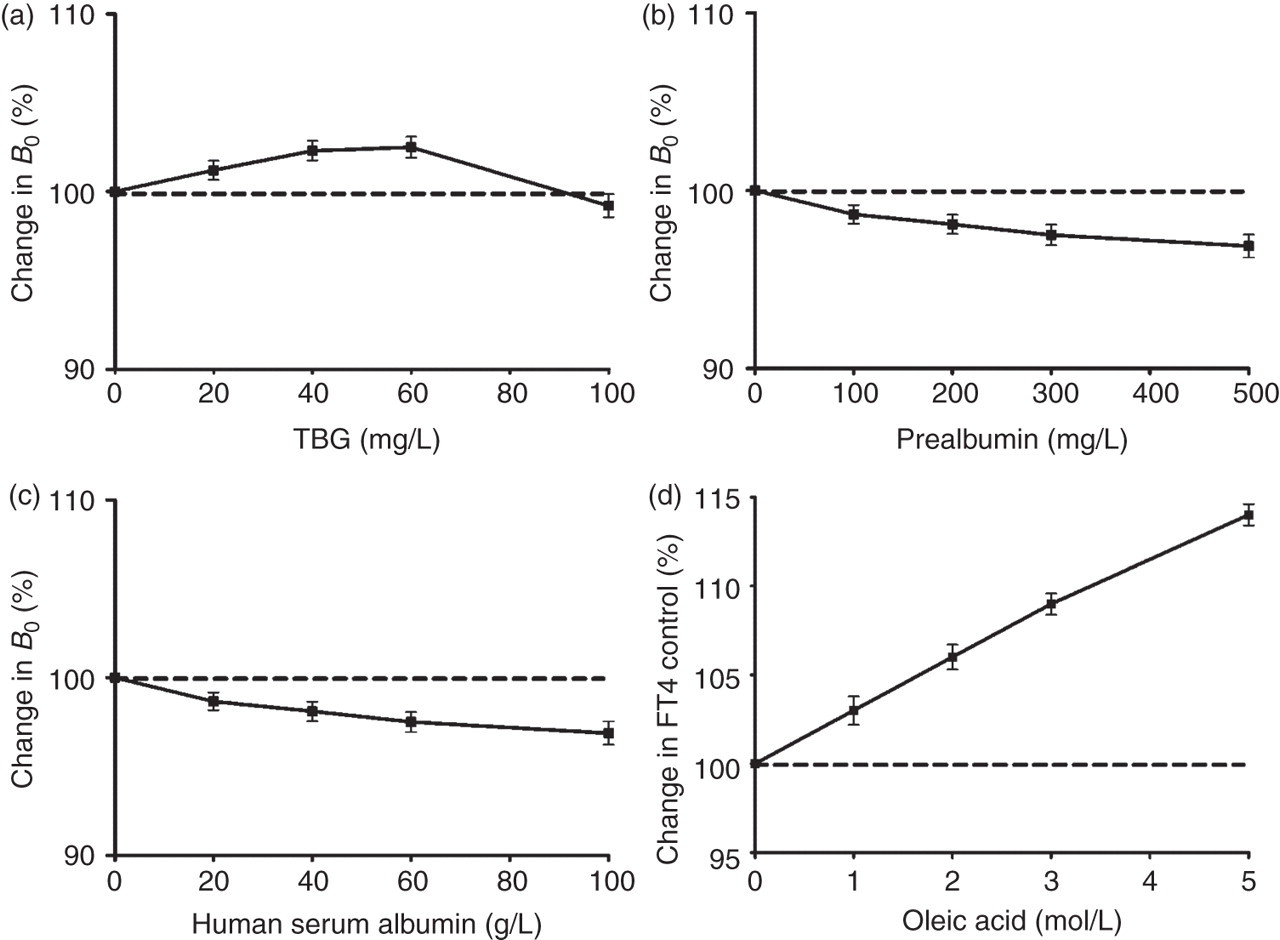
The influence of TBG (a), prealbumin (b), albumin (c) and oleic acid (d) added at various concentrations thereof. Change in B 0 (%) = (fluorescence counts at the zero standard with added protein/fluorescence counts at the zero standard without added protein) × 100 − 100. Change in FT4 control (%) = (concentration of FT4 control serum with added oleic acid/concentration of FT4 control serum without added oleic acid) × 100 − 100. Vertical bars indicate the ±SD (n = 3) for each concentration of the added thyroxine-binding proteins. FT4, free thyroxine; TBG, thyroxine-binding globulin
When various concentrations of oleic acid (NEFA) were added to delipidized euthyroid control sera, the variation found in FT4 was shown in Figure 6d. Adding oleic acid up to 5 mmol/L increased the estimate of FT4 by 15%, suggesting potential interference of oleic acid when using the new FT4 assay.
Comparison with DELFIA® FT4 Assay
The relationship between the new FT4 assay (legend x) and DELFIA® FT4 Assay (legend y) in all patients is shown in Figure 7. A significant correlation (r 2 = 0.975, P < 0.001) between the two assays was observed.
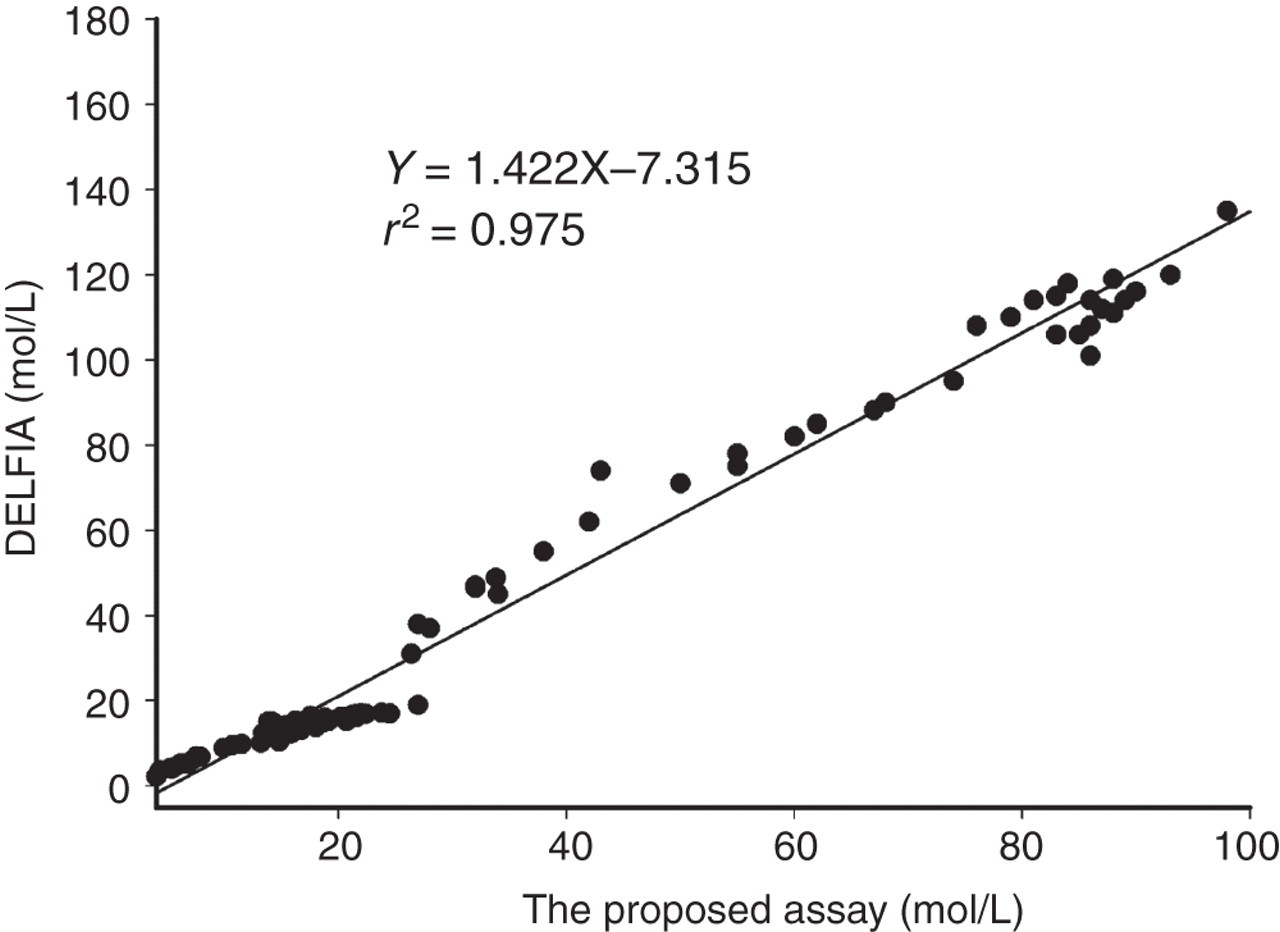
Correlation between results measured by the new TRFIA for FT4 and results measured by DELFIA® FT4 Assay (n = 110)
Good agreements between the two FT4 methods were achieved in all patient groups. Since the DELFIA® FT4 Assay is the standard assay used in clinical settings, this result indicated that the newly developed FT4 assay could be used as a diagnostic tool and is comparable, acceptable and useful for routine analysis of thyroid functions.
Discussion
In the current study, we have demonstrated the characteristics and validity of the new TRFIA for FT4 in the aspects of (i) sensitivity, precision and specificity; (ii) within-assay and between-assay CVs; (iii) interference by the serum thyroxine-binding proteins and NEFA; and (iv) apparent agreement with the DELFIA® FT4 Assay in various serum samples from patients.
Prior research had shown that the existence of a long spacer between the hapten and the carrier protein was important for binding and improves the assay sensitivity and CVs. 27–29 We set out to test this potentially helpful modification in our new FT4 assay by introducing a six-carbon long spacer arm between T3 and rabbit IgG molecule in our studies. Towards this end, a preactivated bifunctional reagent, suberic acid bis(N-hydroxysuccinimide ester), was used to obtain the N-succinimidyl ester derivative of T3 via a simple one-step synthesis. The desired product, N-succinimidyl ester derivative of T3, was synthesized by using 2.3-fold molar excess of the reagent suberic acid bis(N-hydroxysuccinimide ester) in the solvent of N,N-dimethylformamide. The final product was obtained after purification using TLC plates.
The developed method was a typically heterologous competition based on hapten heterology. Using T3-IgG conjugate instead of T4-IgG conjugate as the solid phase may have two advantages: (i) the affinity of the anti-T4 antibody for T3 was only about 1.5% of that for T4 (Table 3). Consequently, this drastically reduced affinity of the antibody to the hapten used in the assay made the dose–response curve steep, leading to a high sensitivity; and (ii) a much higher signal was obtained. It should be pointed out that the very low affinity of the anti-T4 antibody for T3 did not result in a weak immunoreaction (i.e. a low signal) in this proposed assay. In fact, the signal produced through immunoreaction was higher by using T3-IgG conjugate as solid phase than using T4-IgG conjugate as solid phase (data not shown). The reason was probably that T3 displayed more than 10-fold weaker protein-binding capacity in the serum than T4 does, which prevented serum thyroxine-binding proteins from binding to solid-phase T3-IgG conjugate and enhances the immunoreaction between solid-phase T3-IgG conjugate and europium-labelled antibody, thus producing a much higher signal. In addition, binding of T3 to thyroxine-binding proteins could be further reduced by conjugating its alanine side chain to a large protein (rabbit IgG), according to the references. 6,30,31 The resulting conjugation caused sufficient steric hindrance to keep serum proteins from binding to the solid-phase conjugation. As our experimental results showed exactly, the absence of interference by serum thyroxine-binding proteins (TBG, prealbumin and albumin) under the assay conditions indicated that the present assay was not affected by the high concentrations of human serum albumin, prealbumin and TBG. This was in agreement with the findings of Christofides et al. 13
High NEFA concentrations have been considered to be capable to displace quantities of bound T4 from the T4-binding sites of serum proteins and can increase the estimate of FT4, 32 but normal physiological concentrations of NEFA (<0.3 mmol/L) are unlikely to displace T4 and thus cause an increase in FT4. However, NEFA concentrations increased from 0.4 to 2.1 mmol/L after storage of samples at 4°C. 33 Thus, in order to investigate the robustness of FT4 assay to increases in NEFA, the experimental addition of NEFA in this proposed FT4 assay is of importance when inappropriate storage and handling of samples may be taken into account. As shown in Figure 6, up to 2.5 mmol/L concentration of oleic acid would cause only about 7% increase in FT4. On the other hand, in patients receiving heparin injections, blood lipoprotein lipase is activated, that is, NEFA generated in vitro from such patients by enzyme action subsequent to blood sampling. The adventitious NEFA produced in vitro, often to very high concentrations (5 mmol/L or higher), may lead to an increase in FT4. However, BSA added in the assay ingredients countered this artificial increase of FT4 concentration by mopping up the NEFA present in the sample (because NEFA can be bound by BSA), thereby bringing most values of FT4 concentration back to normality. 3 But it raises another question, that is, exogenous BSA in the assay system may sequester T4 from the serum equilibrium system and distort true FT4 values in serum. 22 Therefore, the presence of BSA in the assay ingredients appears to be a trade-off between increased robustness of the assay to adventitious increases in NEFA and decreased robustness of FT4 determination to changes in T4-binding capacities in serum.
For optimum effects, the BSA content in the labelled-antibody solution was set to 1 g/L in the labelled-antibody solution in this new TRFIA, in which the addition of oleic acid up to 5 mmol/L was observed to increase the FT4 measurement by 15%, causing possible interference of the estimate of FT4.
The agreement between the new TRFIA for FT4 and DELFIA® FT4 assay was very good, with the square of the correlation coefficient of 0.975 for results from various samples. In addition to the observed good agreement with the DELFIA® FT4 assay, the new TRFIA for FT4 also demonstrated a lower detection limit (0.6 pmol/L), a wider linear detection range (2.5–120 pmol/L) and a shorter immunoreaction time (60 min) compared with the commercial DELFIA® FT4 assay, which shows a detection limit of about 2.0 pmol/L, a linear range of 2.8–80pmol/L11 and about a three-hour immunoreaction time.
Conclusion
The new one-step TRFIA for FT4 measurement displays simplicity, accuracy and high sensitivity towards detecting FT4 levels in clinical samples. As such, it exhibits great potential for the clinical analysis of FT4. This new advance in FT4 measurement successfully combines one-step labelled-antibody heterologous immunoassay with highly sensitive DELFIA successfully. Upon further modification, it is envisioned that the combination of heterologous immunoassay with highly sensitive TRFIA may be applicable to the measurement of other free ligands (including free T3, testosterone, etc.), where improved sensitivity and reduction of interferences by endogenous binders would be highly desirable. Accordingly, we expect that this new technique may have a wide application in the measurement of free fraction of ligands in biological fluids.
DECLARATIONS
