Abstract
Background
The investigation and effective management of phaeochromocytoma involves biochemical measurement of either conjugated total urine or plasma free metadrenalines. Current analytical methods include enzyme-linked immunosorbent assays, high-performance liquid chromatography (HPLC) with electrochemical detection (ECD) or liquid chromatography tandem mass spectrometry (LCMS/MS). Since the first two methods are either extremely laborious, necessitate low sample run numbers, result in slow turnaround times or are subject to analytical interference, a robust, routine clinical method is not achievable. We established a novel sample preparation method to measure plasma free metadrenalines using LCMS/MS.
Methods
Three different solid-phase extraction (SPE) methods were compared: hydrophilic–lipophilic balance sorbent (HLB), weak cation exchange (WCX) and mixed mode cation exchange (MCX) and their ability to remove interfering compounds prior to LCMS/MS analysis. Maximum recovery of plasma free metadrenaline and plasma free normetadrenaline were achieved by positively charging compounds prior to SPE application.
Results
Compared with HLB and WCX cartridges, MCX extraction resulted in chromatography without co-eluting interference with superior assay precision and accuracy. Additionally, samples that could not be quantified because of interference using HPLC/ECD could be readily assayed using this new method.
Conclusions
The use of the MCX SPE method with LCMS/MS detection provides an improved assay to measure plasma free metadrenalines in comparison to many available alternative methods.
Introduction
The investigation and management of phaeochromocytoma necessitates biochemical measurement of either deconjugated urine or plasma free metadrenalines. 1,2 The use of plasma free metadrenalines is the most sensitive and specific method for the detection of pheochromocytoma compared with urine total metadrenalines and catecholamines. 3–8 Furthermore, an accurate, sensitive method is required to detect small tumours before significant clinical manifestations arise. 9,10 Initially, a method was established to measure plasma free metadrenalines based on the published method of Lenders 11 using solid-phase extraction (SPE) of plasma samples followed by analysis of plasma free metadrenalines by high-performance liquid chromatography (HPLC) with electrochemical detection (ECD). Although accurate, the HPLC/ECD method required 1 mL of heparinized plasma, was labour intensive, sample runs were small, turnaround time was suboptimal and analytical interference was sometimes noted, requiring sample recollection.
Consequently, a new method was established to measure plasma free metadrenalines using a mixed mode cation exchange (MCX) SPE cartridge sample clean up and subsequent analysis by isotope dilution liquid chromatography tandem mass spectrometry (LCMS/MS). MCX cartridges use both a reverse phase interaction and a sulfonic acid strong cation exchange mode to allow maximum retention of biogenic amines with the removal of weak bases to minimize ion suppression. The LCMS/MS assay requires 500 μL of plasma sample, can accommodate 40 patient samples in each analytical run and has a turnaround time of less than 24 h. Plasma samples demonstrating non-specific interference using HPLC with ECD detection could be assayed and reported using the new and more specific LCMS/MS technique. This LCMS/MS assay demonstrated acceptable agreement with the HPLC/ECD assay and excellent agreement with the Royal College of Pathologists Australia (RCPA) Quality Assurance program (QAP). Some ion suppression issues noted with the internal standard are modest and were readily addressed by sample dilution.
Method development
The first published method evaluated 12 required isopropanol precipitation of plasma proteins with separation, evaporative concentration and reconstitution of the supernatant sample before analysis by LCMS/MS. This LCMS/MS method provided accurate results, but was not acceptable for routine use due to frequent and considerable ion suppression. Within run internal standard coefficients of variation (CV) were often as high as 50%. A number of different gradients and analytical columns including ultra performance liquid chromatography (UPLC) were trialled to minimize suppression, but none were successful.
We compared various SPE preparation techniques including Waters hydrophilic–lipophilic balance sorbent (HLB) and weak cation exchange (WCX) cartridges; however, results were inconsistent and in the case of WCX, produced poor chromatography. The Waters MCX cartridges demonstrated improved chromatography and analytical precision.
MCX assay protocol
Charcoal stripped serum (SeraCon 2 CD, cat. no. BB220111L) from SeraCare spiked with pure metadrenalines were used as calibrators.
Quantification was performed using isotope dilution multiple reaction monitoring with the following transitions: metadrenaline (179.7 → 147.6), normetadrenaline (165.8 → 133.6), D3 metadrenaline (182.8 → 150.7) and D3 normetadrenaline (168.7 → 136.6). The cone voltage was set at 35 V for the metadrenalines and 27 V for the normetadrenalines. The collision energy was set at 18 eV for all analytes.
Preparation of plasma samples involved considerable experimentation to provide high analytical recovery and precision and is summarized below. The use of formic acid was applied to positively charge plasma metadrenalines before each wash protocol, which were implemented to remove ion-suppressing interfering compounds.
Sample extraction
MCX cartridges were treated according to the following protocol:
1.2 mL 1% ammonia in methanol; 3 × 2.5 mL de-ionized water; 2.5 mL 0.001% formic acid; 500 μL sample is diluted with 1 mL 0.001% formic acid and 50 μL of 20 nmol/L deuterated metadrenalines and loaded into the cartridge; Wash with 1.2 mL 10 mmol/L phosphate buffer, pH 8.5; 1.2 mL de-ionized water; 1.2 mL methanol; Elute with 1.3 mL 5% ammonia in methanol, into a Waters Max™ recovery vial; Dry eluate in a MiVac centrifugal evaporator at 50°C for one hour; Reconstitute in 50 μL 0.1% formic acid and mix; Add 450 μL acetonitrile and mix. Pellet any precipitate prior to analysis.
The elution gradient is shown in Table 1, followed by 2.5 min equilibration at initial conditions. The flow rate was 0.3 mL/min.
Elution gradient for sample extraction
The functional sensitivity was determined using patient samples assayed in triplicate and was defined as the minimum concentration with a CV% of less than or equal to 20%. The analytical sensitivity was defined as a concentration with a signal to noise ratio of >3 for both analytes.
Results
The functional sensitivity was determined to be 100 pmol/L for normetadrenaline and 60 pmol/L for metadrenaline. The analytical sensitivity was 15 and 7 pmol/L for normetadrenaline and metadrenaline, respectively.
Extraction efficiency experiments were performed on two patient pools by spiking with pure material prior to extraction and comparing this result to non-extracted material. Extraction recoveries varied from 73% to 102% and 85% to 103% for normetadrenaline and metadrenaline, respectively. Each batch included an unextracted internal standard to check for between run consistency and was found to be approximately 80% for both internal standards. Recovery experiments were also performed for each change of lot number for the MCX cartridges.
Intra-assay precision data for this method were performed using 10 replicates of pooled patient plasma and is summarized in Table 2. Inter-assay precision was determined over 24 batches, and provided CVs of 7.2% (mean 680 pmol/L) and 6.6% (mean 3770 pmol/L) for normetadrenaline and 7.5% (mean 480 pmol/L) and 5.9% (mean 1800 pmol/L) for metadrenaline.
Intra-assay precision using three separate patient plasma pools (N = 10)
CV, coefficient of variation
A representative chromatogram obtained from a patient with phaeochromocytoma is shown in Figure 1.
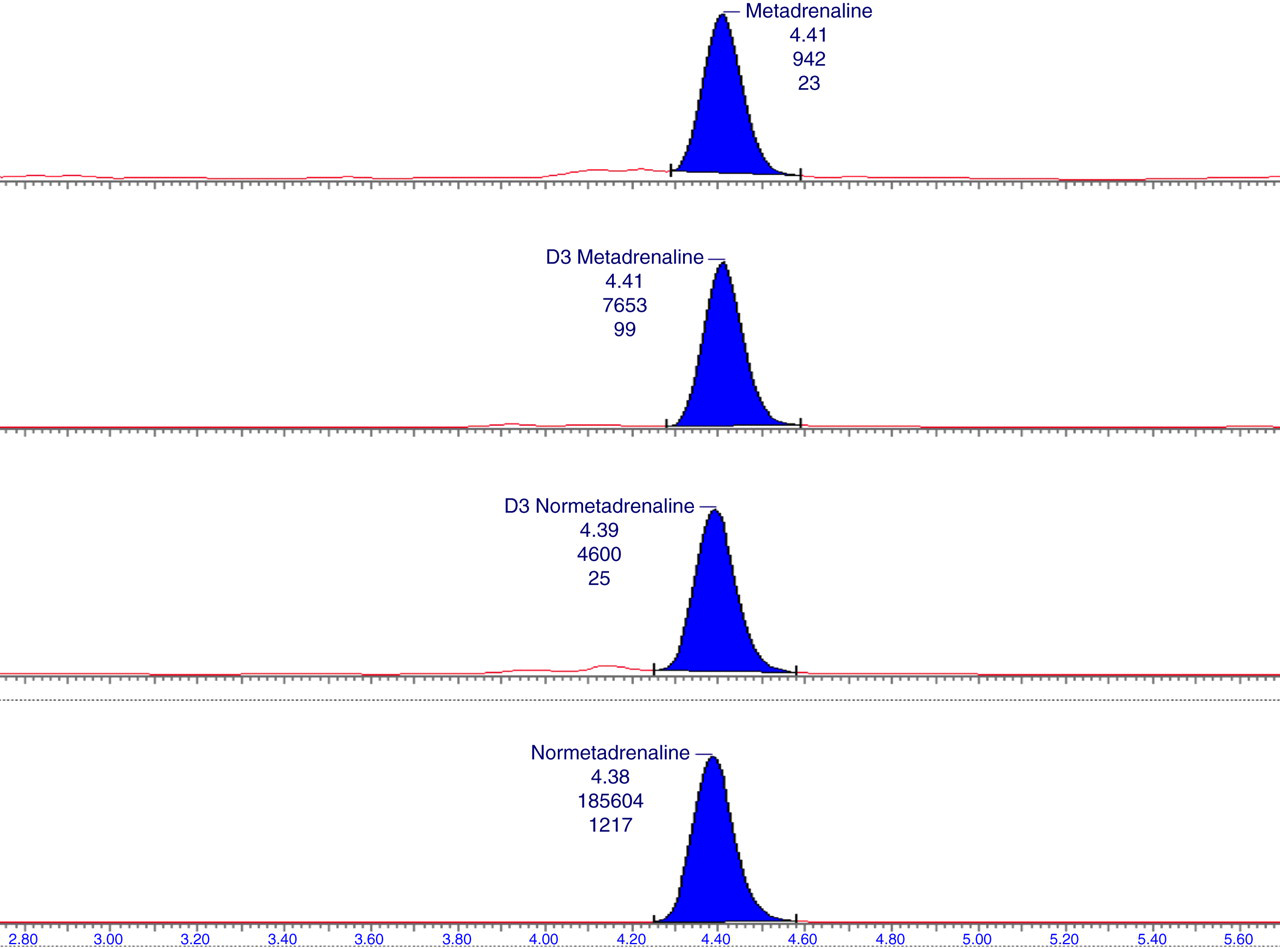
Chromatogram from a patient with confirmed phaeochromocytoma. The normetadrenaline concentration was 1850 pmol/L and the metadrenaline concentration was 80 pmol/L. Transitions used were metadrenaline (179.7 → 147.6), normetadrenaline (165.8 → 133.6), D3 metadrenaline (182.8 → 150.7) and D3 normetadrenaline (168.7 → 136.6). Information for each peak shows compound name, retention time, peak area and signal/noise ratio
Matrix suppression effects were assessed by injection of plasma extract during postcolumn infusion of pure compound. This showed no co-elution of matrix ion suppression and the analytes of interest (Figure 2).
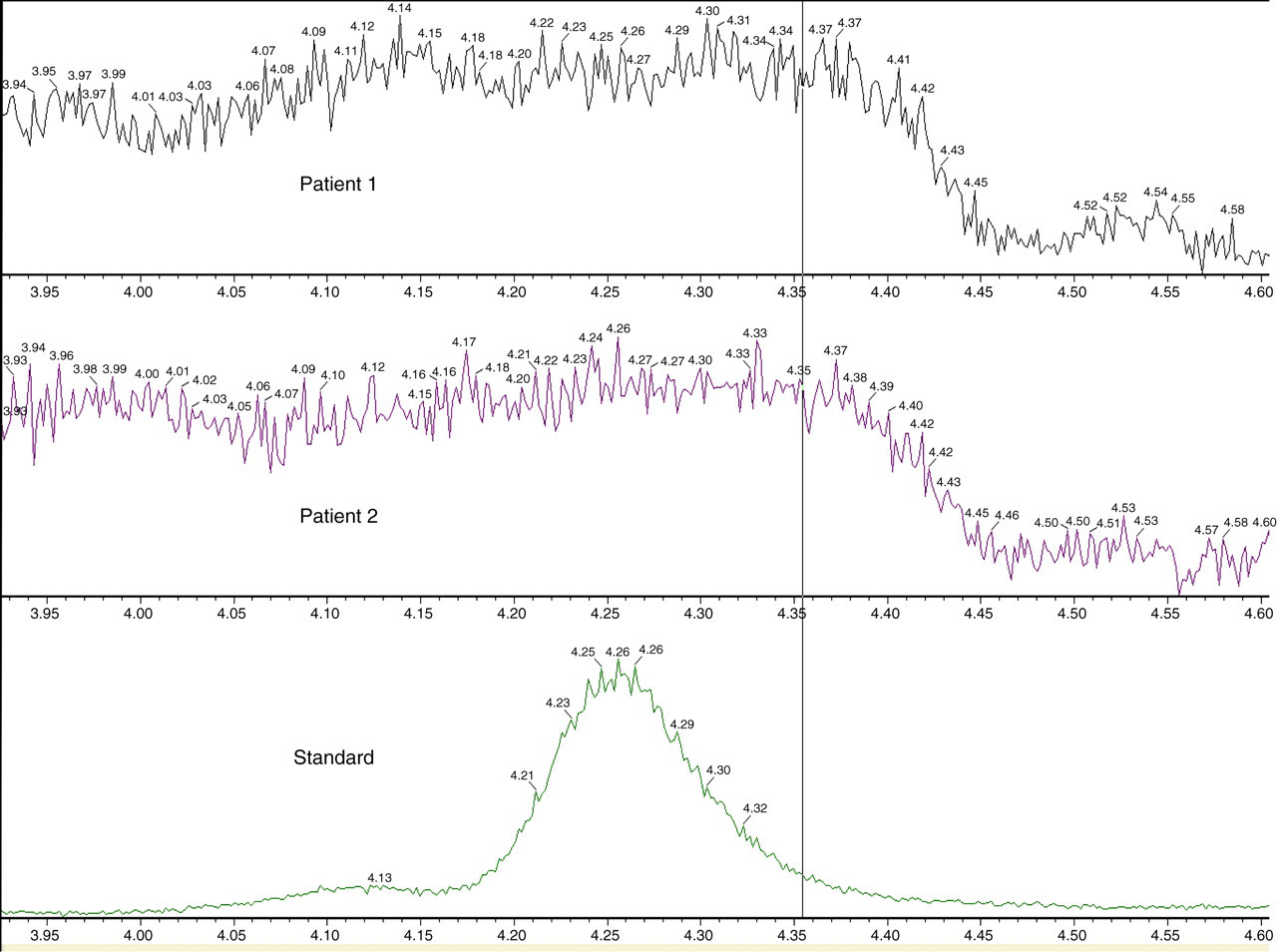
Postcolumn infusion of pure metadrenaline during injection of two patients' extracts, with a standard below showing retention time. This illustrates the separation of an ion suppression region from the elution of the analytes
A patient comparison (106 samples) between the LCMS/MS method and the HPLC/ECD method showed excellent agreement (R 2 = 0.99, y = 1.0706x − 63.281 and R 2 = 0.99, y = 0.8898x − 32.702 for metadrenaline and normetadrenaline, respectively) as depicted in Figure 3. Deming regression was used for method comparisons. The reference ranges used for this assay are <660 pmol/L for normetadrenaline and <300 pmol/L for metadrenaline (n = 122).
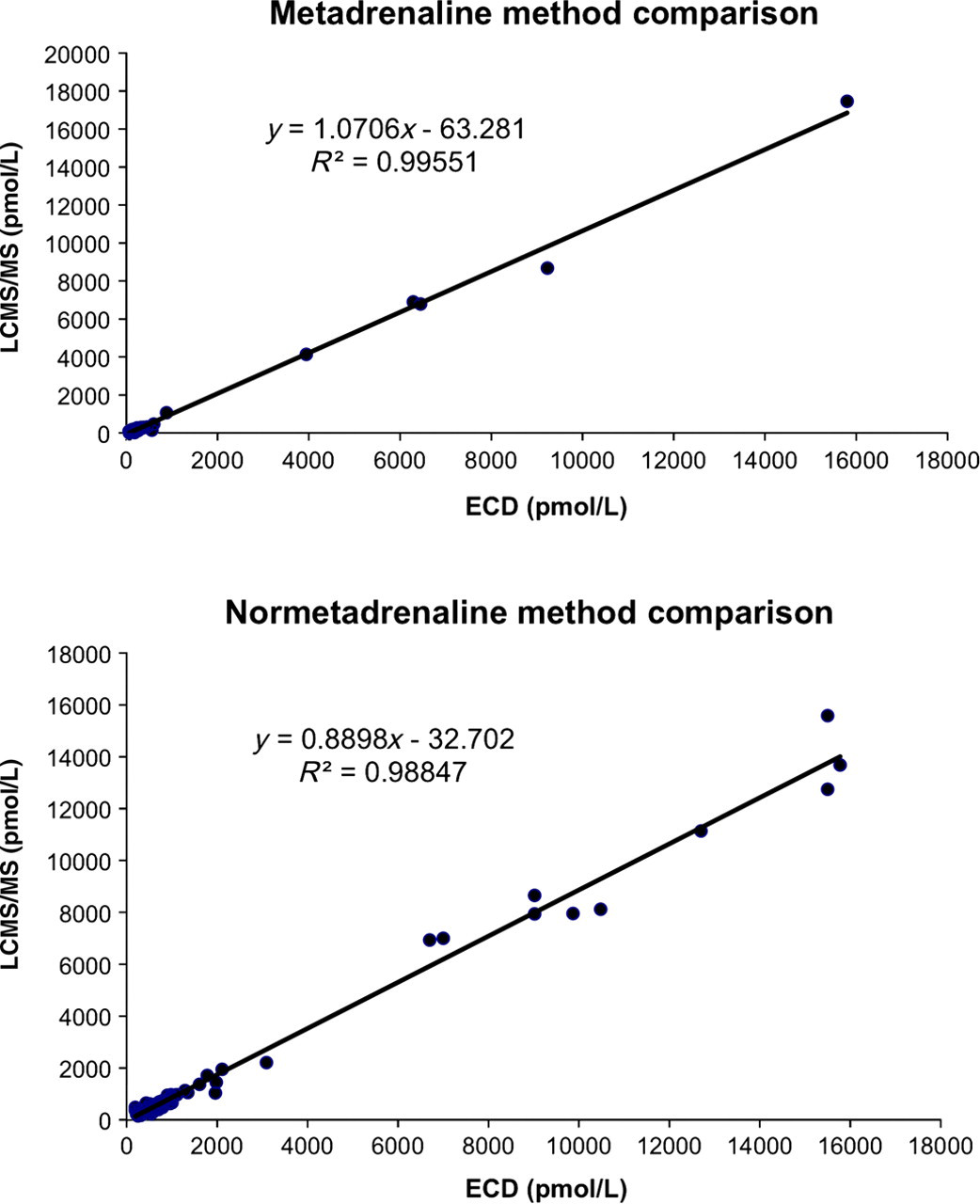
Method comparison showing excellent agreement between the previous HPLC with ECD method versus the new LCMS/MS method. ECD, electrochemical detection; HPLC, high-performance liquid chromatography; LCMS/MS, liquid chromatography tandem mass spectrometry
External quality assurance samples (RCPA QAP) were also assayed and showed excellent agreement with the median concentrations for both normetadrenaline and metadrenaline (Figure 4).
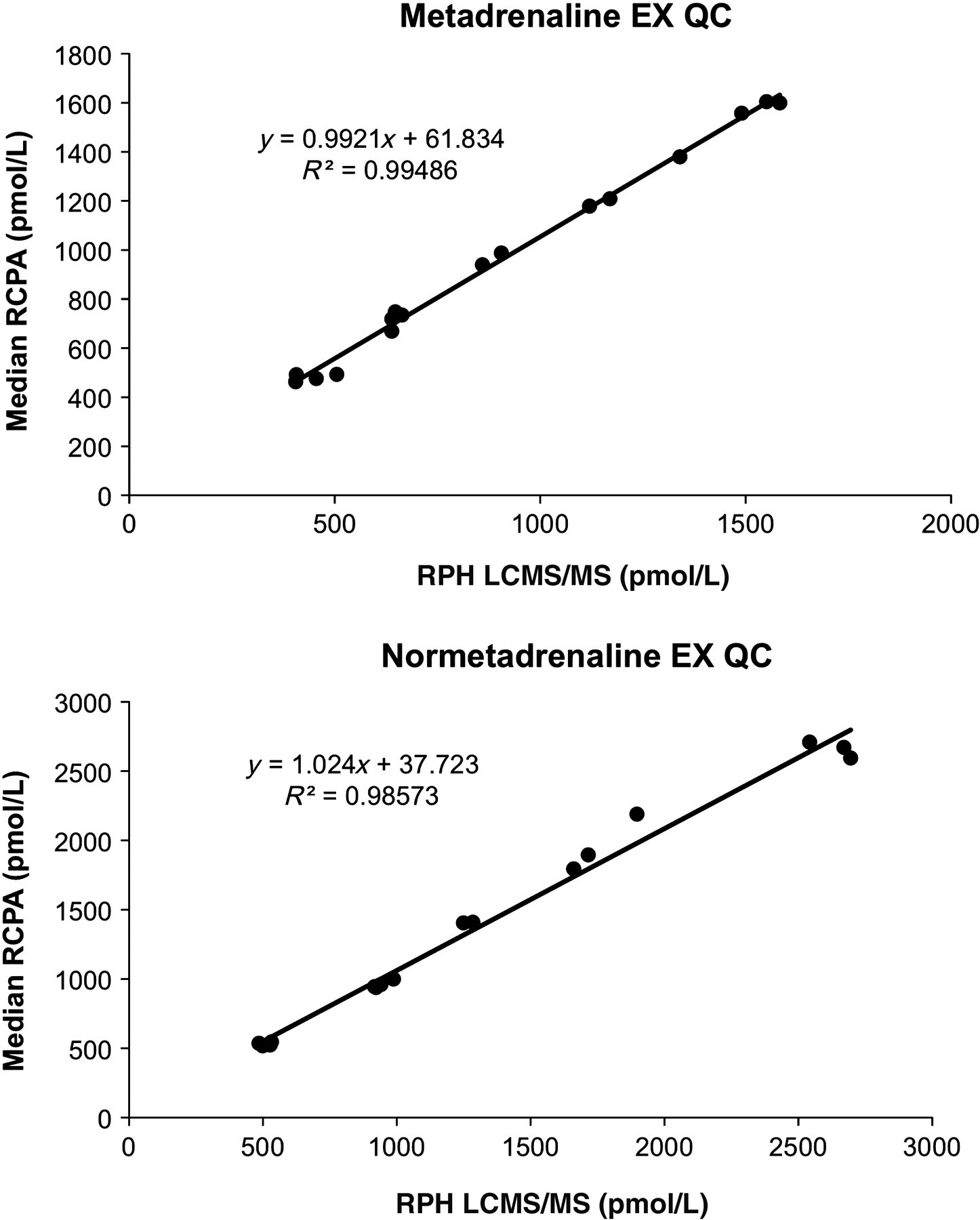
Comparison of median Royal College Pathologists Australia Quality Assurance Program samples versus our new LCMS assay for metadrenaline and normetadrenaline. This involved 15 laboratories and five of these used LCMS/MS methods. LCMS/MS, liquid chromatography tandem mass spectrometry
To ensure stability of the two compounds being assayed, samples were collected onto ice and frozen within four hours of collection. Samples assayed both on the day of collection and again up to 32 d later showed no change in both normetadrenaline and metadrenaline concentrations (n = 20). Furthermore, extracted samples were also stored in the autosampler and show no deterioration after 72 h at room temperature (n = 30).
Investigation into carryover effects were performed during each assay and did not demonstrate any evidence of increased background signal on blank samples.
Signal response linearity was confirmed by serial dilution of patient samples with stripped serum giving R 2 = 0.99 with an intercept at the origin.
To complete the method validation, we examined 12 patient samples that showed considerable interference with the HPLC/ECD assay and in all cases both normetadrenaline and metadrenaline could be quantified with no interference using LCMS/MS.
Discussion
The MCX SPE technique with LCMS/MS detection represents a sensitive and extremely accurate method to measure plasma free metadrenalines and is a suitable replacement, in our opinion, for the less specific and more laborious assays traditionally used to investigate patients with presumed phaeocromocytoma. The importance of pre-analytical preparation and the differing results of SPE methodologies is likely to be appreciated by more laboratories as they adopt sophisticated methods of analysis using tandem mass spectrometry.
The measurement of urinary deconjugated metanephrines using LCMS/MS has been previously described. 11,13–16 Plasma catecholamines and their metabolites have also been measured using a time-of-flight instrument. 17 Previous assays measuring plasma free metadrenalines in the diagnosis of phaeochromocytoma include enzyme immunoassays 15 and HPLC methods with ECD. 11 More specific LCMS/MS based assays have also been published using Oasis HLB SPE clean up, 18 using LCMS/MS with an automated online SPE system 19 and more recently, using a 96-well microtitre plate format and WCX sample clean up. 20
Inferior chromatography was observed when using WCX SPE cartridges compared with the MCX cartridges. The LCMS/MS assay showed improved precision, recovery and accuracy compared with HPLC/ECD and other methods employed within Australia. No interfering peaks have been observed during the routine clinical use of the LCMS/MS assay. The sensitivity of the LCMS/MS assay may enable adaptation to a microtitre plate format with reduced sample volume.
The use of the MCX SPE technique with LCMS/MS detection provides improved assay performance in the measurement of plasma free metadrenalines in comparison to HPLC methods and may prove to be superior in comparison to many available alternative methods, such as HLB and WCX pre-analytical preparation methods. The MCX SPE method with LC/MSMS detection also resolves most of the limitations of HPLC/ECD, allowing higher throughput analysis, increased sample batch numbers, less sample re-runs and improved turnaround times.
DECLARATIONS
