Abstract
Background
Currently, no clear guidelines exist for the most appropriate tests to determine sample quality from centrifugation protocols for plasma sample types with both lithium heparin in gel barrier tubes for biochemistry testing and citrate tubes for coagulation testing.
Methods
Blood was collected from 14 participants in four lithium heparin and one serum tube with gel barrier. The plasma tubes were centrifuged at four different centrifuge settings and analysed for potassium (K+), lactate dehydrogenase (LD), glucose and phosphorus (Pi) at zero time, poststorage at six hours at 21 °C and six days at 2–8°C. At the same time, three citrate tubes were collected and centrifuged at three different centrifuge settings and analysed immediately for prothrombin time/international normalized ratio, activated partial thromboplastin time, derived fibrinogen and surface-activated clotting time (SACT).
Results
The biochemistry analytes indicate plasma is less stable than serum. Plasma sample quality is higher with longer centrifugation time, and much higher g force. Blood cells present in the plasma lyse with time or are damaged when transferred in the reaction vessels, causing an increase in the K+, LD and Pi above outlined limits. The cells remain active and consume glucose even in cold storage. The SACT is the only coagulation parameter that was affected by platelets >10 × 109/L in the citrate plasma.
Conclusion
In addition to the platelet count, a limited but sensitive number of assays (K+, LD, glucose and Pi for biochemistry, and SACT for coagulation) can be used to determine appropriate centrifuge settings to consistently obtain the highest quality lithium heparin and citrate plasma samples. The findings will aid laboratories to balance the need to provide the most accurate results in the best turnaround time.
Introduction
Choice of optimum centrifuge settings for blood samples (force and duration) plays a pivotal role for sample quality, accuracy and turnaround time (TAT) of results. It is estimated that pathology data influence up to 80–90% of clinical decisions. 1 It is well documented that the preanalytical phase is the predominant phase contributing to pathology errors. 2 Centrifuges perform one of the most critical steps in the preanalytical phase to separate cells and serum or plasma. High-quality plasma or serum, in this context, can be defined as sample totally free of blood cells, cellular content and microclots. The establishment of the centrifuge protocols is usually left to laboratories as there is no definite information on the settings or suitable tests to use in checking sample quality. As a consequence, settings may vary between routine samples and urgent or ‘stat’ samples.
Centrifugation protocol studies for biochemistry lithium heparin plasma and serum samples range from those recommended by tube manufacturers, e.g. BD 1100g and 10 min for both sample types; 3 Greiner for lithium heparin tubes at 2200g for 15 min and 1800g for serum tubes, 4 to those used by laboratories which are as high as 13,600g for two minutes. 5 The Clinical and Laboratory Standards Institute guideline with respect to centrifugation of biochemistry samples recommends tube manufacturers' literature be consulted and the use of temperature-controlled centrifuges to prepare samples for temperature-sensitive analytes (e.g. potassium [K+], adrenocorticotropic harmone). 6
What is often not considered in choosing centrifuge protocols is the requirement to keep specimens for re-testing or reflective testing, which could be days later and the impact on sample quality from prolonged storage. In fact, the guidelines in Australia require biochemistry specimens be kept for up to seven days. 7 In other countries, guidelines stipulate biochemistry samples be kept for a minimum period of 48 h. 8,9 In all laboratories, samples are kept for up to eight hours at room temperature before being stored at 2–8°C. There are a number of published studies on analyte stabilities in uncentrifuged serum samples. 10–12 Apart from a study by Bailey, 13 our searches failed to find any recent studies looking at stabilities of analytes over time and temperature in lithium heparin plasma tubes with gel barrier. Lithium heparin plasma is a more complex matrix than serum. When the lithium heparin plasma is collected in gel separator tubes, the plasma will always contain some blood cells and cell matter and a ‘buffy-coat-like layer’ between the gel barrier and the plasma, 14 which renders the plasma less stable for prolonged storage.
Another aspect of centrifuge setting evaluations for biochemistry analysis is the use of large analyte panels with duplicate testing immediately after centrifugation to look for clinical significant changes to evaluate sample quality. 15 One such study has even performed triplicate analysis. 16 This rarely provides additional information that a select few analytes cannot provide.
Unlike biochemistry guidelines for centrifugation settings, there is a specific guideline for centrifugation settings for plasma-based coagulation studies. Citrate plasma must be consistently platelet-poor, i.e. platelet count <10 × 109/L, and swing-out bucket rotor centrifuges are recommended to minimize contamination of the plasma with platelets and other cells. 17 This will optimize sample quality for routine coagulation tests such as the activated partial thromboplastin time (APTT), prothrombin time/international normalized ratio (PT/INR) ratio and thrombin clotting time. Additionally, samples for routine lupus anticoagulant testing 18 or samples to be stored frozen for referral must be rendered platelet-free not only platelet-poor, 19 prior to testing. The latter is particularly important to avoid activation issues with thawed coagulation samples. The limitation of these guidelines is that they do not outline what are the acceptable limits of change in clotting parameters or how to calculate them. Again, reported centrifuge settings used for coagulation testing range from 1500g for 10 min 20 to 11,000g for one minute, 21 and performing analysis in duplicate. 20
The objective of this study is to evaluate the influence of centrifuge settings with swing-out bucket rotor centrifuges on sample quality in lithium heparin plasma in gel separator tubes using K+, glucose, lactate dehydrogenase (LD) and inorganic phosphorus (Pi) over three different time periods (immediately postcentrifugation, 6 h post 21°C storage and post 6 days at 2–8°C), and citrated plasma using APTT, PT, INR, derived fibrinogen (FibD) and surface-activated clotting time (SACT) immediately postcentrifugation.
Materials and methods
Volunteers and sample processing
Blood samples were collected with consent, as approved by the Princess Alexandra Hospital Ethics Committee, from 14 healthy laboratory participants all >18 years of age, by an experienced phlebotomist using a BD Vacutainers Safety-Lok Blood Collection Set with 21G needle (REF 367286) into Greiner Vacuette® citrate tubes (Cat No. 454327), lithium heparin (Cat No. 456083) and serum (Cat No. 456078) tubes. The lithium heparin and serum tubes contained a gel barrier. The tubes were centrifuged in a StatSpin® Express 4 (SSE4) with fixed settings, and a Heraeus Multifuge 1S-R (HM). For the biochemistry study the following tubes were collected: a serum tube and four lithium heparin samples. The tubes were prepared as recommended by the manufacturer. A lithium heparin tube was centrifuged in each of these settings within 30 min postcollection: (a) three minutes, 4000g in SSE4; (b) 10 min, 3100g in SSE4; (c) 10 min, 3000g, 20°C in HM; and (d) four minutes, 6000g, 20°C in HM. The serum sample was centrifuged for 10 min, 3000g, 20°C in HM. For the coagulation study three citrate tubes were collected and centrifuged within 30 min of collection: (a) three minutes, 4000g in SSE4; (b) 10 min, 3100g in SSE4; and (c) 10 min, 3000g, 20°C in HM. Analysis was performed within two hours postcentrifugation.
To assess the effect of platelets on the SACT, two citrates were collected from a further 10 volunteers. One citrate was centrifuged at 100g for 10 min to obtain platelet-rich plasma (PRP), the other centrifuged at 3000g for 10 min to obtain platelet-poor plasma (PPP). Baseline platelet counts and SACT were performed on each PPP, and then the platelet count was slowly increased by addition of small volumes of the respective PRP and platelet counts and SACT recorded.
Analysis
The biochemistry analysis was performed on a Beckman DxC800 general chemistry analyser (Beckman Coulter, Fullerton, CA, USA), and the coagulation analysis was performed on an ACL TOPCTS (Instrumentation Laboratory, Lexington, MA, USA) using Thromborel S (Siemens Healthcare Diagnostics, Marburg, Germany) for the PT and FibD, TriniCLOT aPTT HS (Trinity Biotech, Wicklow, Ireland) and HemosIL 0.025 mol/L CaCl2 (Instrumentation Laboratory, Lexington MA, USA) for the APTT and VitaCLOT (Vital Diagnostics Pty Ltd, Bella Vista, NSW, Australia) and HemosIL 0.025 mol/L CaCl2 (Instrumentation Laboratory) for the SACT. Only single analysis was performed for all analytes. The mean and standard deviation for each analyte was calculated and reported.
The total analytical imprecision expressed by the between-run coefficient of variation (CV) for the analytes was as follows: K+ (1.6% at 3.9 mmol/L and 1.6% at 6.3 mmol/L), glucose (2.8% at 4.9 mmol/L and 2.7% at 15.7 mmol/L), LD (2.7% at 156 U/L and 2.3% at 432 U/L), Pi (3.2% at 0.99 mmol/L and 3.1% at 2.90 mmol/L), APTT (2.3% at 32.2 s and 1.5% at 81.7 s), PT (3.6% at 10.4 s and 3.1% at 39.0 s), INR (3.6% at 1.0 and 3.1% at 3.5), FibD (3.0% at 2.5 g/L and 2.6% at 1.6 g/L) and SACT (3.0% at 72 s and 5.6% at 127 s). The platelet count was performed on a Sysmex XT-2000i (Sysmex, Kobe, Japan).
Statistical analysis
The mean and standard deviation were calculated for each analyte at the different centrifugation setting and time delay. From the imprecision data the least significant change (LSC) (2.77√analytical coefficient of variation) was calculated to determine the maximum allowable difference in test results between centrifuge settings and time delay. 12 The platelet count and SACT mean and SD were calculated for the PPP/PRP mixtures and results were graphed using GraphPad Prism version 4 software (San Diego, CA, USA).
Results
The biochemistry test results are summarized in Table 1. The haemolysis index measured on the Beckman DxC800 analyser in all samples was <0.5 g/L haemoglobin. The data indicate that plasma is less stable than serum, and plasma sample quality is higher with longer centrifugation time, 10-min setting on the SSE4 centrifuge and at much higher g force with shorter time on the HM centrifuge. As expected over time, blood cells present in the plasma undergo lysis or are damaged when transferred in the reaction vessels, causing an increase in the K+, LD and Pi above the LSC limit. The LD showed changes in the plasma, increased above the LSC limit even post six hours storage at 21°C. In serum the LD activity decreased post six days storage at 2–8°C. This decrease is in line with published findings, serum LD activity, specifically due to fraction four instability falling rapidly in the first week at 0–4°C. 22 The cells remain active even in cold storage and consume glucose leading to changes outside the LSC limit post six days storage. The analytes selected demonstrated that the lithium heparin plasma quality was influenced by centrifugation settings and that they are suitable candidates to be used as indicators of sample quality.
Biochemistry data at various centrifugation settings and poststorage
LiHP, lithium heparin plasma; S, serum; SSE4, StatSpin® Express 4; HM, Heraeus Multifuge 1S-R; LSC, least significant change; LD, lactate dehydrogenase
Values in bold are outside the expected limits
The coagulation results are summarized in Table 2. The data clearly indicate that the SSE4 three-minute setting fails to meet the CSLI recommendations for producing citrate PPP with platelet counts <10 × 109/L. Apart from the SACT, with the SSE4 three-minute setting, no other analyte results were compromised when the platelet count was above the CLSI recommended limit. Besides the platelet count, the SACT appears to be a suitable parameter that can be utilized as an indicator of citrate plasma sample quality. To test the effect of increasing platelet count on SACT, we prepared 10 sample sets with increasing platelet concentrations. The relationship is clearly illustrated in Figure 1.
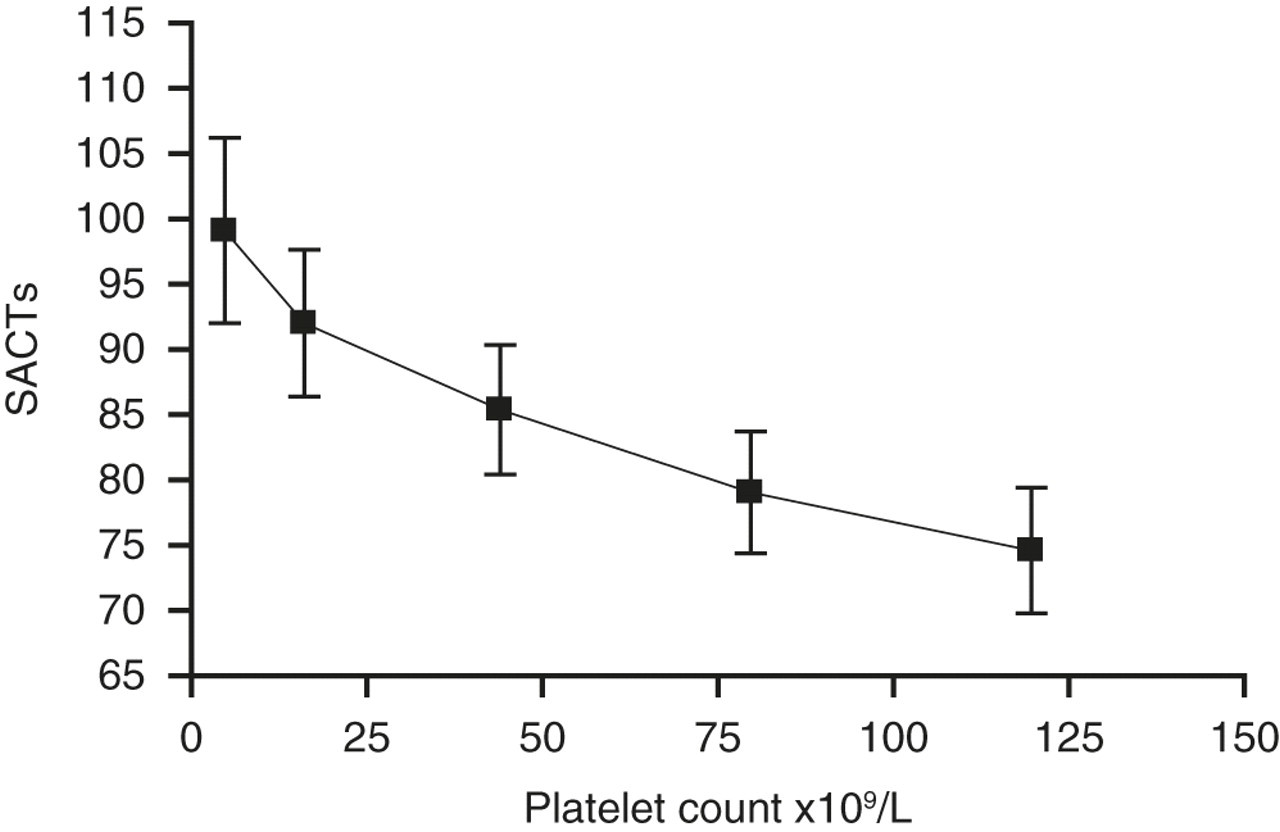
Relationship between platelet count and surface-activated clotting time (SACT)
Coagulation study data with citrated plasma analysed immediately after centrifugation
APTT, activated partial thromboplastin time; PT, prothrombin time; INR, international normalized ratio; FibD, derived fibrinogen; SACT, surface-activated clotting time; SSE4, StatSpin® Express 4; HM, Heraeus Multifuge 1S-R; LSC, least significant change
Value in bold is outside the expected limits
Discussion
It is well accepted that TAT and sample quality (error rates) are key indicators of the quality of laboratory services, particularly for urgent samples. According to Valenstein, 80% of complaints laboratories receive are about TAT, 23 and it is estimated that 87% of dissatisfaction complaints on TAT originate from emergency departments. 24 Despite such complaints, sample quality and analytical reliability must not be compromised by use of reduced centrifugation times to meet TAT demands.
Although plasma offers improved TAT of results for biochemical tests by ∼30 min, it needs to be treated with caution for reflective and re-testing post prolonged storage. Our data showed that the more complex plasma sample will continue to exhibit changes in biochemical analytes more so than serum, due to the presence of blood cells and cellular content. Thus, unless TAT is critical and patients are unlikely to be anticoagulated or samples are unlikely to be taken from vascular catheters that can be grossly heparin contaminated, serum in tubes containing gel barrier offers greater analyte stability upon prolonged storage at 2–8°C. This is also likely to hold true for storage of samples at −20 or −80°C, where cells present in the sample will be lysed during the freezing process and can alter sample composition.
The biochemical analytes that have been selected for this study, K+, glucose, LD and Pi are very sensitive to cellular influence. These three analytes are present in red cells in much greater concentrations than in the plasma: K+ ∼25 times, 25 Pi ∼7 times 10 and LD 4000 times. 26 They are also present in the cytoplasm of all blood cell types and in much greater concentration than in the serum or plasma. Blood cells present in the plasma will continue to be active and consume glucose, releasing lactate, etc. In a tertiary-level hospital like ours, there is a very high demand for short TAT for critically ill patients. Additionally, a very large percentage of these patients, up to 10%, are on anticoagulant therapy; 27 hence lithium heparin plasma is the preferred sample type for biochemistry analysis for patients from critical care units.
Similar TAT issues are relevant to coagulation testing. Previously, we found no significant differences in the PT/INR, APTT and FibD when high-speed (4440g), short-time (2 min) centrifugation is used to improve the efficiency for urgent requests. 28 As with biochemistry samples, there are a variety of citrate preanalytical sample handling issues, including the citrate whole-blood storage time and temperature, prior to centrifugation, centrifugation time and temperature, centrifuged sample storage or aliquot freeze/thawing, which may impact on result accuracy. However, various centrifugation protocols rarely show any clinically significant differences in these parameters unless the sample is stored for more than four hours prior to analysis. 29–31 Therefore, when assessing centrifuge protocols, these routine coagulation parameters are less than ideal for assessing sample quality.
The SACT uses a phospholipid-free suspension of an aluminasilicate mineral to activate the contact factors in an APTT-like assay. The low phospholipid content of this reagent makes it very useful for detecting lupus anticoagulants (LA) in platelet-free samples. 32 We hypothesized that the SACT result would decrease as platelet count increases, in the same way confirmatory tests with high phospholipid content tend to normalize in LA diagnostic test panels. 18 As shown in Table 2 and Figure 1, there is an inverse relationship between sample platelet count and the simple SACT, which can be a useful marker of sample quality.
Obtaining a highest sample quality ensures that the results and other laboratory data integrity are more reliable for patient care decisions and reduces unfavourable outcomes for patients and health providers. Since each laboratory is required to perform its own centrifugation studies, the results of this study indicate that a small number of laboratory parameters can be used to determine appropriate centrifuge settings. K+, glucose, LD and Pi are suitable for biochemical testing of lithium heparin plasma and the platelet count, and SACT are suitable for citrate plasma for coagulation testing to ensure the highest quality sample is obtained.
The SSE4 data indicated that the 10-min duration setting would render the centrifuge suitable for small laboratories or where separate centrifugation is taking place of urgent samples. Despite the obvious TAT benefit offered by the three-minute SSE4, our data suggest that sample quality is not optimal under these conditions. The data indicate that centrifuges and settings can be shared to prepare samples for biochemistry and coagulation testing. Although this study has limitations such as not evaluating the SSE4 five-minute setting, it is designed to provide data on analytes that would be most appropriate for laboratories to utilize in their own studies.
In summary, rapid TAT and having accurate results is equally important in patient safety and reducing inappropriate utilization of scarce resources by health-care systems. Accurate results can only be obtained if laboratories perform analysis on the highest sample quality, which in part is influenced by centrifugation settings. The study clearly illustrates, for plasma samples, that a limited but sensitive number of assays can be used to determine appropriate centrifuge settings to consistently obtain the highest quality lithium heparin and citrate plasma samples for biochemistry and coagulation testing, respectively.
DECLARATIONS
