Abstract
Hypogonadism in the male results from inadequate testicular function, especially defects in androgen synthesis and secretion, or action. Androgen action is important throughout normal male development: in the fetus, puberty, adult life and old age. Regulation is by variable activity of the hypothalamo-pituitary axis at different phases of the life span. Clinical aspects include: genetic aspects presenting at birth and pubertal failure/arrest. Aspects in adult life embrace sexuality, somatic symptoms and osteoporosis. Acquired causes of hypogonadism may arise from various forms of testicular damage (primary hypogonadism), pituitary and hypothalamic disorders, as well as aetiologies acting at several sites. Measurement of testosterone (T) is crucial to the diagnosis of hypogonadism and the technologies continue to develop, with recent major advances. A growing problem relates to the diagnosis and treatment of hypogonadism in the ageing male. T therapy is available in several forms, with major improvements in more newly available modalities.
Introduction
Male hypogonadism results from inadequate testicular function. This may be significant at all phases of life, from sexual differentiation in fetal life, through puberty to adult life, involving virilization, fertility, anabolic responses, psychological effects and finally the changes in old age. 1 Androgens, particularly testosterone (T) and dihydrotestosterone (DHT) play a key role, but other relevant testicular hormones and paracrine agents include Műllerian inhibitory hormone, inhibin B and the activins. 2
Regulation of testicular function involves the pituitary gonadotrophins, follicle-stimulating hormone (FSH) and luteinizing hormone (LH), which in turn are controlled by the pulsatile release of gonadotrophin-releasing hormone (GnRH) from the medial-basal hypothalamus, reaching the pituitary gland via the hypothalamo-hypophyseal portal blood vessels along the pituitary stalk. 3
Androgen synthesis and action
T, the major androgen, is largely produced by the Leydig cells of the testes (3.7 ± 2.2 mg/d in adult men), 4 less than previous estimates, up to 7 mg/d. 5 The recent lower estimates derive from the use of stable labelled compounds, analysed under steady-state conditions. The principal substrate for its synthesis is from cholesterol esters stored in fat vacuoles of the Leydig cells. 6 The initial, rate-limiting step in bio-synthesis involves a three-stage side-chain cleavage (ssc) on the mitochondrial inner surface yielding pregnenolone. The crucial enzyme, cytochrome P450ssc or 20,22 desmolase, is driven by LH. 7 T biosynthesis can take two pathways; that preferred in men, Δ5, has the double bond between carbon atoms 5 and 6 in the B ring of the steroid molecule, whereas in the Δ4 pathway the double bond is in the A ring between carbon atoms 4 and 5. 8
These subsequent steps take place in the endoplasmic reticulum. The weak Δ5 androgens are also synthesized by the adrenal cortex and small amounts peripherally converted to T. Peripheral conversion of T to the more potent DHT occurs especially in target tissues: the external genitalia, prostate and androgen-dependent hair follicles, catalysed by 5α-reductase. 9 T is also converted to oestradiol by aromatase, which is of major importance in securing epiphyseal fusion of long bones, and for some actions of T in the central nervous system.
Physiological role of androgens throughout normal male development
Fetal and early postnatal life
The classic studies of Alfred Jost in rabbits more than 50 years ago have provided the foundation for the understanding of sexual differentiation applicable to most mammals including man. 10 The initial stage of sex determination is genetic, with the primary role of the Y chromosome, on which is situated the SRY gene – necessary, but not sufficient for turning the early bipotential gonad into a testis (numerous other genes downstream are also implicated). 11 By a carefully timed series of supremely skilful experiments including gonadectomy, gonadal transplants and implants of crystals of T, he demonstrated that the default position was the female phenotype, to which the ovary made no obvious contribution as it was seen in genetically normal males castrated early, before the differentiation of the testes. 12 This is characterized by the persistence of Műllerian duct derivatives, the uterus and Fallopian tubes. The male phenotype involves the activity of the differentiated testis's hormonal secretions namely Műllerian inhibitory hormone leading to the failure of such products to grow into the mature organs of uterus and Fallopian tubes, and shortly thereafter T acting in a paracrine fashion to promote the development of the Wolffian duct system. 13
Much later in pregnancy, there is the important further masculine differentiation of the external genitalia mediated by the local production of DHT from T by 5α-reductase, leading to scrotal fusion, phallic development and testicular descent as well as the differentiation and growth of the prostate and other accessory sexual organs (Figure 1). 14 These late testicular endocrine actions are at least in part driven by fetal LH secretion and are continued for a few months into postnatal life, before the early to mid-childhood inhibition of the hypothalamo-pituitary-testicular axis supervenes.
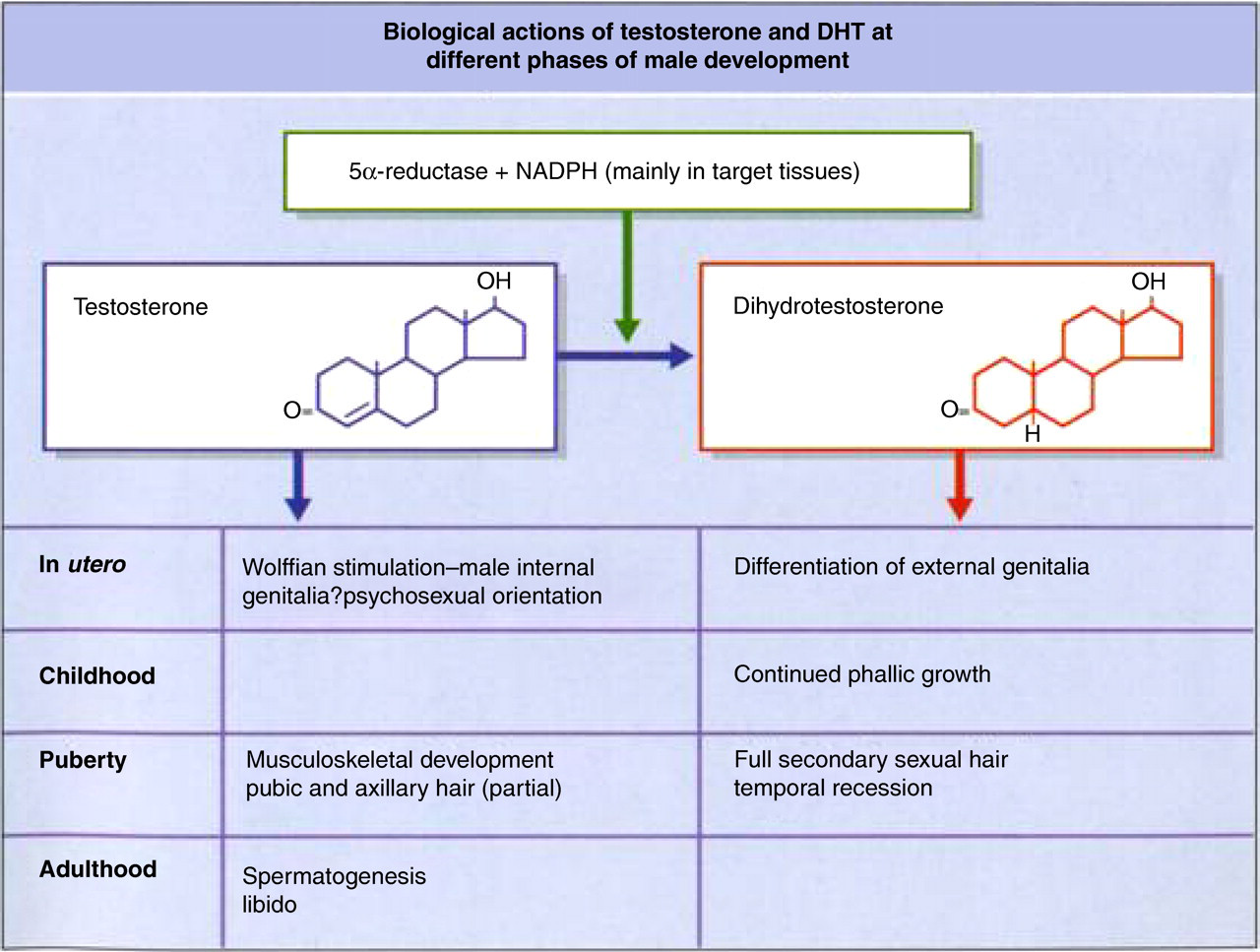
Roles of testosterone and DHT at different phases of male lifespan. DHT, dihydrotestosterone
Puberty
The events of puberty involve re-awakening of the pulsatile pattern of GnRH release and consequent gonadotrophin secretion, initially with a predominantly FSH output. This then reverses to primarily nocturnal LH pulses, later extending throughout the whole 24-h period by the end of puberty (Figure 2). 15 Despite this, a circadian rhythm of T secretion persists: greatest in the morning, falling to a nadir in the afternoon and evening. 16 Mechanisms initiating puberty are not fully understood, but involve genes such as Kisspeptin and somatic signals such as leptin, pineal gland activity and alteration in the balance of inhibitory neurotransmitters such as γ-amino-butyric acid and neurostimulators such as glutamate. 17 There are significant differences between the sexes in the course of puberty. In boys the onset is generally about a year later than girls and the growth spurt is near its end, associated with massively augmented growth hormone secretion. 18 The clinical stages through puberty in boys are charted according to Tanner for patterns of genital development (the earliest sign being growth of testes to 4 mL) and pubic hair growth. 19 Increases in T and DHT levels play important roles. 1
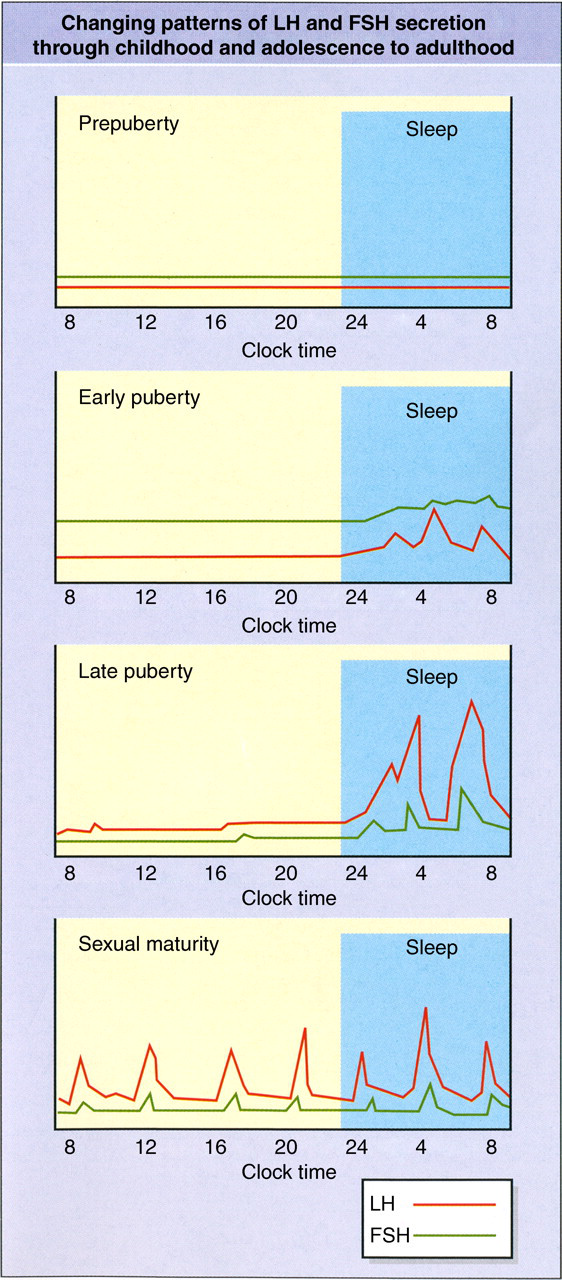
Evolving patterns of pulsatile secretion of LH and FSH from childhood through puberty to adulthood. LH, luteinizing hormone; FSH, follicle-stimulating hormone. Please see online for colour version
Adult life
Active pulsatile, GnRH-driven LH and FSH secretion is essential throughout adult life for fully maintained secondary sexual characteristics and function. 20
Virilization
Acquisition of the masculine habitus, with marked muscular development, is largely dependent on T secretion. Patterns of facial, axillary, pubic and body hair growth are determined both by T and also by peripheral conversion to DHT. Genetic and ethnic variations occur in androgen-dependent patterns of hair growth (and baldness). 21 Variations may derive from differences in local 5α-reductase or androgen receptor responsiveness. The latter is often related to the length of a highly polymorphic polyglutamine tract in the androgen receptor, encoded by CAG repeats in exon 1 of the androgen receptor gene, with increasing numbers associated with lower androgen action. 22,23
Other somatic effects of T
The skeletal effects of T affect ultimate height by epiphyseal fusion (mediated via aromatase conversion of T to 17β-oestradiol), also bone accretion and maintenance through anabolic actions and antiresorptive effects. 24 T positively influences the haemoglobin concentration and haematocrit–men have a higher incidence of ischaemic heart disease and more adverse lipid profiles than women, suggesting that T is atherogenic. Recent work has challenged the concept that T is atherogenic, as over simplistic. 25
Sexual function
T is important for many aspects of sexual function, including fertility, probably due to paracrine effects of the high ambient intratesticular T concentration in the healthy adult male. 26 Prostate function is dependent on local DHT generation. 27 Other important T effects include libido as well as such other psychological domains as spatial sense and (as yet not fully resolved) aggression. 28 T is an essential component for erectile function. Recent animal studies have demonstrated a neural circuit in the lumbar spinal cord with T-responsive, gastrin-releasing peptide neurones projecting to the lower lumbar regions involved in erection and ejaculation. 29
Clinical aspects of hypogonadism
Genetic influences presenting at birth
Early failure of T production from the fetal testis (9th to 14th week of gestation), or failure of androgen receptor response, lead to failure of sexual differentiation. Causes include several, recessively inherited mutations of critical enzymes involved in steroid biosynthesis affecting T and sometimes adrenocortical function. They include 3β-hydroxysteroid dehydrogenase II deficiency, 17α-hydroxylase/17,20-lyase deficiency and 17β-hydroxysteroid dehydrogenase type 3 deficiency in 46,XY individuals. 30 The degree manifest varies from the most extreme with a female phenotype, having poor phallic development, unfused labia with perineal urethral opening and testicular mal-descent. Műllerian inhibitory hormone being unaffected leads to involution of the Műllerian structures, hence there is a blind ending vagina. In the complete forms there is absent secondary sexual hair, full breast development, but the nipples are rudimentary. 31 Height is often tall owing to the action of growth-related genes on the Y chromosome. 32 Lesser degrees of T failure of production or insensitivity are linked with a states of intermediate or ambiguous sexual phenotypes ranging from some virilization up to just minor degrees of hypospadias or some forms of idiopathic male infertility. 33
Deficient T production later in pregnancy up to the 24th week of gestation may cause micropenis and abnormal testicular descent. An interesting variant is caused by 5α-reductase deficiency leading to impaired DHT production. At birth, these genotypic males have ambiguous external genitalia, clitoral-like phallus, bifid scrotum, blind vaginal pouch and inguinal or labial testes (Figure 3). Raised as girls, at puberty they virilize with T-related development of male habitus and muscularity, testicular descent and spermatogenesis, but have markedly reduced facial hair, acne and frontal hair recession. They develop male psychosexual characteristics after puberty. In a highly consanguineous community in the Dominican Republic where this gene mutation is common, most affected people switched at puberty to a male gender role. 34 The process was accepted as normal and because of the concomitant phallic growth was termed ‘guevedoces’: penis at 12.
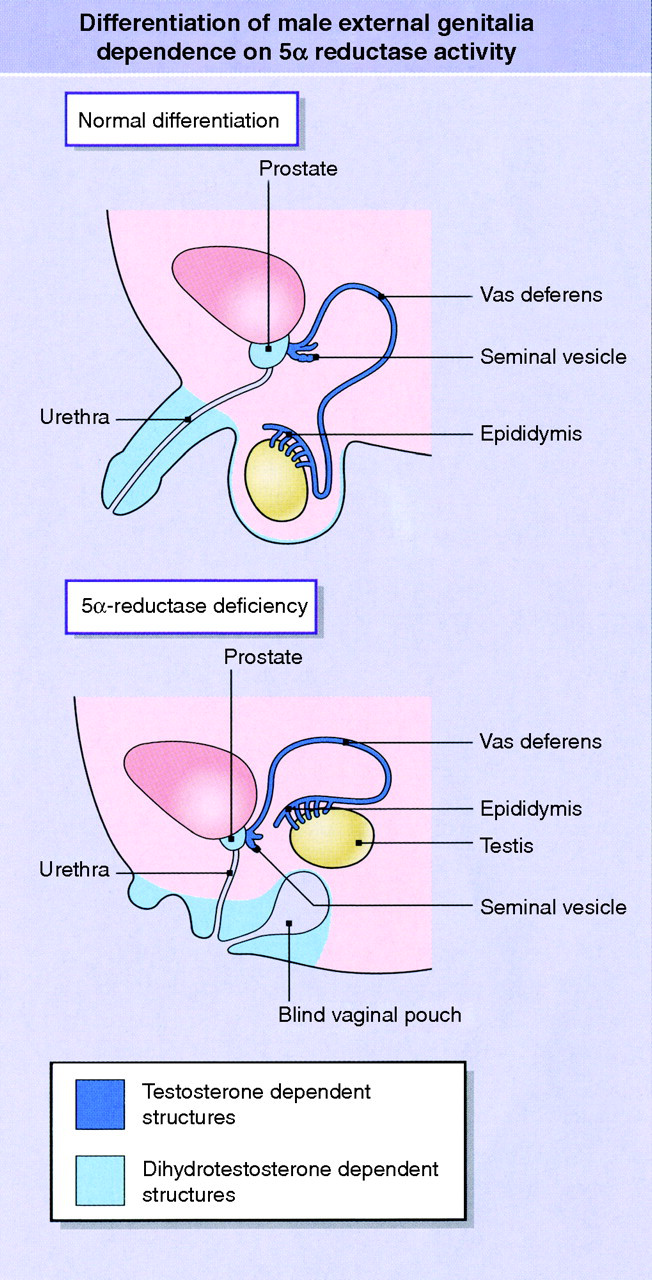
Anatomical consequences at birth of 5α-reductase deficiency. Please see online for colour version
The opposite situation is seen in the persistent Müllerian duct syndrome, usually due to either mutations in genes coding for anti-Müllerian hormone production or the hormone receptor. 35 These individuals are unequivocally phenotypically male and virilized, though, as well as possessing a uterus and Fallopian tubes, have defective testicular descent. The majority of affected subjects are infertile.
Pubertal failure or arrest
Since childhood is largely a period of sexual quiescence, unless there are syndromes with characteristic somatic or developmental abnormalities, the next stage at which hypogonadism manifests is when the normal pattern and timing of puberty fails to occur. 36 This may be subtle in boys, whereas in girls the early features of breast development and growth acceleration (or their absence) are conspicuous, while menarche is a landmark event, albeit late in the process. The hypogonadal boy's growth often falls behind that of his peers as theirs accelerates, but if the underlying problem persists, the long bones continue growing, leading to the span exceeding the standing height by 5 cm or more (since the arms are held outstretched in series, in contrast with the parallel position of the legs). This constitutes eunuchoidal proportions. Eventually the failure of virilization becomes obvious, often leading to loss of confidence and self-esteem before medical referral.
Hypogonadism in adult life
The principal features relate to sexual function, general somatic symptoms, especially fatigue or weakness, and osteoporosis. The spectrum is wide, varied in severity and speed of onset, and related to the underlying cause. Acquired hypogonadism in adult life may have widely differing effects on secondary sexual characteristics and performance.
Sexual function
The chief consequence of hypogonadism is loss of libido, often associated with erectile dysfunction. The latter, however, is commonly not overtly associated with endocrine dysfunction, although subtle degrees of hypogonadism are increasingly frequently being diagnosed. 37 Secondary psychogenic components, such as depression and anxiety, often exacerbate the clinical situation and require sympathetic handling. 38 Intercurrent medical conditions may add further aspects as well as causing the sexual problems. Men with acquired hypogonadism may experience hot flushes and vasomotor disturbances like postmenopausal women. These may be severe, especially if the cause is of rapid onset. 39 Hypogonadism may be discovered during investigation of infertility and may be surprisingly asymptomatic.
General somatic symptoms
Fatigue and weakness are common in hypogonadal men, arising from many causes. These include specific effects of low T, in part mediated by the loss of skeletal muscle mass and strength, combined with increased body weight, particularly central obesity. Normocytic anaemia may also contribute. Vasomotor symptoms and sleep apnoea often disrupt sleep patterns. This commonly occurs in hypo-gonadism and problematically may worsen with T substitution therapy. 40 Depression can be very debilitating and partly come from awareness of loss of body and facial hair, muscle bulk and height from kyphosis. The skin typically becomes sallow and thin, with multiple fine facial wrinkles especially vertically oriented above the upper lip. 41 Many of these features are accompaniments of ageing, hence the debate about the diagnosis and management of hypogonadism in older men.
Osteoporosis
As the male population lives longer, the incidence of osteoporosis rises and the role of hypogonadism is probably underestimated. 42 In the late 1980s after the installation of one of the first dual-energy X-ray absorptiometers in Leeds, it was hard to obtain normative male bone density data. This was helped by university students recruiting their fathers. Those who had low readings were invited to be further investigated. A substantial number were found to be clearly hypogonadal, despite presumed paternity, often with remarkable paucity of classical clinical features. This remains anecdotal in the absence of reliable data on the percentage of cases attributable to hypogonadism, despite the large body of recent literature addressing T concentrations in the ageing male (see below). A number of studies have found that the closest hormonal correlate of bone mass is with the bio-available oestradiol concentration (most of which is derived from aromatization of T). 42
Causes of male hypogonadism
It is conventional to classify the underlying causes into a primary group, where the defect lies at the testicular level, and secondary causes arising from deficient secretion or action of pituitary gonadotrophins. A number of conditions, however, may act at more than one site to produce hypogonadism; these are often chronic, multisystem diseases, but also include the increasingly recognized age-related changes in male gonadal function.
Primary hypogonadism
Trauma
Dating back into prehistory, castration was used for various religious, punitive and social purposes and there remain indications in the management of prostate cancer, testicular malignancy (which may be bilateral), Fournier's gangrene and male-to-female gender reassignment. 43 Testicular damage can also arise from vascular causes such as testicular torsion, or iatrogenically in the attempted surgical correction of cryptorchidism and as an inadvertent consequence of inguinal hernia repair.
Drugs
Alkylating agents such as cyclophosphamide, busulphan and chlorambucil are notoriously damaging to seminiferous tubules, causing infertility, rather than causing impaired hormone production. These effects are dose-dependent, with high doses leading to permanent azoospermia. Other cytotoxic drug types are much less harmful, usually leading, at most, only to transitory suppression of spermatogenesis. 44 Drugs that predictably interfere with androgen synthesis include aminoglutethimide, etomidate and ketoconazole. Drugs blocking action at the androgen receptor include spironolactone, cyproterone acetate and flutamide. 45
Ionizing radiation
This is very toxic to the germinal epithelium, resembling the effects of alkylating drugs. 46 Radioiodine, in the doses used for treatment of thyrotoxicosis, has not been associated with infertility, at most just short-lived depression of the sperm count. High doses used in patients with differentiated thyroid cancer are probably more likely to cause adverse effects, especially with repeated administration. 47
Chromosomal disorders
Klinefelter's syndrome is typically caused by a 47 XXY karyotype, while less severe features may be seen with 47 XXY/46 XY mosaicism and much more severe with increasing supernumerary X chromosomes. Classically there are small firm testes with markedly hyalinized, atrophic seminiferous tubules and azoospermia. Testicular biopsies may recover occasional spermatozoa, at least in younger adults.
Patients are often tall and display feminized fat distribution and gynaecomastia as well as eunuchoid proportion, but others are thin and narrow chested, while some may look no different from normal apart from their small testes. Klinefelter's syndrome is a recognized cause of leg ulcers in men. Diagnosis prepubertally is rare – sometimes it is suspected on the basis of problems in psycho-social development or long limbs. Puberty may fail to develop or undergo early arrest, but commonly normal T concentrations are seen in early adult life, which then decline markedly with the development of hypogonadism after a decade following completion of puberty. The phenotypic picture is also influenced by the number of CAG triplet repeats in the androgen receptor gene. 48
Down's syndrome (trisomy 21) is usually associated with infertility not hypogonadism.
Elevated testicular temperature
Persistent failure of descent of the testes into the scrotum is associated with infertility, attributed to the raised temperature within the abdominal cavity, since early surgical correction improves the chances of fertility. Furthermore the intra-abdominal site predisposes to malignant transformation in cryptorchid testes. Other factors may be operative as in cases of unilateral cryptorchidism, there are often abnormalities noted in the scrotal testis. 49
Varicocele is often blamed for subfertility or mild feminizing clinical features such as gynaecomastia, where again the mechanisms suggested have included the reduction in abdomino-scrotal temperature gradient to 0.1°C from a normal difference of 2.2°C. 50 Abnormalities of thermoregulation and circulation are cited as the cause of impaired spermatogenesis in spinal injuries leading to paraplegia or quadriplegia. 51
Infective and inflammatory disorders
Mumps is the best known viral cause of orchitis, but not appearing in prepubertal boys. Permanent sequelae follow bilateral involvement, principally engendering fertility problems. 52 Bacterial epididymitis and orchitis may result from ascending urethral, bladder or prostatic infections. Patients with AIDS frequently develop hypogonadism and infertility. Granulomatous conditions may affect the testes and lepromatous leprosy represents a common cause of testicular damage in many tropical countries. 53 Autoimmune involvement is seen in patients with forms of polyglandular syndrome and is especially associated with primary hypoadrenalism when there are sex steroid cell antibodies directed at common epitopes in both glands. 54
Secondary hypogonadism
Pituitary disease
Hypogonadism tends to occur early in the development of functionless, non-secretory tumours, usually shortly after growth hormone deficiency is biochemically identifiable. LH is more markedly affected than FSH. 55 Hyperprolactinaemia is probably commoner in men than is generally recognized, in whom, by the time of diagnosis, tumours are often large and cause mass effects such as headaches, compression of the optic chiasm and even cerebrospinal fluid leak (rhinorrhoea). 56 High circulating concentrations of prolactin indicate prolactin-secreting tumours, usually large, but concentrations below 2000 mU/L may derive from normal pituitary tissue, which, due to distortion of the pituitary stalk by a large functionless pituitary tumour, is deprived of the usual inhibition of prolactin secretion exerted by dopamine secreted from the hypothalamus into the portal veins running down to the anterior pituitary gland. Hyperprolactinaemia has central effects suppressing libido. 57 It also inhibits gonadotrophin secretion by making the central hypothalamic pulse generator driving gonadotrophin production slow down the frequency of pulses.
Further damage to gonadotrophin secretory capacity may result from neurosurgical adenoma removal and over a longer timecourse after pituitary radiotherapy.
Hypothalamic disease
The commonest manifestation is constitutional delay in growth and development, a physically benign condition in which the onset/tempo of puberty is delayed, but otherwise normal. It is often familial, affecting father, uncles or other close male family members. Because of the psychological stress it may cause, there is a readiness to treat with low-dose T for a matter of months to advance virilization and skeletal growth and probably also to encourage the initiation of endogenous pubertal development. As long as T doses used in the treatment are not excessive, no damage or delay to natural development ensues. 58 It is always important and often difficult to distinguish this very common situation from psycho-social or physical abuse, or more profound organic causes of hypothalamic hypogonadism, including Kallmann's syndrome where there are other physical features, notably anosmia/hyposmia and a variable number of other features including cleft lip and palate, renal tract anomalies and mirror movements (bimanual synkinesia). 59
A number of genetic mutations have been identified, but they only account for a minority of cases. 60 In part, the developmental failure often entails failure of migration of GnRH cell bodies from the olfactory placode in the embryonic forebrain to the medial-basal hypothalamus, from there to connect with the hypothalamo-pituitary portal blood vessels. 61
Another group of hypothalamic disorders is neoplastic, of which one of the commonest examples is craniopharyngioma, a benign tumour (with extremely rare exceptions) but which is associated with higher morbidity and mortality than pituitary tumours. 62 As with many structural hypothalamic disorders, diabetes insipidus often occurs as well as anterior pituitary failure. Craniopharyngioma is a frequent cause of hypopituitarism in childhood, but can present at any time from infancy to old age. A variety of hypothalamic tumours occur, often malignant, and include metastases from extra-cranial primaries. 63 Of particular note are germinomas, because of their propensity to spread suddenly and rapidly along the optic tracts leading to irreversible, profound visual loss, 64 and secondly in rare circumstances, instead of leading to a panhypopituitary state, cause a paradoxical sexual precocity due to secretion of human chorionic gonadotrophin, alongside deficiency of other pituitary hormones, save for prolactin, which may be moderately raised due to dopamine deficiency. 65 Head injury and cranial irradiation are both capable of producing hypopituitarism, not only via direct pituitary damage but also at a hypothalamic and/or pituitary stalk level, again inducing joint anterior and posterior lobe hormone deficiency plus raised levels of prolactin. 66,67 Similar hormonal features may be caused by Langerhans cell histiocytosis, xanthoma disseminatum, tuberculous mengitis, Wegener's granuloma, lymphocytic hypophysitis (usually associated with pregnancy, but has been seen in men) and cranial sarcoidosis – reputedly causing disturbance of thirst, as much as diabetes insipidus – and pituitary abscess. 68
Disorders acting at other/several sites
Hepatic effects
Elevation of sex hormone binding globulin (SHBG), especially if associated with diminished reserve of T production, may reduce free T concentration without markedly changing total T, as most often seen in ageing men, but also liver disease, especially alcoholic cirrhosis. 69 In thyrotoxic men the total T may be elevated due to SHBG rise, yet free T low. 70 Exogenous oral oestrogen is occasionally used in men with prostate cancer; endogenous oestrogens may derive from rare germ cell tumours, or arise from disturbed metabolism in men with cirrhosis.
Iron overload
Haemochromatosis may lead to iron deposition in many organs, including the testes and also the anterior pituitary, where there is a predilection for the gonadotroph cells, leading to a variably mixed picture of primary and secondary hypogonadism. 71,72 Similar effects are seen in patients with thalassaemia requiring lifelong multiple blood transfusions, though iron-chelating drugs can ameliorate but not reverse the situation. 73 In haemochromatosis, the treatment is venesection as indicated by haemoglobin and haematocrit values.
Chronic renal failure
Impaired spermatogenesis and testicular damage often leads to infertility in chronic renal failure, with abnormalities at all levels of the hypothalamo-pituitary-gonadal axis. 74 Sexual dysfunction is common, especially in patients on dialysis. 75 Hyper-prolactinaemia also complicates chronic renal failure very frequently. 76 It has been postulated that the elevated levels of parathyroid hormone may play a role in the development of impotence in uraemic men. 77
Diabetes mellitus and the metabolic syndrome
The high prevalence of sexual dysfunction is well recognized, with many contributory factors, including micro- and macrovascular disease, neuropathy as well as endocrine disturbances. In type 1 diabetic males, erectile dysfunction is exceedingly common and associated with adverse cardiovascular risk factors. 78 In type 2 diabetes and males with the metabolic syndrome, there are also high numbers with erectile dysfunction and T deficiency, again closely associated with increased cardiovascular risk. 79,80 Whereas moderate obesity (body mass index [BMI] 30–35) is associated with lower serum total T, resulting from decreased serum SHBG, morbidly obese men (BMI > 40) also tend to have decreased free T as a consequence of diminished gonadotrophin secretion. 81
Hormone biochemistry and diagnosis
The key measurement in assessment of male gonadal function is serum T. Relevant biological considerations include the circadian rhythm, mentioned above, which may exceed a 30% difference between the peak morning values and those obtained later in the day. 82 Hence any borderline measurements obtained in the afternoon or evening need repeating in the morning. Severe acute illness depresses serum T, so definitive diagnosis requires reassessment on recovery. 83 The wide range of additional biological and technical issues involving T measurements is addressed below. At the stage of initial diagnosis, however, it is almost always necessary to assess gonadotrophin (LH and FSH) secretion to determine if there is an obvious primary or secondary cause of any low production of serum T. This is not normally problematical, despite the pulsatile secretion of LH in particular, since in established gonadal failure the elevation of the gonadotrophins is in general clear-cut, even though with secondary failure the levels of gonadotrophins often overlap with the lower part of the reference range.
The hCG test continues to be valuable, especially to assess the presence of responding Leydig cell tissue in cryptorchid boys. Studies claim to show that recombinant hCG yields equivalent responses to the classical extracted hormone. 84 Published studies, have used varied protocols and too small numbers to establish definitive normal responses. Neither the GnRH test nor the clomifene test is much used nowadays in the differential diagnosis of hypogonadism as they rarely add much information with modern quality gonadotrophin assays.
In patients with secondary hypogonadism (pituitary or hypothalamic disorders) prolactin should be measured, as high levels can usually be normalized by dopamine agonist drugs. 68 Current concerns that drugs of this class containing ergot alkaloid moieties may cause serious fibrotic reactions especially with cardiac valves are essentially confined to the prolonged high doses used in treatment of Parkinson's disease, not the much lower ones generally required to manage hyperprolactinaemia. 85,86
Measurement and interpretation of circulating T
The first line and, in most clinical laboratories, only androgen measurement is the serum total T concentration. It has a chequered history as technologies were developed to cater for (and encourage) increasing demand from clinicians for T measurements in patients. A consequence has been the widespread adoption of fully automated immunoassays with no prior steroid extraction, much more user-friendly but open to various types of interference. 87,88 The rather surprising lack of a universally accepted standard preparation of T also did not help. Matters in the USA were subjected to a critical analysis of the commercially available assays and the validity of many of the reference ranges used in a position statement from the Endocrine Society. 89 In the UK the situation appears to be rather better and improving with the increasing number of laboratories using tandem mass spectrometry. 90,91 There remain concerns how reference ranges are established.
The biologically relevant component of circulating T is generally reckoned to be non-SHBG bound; whether this is solely free T or ‘bioactive T’ (where the fraction loosely bound to albumin is added to the free T) remains debatable.
92
This may be of little practical consequence, since many studies have indicated that the two measurements are very highly correlated.
93
The gold standard method for estimation of the free T fraction remains isotope dilution equilibrium dialysis,
94
but alternatives involving ultrafiltration and direct measurement of the free fraction using mass spectrometry are being developed.
95
Analogue methods purporting to measure free T directly have proved inadequate.
96,97
Meanwhile, there have been several mathematical formulae published to calculate free T and bioactive T from measurements of total serum T, SHBG (and albumin).
92,98–101
An on-line calculator is available at
The assessment of the biological action of T is further complicated by studies of some common polymorphisms of the SHBG gene, which appear not only to affect the SHBG levels and hence T transport, but also seem to suggest SHBG feedback action on LH, leading to slightly increased free T concentration accompanying the raised SHBG. 103 Finally, T action might be modulated by polymorphisms in the androgen receptor and individual differences in androgen sensitivity. 104
Prevalence of hypogonadism and changes in circulating T in old age
This important area continues to raise questions defying definitive answers. Many of the distinct pathological entities leading to male hypogonadism described earlier are associated with diagnostic clinical features and biochemistry, usually including markedly low circulating T concentration (unless there are issues of impaired androgen response). Many are uncommon or extremely rare, but nevertheless illustrate aspects of androgen-related physiology and, where it is appropriate, the use of T substitution therapy raises no problems.
The clinical conundrums largely arise from the now very clearly delineated age-related decline in circulating T concentration, stretching from early adult life steadily into very old age, 105 without any suggestion of the kind of abrupt drop in oestradiol levels seen at the menopause in women. 20 The male situation is compounded by the reverse rise in concentration of SHBG with age, accentuating effects on free T changes. The initial data from cross-sectional studies are strengthened by subsequent major longitudinal ones. 106–108 The crucial dilemma is whether this decline in T secretion over the years warrants intervention and if so, when. Parallels have been drawn between the classical features of hypogonadism and those associated with ageing in the domains of sexuality, strength, stamina, mood and cognition, and physical changes affecting body composition, particularly lean body mass, fat mass and distribution, bone density and haematocrit. 109 The changes, however, are generally more subtle and certainly lack diagnostic specificity. The situation closely resembles the age-related changes in the growth hormone/IGF-1 axis and indeed there are clear areas of interaction between the two systems. 110 In both there is a great spread found across the normal population at all ages. There are confounding effects on the age-related fall in total T, such as the exaggeration seen with increasing adiposity. 111 This is partly due to reduction in SHBG (usually attributed to insulin resistance in obesity), but this is not the complete story, as free T levels also decline faster in the obese as they age. 112 The progressive increase in prevalence of obesity may account for some of the reported secular decline in T levels, although other potential factors such as endocrine disruptors have been suggested. 113 The pathophysiology of age-related gonadal changes include both reduced testicular reserve and diminished hypothalamo-pituitary drive. 105
As yet there is no agreement as to the appropriate level at which to diagnose hypo-gonadism in the elderly. It has been argued that there are no major physiological reasons to suggest that the ageing male is overall more or less sensitive to any given level of circulating T. 112 The variations in analytical techniques and reference ranges currently in use make it impossible to set a universally acceptable figure. In general, figures range for total T from 8 to 11.3 nmol/L.
Surveys range from hospital clinics and general practices to structured samples of the general population – the former tending to yield lower values because chronic diseases, which tend to lower T levels, are more frequently represented in medical settings. 114 There is agreement that clinical and biochemical features should converge before starting treatment with T. 88,112,115
A major deterrent to over ready use of T replacement therapy is economic, because the prevalence of hypogonadism using various parameters in different studies ranges from 20% to 70% in men in their seventh and eighth decades, while the percentage of the population in that age group has doubled since 1950 and is projected to increase another 10% by 2050, reaching nearly 30% of the population by that time. 88,116
A final important area of concern is the potential risks associated with T therapy in the elderly. The immediate first thoughts are usually prostatic disease, especially cancer, but there is no compelling evidence that T either impairs urine flow in benign disease (though prostate volume does increase) or that it increases the incidence of clinical cancer, though its use is contraindicated in the presence of diagnosed disease where it can aggravate locally advanced and metastatic prostate cancer. 117 It is of interest that prostatic cancer itself can induce hypogonadism, reversible on treatment, hence digital rectal examination and prostate-specific antigen measurement remain important before starting T therapy. 118
There is evidence that hypogonadism is an adverse cardiovascular risk factor. Many surrogate factors for cardiovascular disease improve in short-term studies using exogenous T, 119,120 but the Women's Health Initiative and other studies of hormone replacement therapy in women remind us of the dangers of heeding such studies without randomized clinical trials. Monitoring men on T treatment also involves checking for erythrocytosis and sleep apnoea. 117
This leads to the current guidelines on investigation, treatment and monitoring of late-onset hypogonadism in males, which has been as a consensus statement by a multinational panel incorporating recommendations from the International Society of Andrology, the International Society for the Study of the Aging Male, the European Academy of Urology and American Society of Andrology. This has been widely published to obtain maximum access to the multiple relevant specialties, and with which this article aims to be in full accord. 121–126
Available modalities for administration of exogenous T
In line with the international consensus panel's recommendations, only formulations of native T will be considered as appropriate therapeutic agents these days. Formerly, especially in the USA, 17α alkylated derivatives of T were used as these were effective taken orally, but are no longer used (except for large quantities by anabolic steroid abusers in gymnasia and still some athletes) as they may cause benign and malignant hepatic tumours. 127 A transdermal preparation of DHT has been used, particularly in France, for treatment of microphallus in childhood and also gynaecomastia. 128 However, as DHT cannot be aromatized to oestradiol, the use of DHT is a less physiological approach, which might be particularly relevant for skeletal health.
The main preparations used for many years have been various T esters dissolved in oily vehicles and injected intramuscularly. The resulting serum T concentrations achieved over time following injection depend on the rate of hydrolysis of the esters, those with longer aliphatic chains increasing the duration of release (Figure 4). In the UK the brands Sustanon 100 and Sustanon 250 (Organon) are the most popular and are certainly the cheapest. They consist of blends of different length esters, purportedly providing a smooth delivery of T into the circulation for about two or three weeks, respectively. They do not get smart levels, instead quickly produce supraphysiological peaks of T within days of injection, which is followed by a rapid exponential fall to preinjection levels, usually leaving a period of untreated hypogonadism – often symptomatic (as are excessive early levels, in the opposite direction), until this erratic cycle is repeated with the next injection. 1 Pain at the injection site is not rare, despite careful and accurate administration: either immediately, or starting some hours later, when there may be systemic features like slight pyrexia, rise in neutrophil count and C-reactive protein. A better tolerated and with better pharmaco-dynamic profile is T enantate, but it costs much more (despite having only one active constituent). This and the pharmacologically very similar T cypionate are the favoured intramuscular preparations in the USA. In the past, mesterolone, an orally active derivative of DHT, and fluoxymesterone, a 17α-alkylated steroid, were used but have now been dispensed with. Another longstanding preparation in use less commonly are T pellets. These are implanted subcutaneously in the abdomen or gluteal regions. These provide excellent, prolonged and stable physiological T levels. 129 Though cheap, their administration every 4–6 months, involves a sterile, minor surgical procedure using a trocar and cannula and local anaesthesia. It is thus costly in time, inconvenient and requires some skill and care to minimize the occasional extrusion of pellets and even more rarely local infections. For many patients, however, this proved a highly acceptable, reliable mode of T administration when performed by an experienced person, with remarkably few complications.
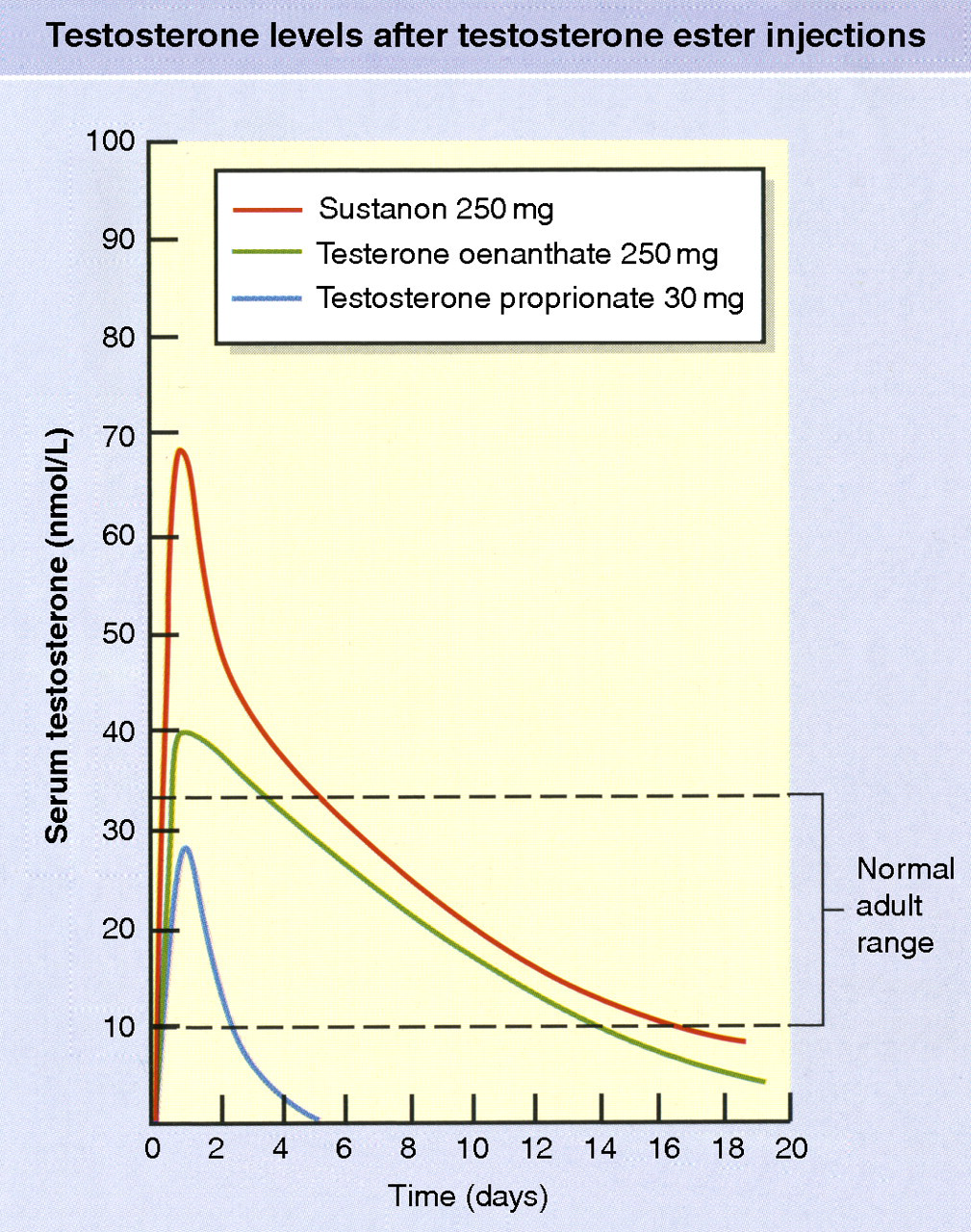
Pharmacodynamic profiles of serum testosterone concentration after single injections of commonly used short-acting testosterone esters. Please see online for colour version
Preparations that have subsequently been developed have proved of rather variable acceptability. The original transdermal patches were applied to the shaved scrotum and though achieving some popularity in the USA failed to do so in Europe. 130 These were followed by patches that could be applied to non-scrotal skin, through which, with the aid of amplifying agents, adequate amounts of T can be absorbed. These are still used by some patients, but cause unacceptable itch and irritation for very many others; also, they are large, often two are required that need replacing daily and are generally not very user-friendly. 131 Although native T on ingestion is largely metabolized on the first pass through the liver, when taken as T undecanoate in oil-containing capsules with a meal, much enters lymphatics in chylomicra by virtue of the long lipophilic side chain and thence into the systemic circulation via the thoracic duct. To secure adequate durable levels of T requires two or more 40 mg capsules as often as five times daily – which rarely proves to be practical. Some must reach the liver as the SHBG concentration falls with its use. It has a niche use for inducing puberty in boys, where only small, short lasting exposures to T are needed. 132
Recent years have seen products transforming the ease and success of administering T. First have been T containing gels rubbed into the skin, which provide rapid and reliable absorption of a day's requirements with one dose. 133 Three versions are available in the UK. They cause strikingly few adverse side-effects, particularly little irritation. Much is made of the need to avoid transferring T to other by avoiding skin-to-skin contact for several hours after application but is not a troublesome precaution. Very good quasi-physiological circadian variations in serum T levels can be obtained after regular morning use. The ability to stop, knowing that most of the absorbed T will disappear in 24 h, encourages its use in the early trials of a patient's response to T therapy, and many men elect to continue using it long term. Now, the most popular ongoing T medication is intramuscular injections of T undecanoate in oil. The usual protocol recommends the second injection should follow the first after six weeks, then leave 12 weeks and measure serum T immediately prior to the third injection. 134 If this T level is mid-normal adult range, subsequent injection should be given at 12-week intervals; if not, alter the interval appropriately to between 10 and 14 weeks to maintain steady average adult male serum T concentrations. A new T product on the market is a buccal tablet made to apply to the gum in the concave depression above the second upper incisor. The dose has to be repeated every 12 h. 135 Not many patients so far choose it in preference to the transdermal gels or long-acting T undecanoate injections.
Conclusion
Male hypogonadism may manifest at any phase in life from the fetus to old age and may result from a wide variety of causes. Diagnosis largely depends on measurement of T, problems with which have been prominent, but are currently well on the way to resolution. T therapy has also undergone major improvements in mode and acceptability of administration.
This paper is dedicated to the memory of Julie Swinburne, endocrine nurse specialist at Leeds General Infirmary, who developed the nurse-led testosterone pellet implant clinic, helped evaluate several new modalities of testosterone therapy and generally supported and educated the large number of patients attending the endocrine clinics. She died aged 38, after a long, painful and courageous fight against aggressive breast cancer.
DECLARATIONS
The figures used in this review are reproduced with permission from Mosby's Color Atlas and Text of Diabetes and Endocrinology, 2nd Reprint, Paul Belchetz, Peter Hammond, Endocrinological Problems in Children and Adolescents, Page 221 Fig 16.2, Reproductive Endocrinology, Page 301 Fig 19.4, Page 305 Fig 19.9, Page 323 Fig 19.30, Copyright Elsevier, First published 2003, reprinted 2004.
