Abstract
Background
The aim of the present study was to evaluate standard reference material (SRM) 1955 commutability as a reference material for serum folate using automated methods. We also designed so as to reduce the intermethod variability present in different automated methods.
Methods
Using a microbiological assay related to the ‘information value’ of SRM 1955 as a comparison method, we investigated the possibility of standardization for the assay values of serum folate as measured by the automated methods (Access, Centaur and Elecsys). In the assay of 50 patient sera by these automated methods, we corrected observed values by the SRM 1955 and compared with comparison values.
Results
The observed values of SRM 1955 Levels I, II and III were within or outside (but near) a 95% prediction interval obtained from patient sera by the automated methods. The normalized residuals obtained from SRM 1955 were within ±3.0 (in SD units), which enabled us to conclude that the SRM 1955 had a physicochemical characterization similar to native serum. Twelve patients were assessed as hypofolataemia (<6.0 ng/mL) and 38 patients as normal (≥6.0 ng/mL). Before correction, folate levels in six of 12 patients were lower than 6.0 ng/mL, and those in seven of 38 patients were higher than 6.0 ng/mL with the automated methods. After correction, low levels were found in four of 12 patients, and normal levels were found in 33 of 38 patients.
Conclusions
The use of SRM 1955 would help to reduce the intermethod variability present in different automated methods for serum folate measurement.
Introduction
Although 5-methyltetrahydrofolic acid (5MeTHF) is the predominant folate vitamer both in plasma and cells, 5-formyltetrahydrofolic acid (5FoTHF), 5,10-methenyltetrahydrofolic acid, folic acid and various other forms of tetrahydrofolic acid (THF) are present in vivo. 5MeTHF acts in methylation reactions, and 5FoTHF acts in nucleotide biosynthesis. 1 Folate deficiency symptoms including megaloblastic anaemia are arteriosclerosis, cancer, depression in the elderly, and impaired growth and neural tube defects in fetuses. In the assessment of an individual's folate status, the amount of folate in the serum, which reflects the recent intake, and red cell folate as a better index of body stores are determined. 2 In practice, folate is being measured in the serum by the use of an automated assay in clinical laboratories. The traditional method for folate measurement (i.e. microbiological assay with Lactobacillus casei) has been almost completely replaced by the automated assay, but there is a considerable lack of agreement regarding the results from the use of different automated methods. 3
In order to reduce the discrepancies among laboratories using different automated methods and achieve uniform results in folate measurement, the World Health Organization (WHO) Exert Committee on Biological Standardization selected the isotope-dilution liquid chromatography tandem mass spectrometry (LC/MS/MS) 4,5 as a candidate reference method. 6–8 Recently, based on WHO recommendations, the National Institute of Standards and Technology (NIST) has developed a standard reference material (SRM 1955 Homocysteine and Folate in Frozen Human Serum) 9 to use as control material needed for standardization of folate measurement. SRM 1955 has been assigned the certified values for total homocysteine and 5MeTHF utilizing the LC/MS/MS, and additionally ‘information values’ for 5FoTHF, folic acid and total folate (i.e. sum of 5MeTHF, 5FoTHF and folic acid).
The aim of the present study was therefore to evaluate SRM 1955 commutability as a reference material for total folate using the automated assays currently available in Japan. This study was also designed to reduce the intermethod variability present in different automated methods. Some automated methods for analysis could measure red cell folate, but we leave the utilization of SRM 1955 for future study.
Materials and methods
Participating laboratories and methods
Four laboratories participated in the folate assays. Three laboratories measured the serum folate concentrations by the automated method based on the competitive protein binding (CPB) assay, i.e. Access (Beckman Coulter, Brea, CA, USA), Advia Centaur (Siemens Healthcare Diagnostics, Deerfield, IL, USA) and Elecsys (Roche Diagnostics, Basel, Switzerland), according to the instructions of the automated methods and the manufacturer's reagents. Analytical precision and linearity ranges of the three instruments were within- and between-day coefficients of variation <10% and 0.5–20 ng/mL, respectively. 3 Although more automated CPB methods are being developed around the world including a radiometric CPB assay (Quantaphase II, Bio-Rad Laboratories, Hercules, CA, USA), the above three instruments are exclusively available in Japan. One laboratory measured the serum folate concentrations by microbiological assay using the assay organism Lactobacillus rhamnosus American Type Culture Collection (ATCC) 27773 (L. casei), which responds to a wide variety of folate vitamers such as 5MeTHF, 5FoTHF, THF and folic acid. 10–12
Study design
Folate concentrations in SRM 1955 and a total of 50 serum samples were measured by each of the four laboratories using their own folate assay procedure and calibrants. Here, 50 serum pools were residual aliquots of the serum of hospitalized patients considered to have hypofolataemia. Our study was in compliance with the rules for human experimentation of the Japanese Society of Laboratory Medicine.
SRM 1955 was composed of three vials, i.e. Level I, II and III (1.0 mL of frozen serum each).
9
Level II was prepared from a pool of human serum with no additives. Level I was prepared by diluting the serum pool 1:1 (volumetrically) with phosphate-buffered saline (PBS), pH 7.04. Level III was prepared by adding a known amount of 5MeTHF and homocysteine to Level II. Assigned concentrations of 5MeTHF (certified value), 5FoTHF and folic acid were determined by the LC/MS/MS analysis (the candidate reference method). NIST provided a ‘certified concentration value’ only for 5MeTHF, because a complete evaluation of uncertainty for 5FoTHF and folic acid has not been available in LC/MS/MS analysis. Total folate was the ‘information value’ that was calculated as the sum of the molar concentration of 5MeTHF, 5FoTHF and folic acid, and the assay values were expressed as ng/mL in terms of their folic acid equivalent. For conversion to mass units (ng/mL), WHO recommended that molar units have to be multiplied by a factor of 0.441.
6,7
The ‘information value’ was 2.65 ng/mL for Level I, 5.78 ng/mL for Level II and 18.08 ng/mL for Level III. Each SRM 1955 level was thawed at room temperature (
Data analysis
For the assessment of SRM 1955's commutability, the regression statistics reported by Nelson et al. 13 were applied to the data by automated methods. Here, we used the microbiological assay as the comparative assay, because the microbiological assay almost completely recovered the folate vitamers added to the serum samples, and their total folate values were generally in agreement with the LC/MS/MS. 14
In the linear regression analysis, each evaluated assay (observed values of Access, Advia Centaur and Elecsys, not corrected for SRM 1955) was assigned to the y-axis, and the comparative microbiological assay (corrected values related to SRM 1955) was utilized as the reference assay (x-axis). A regression equation was obtained from the 50-patient specimen data for each evaluated assay-comparative assay pair, and the 95% prediction intervals (95% PI)
15
for the patient specimen data about the resultant regression lines for each evaluated assay were calculated using a two-tailed test. The resultant slope and intercept values were then used to calculate the predicted y values (ŷ) for each sample x value:
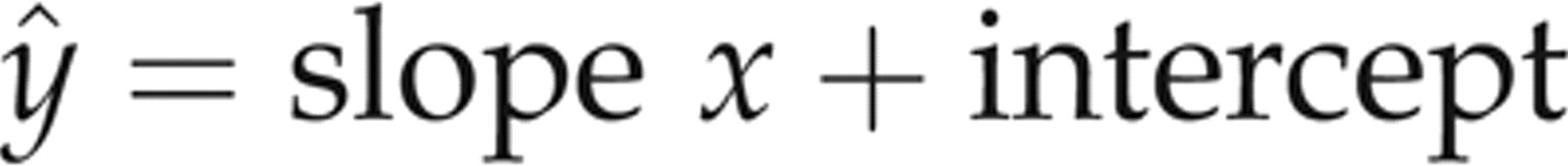
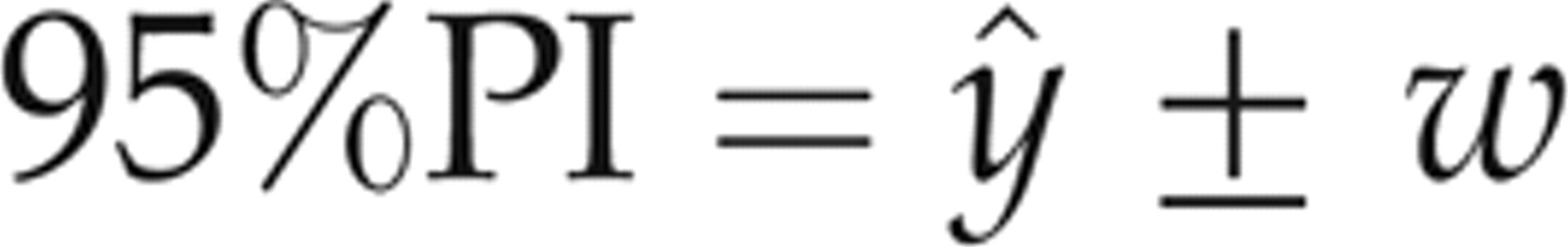
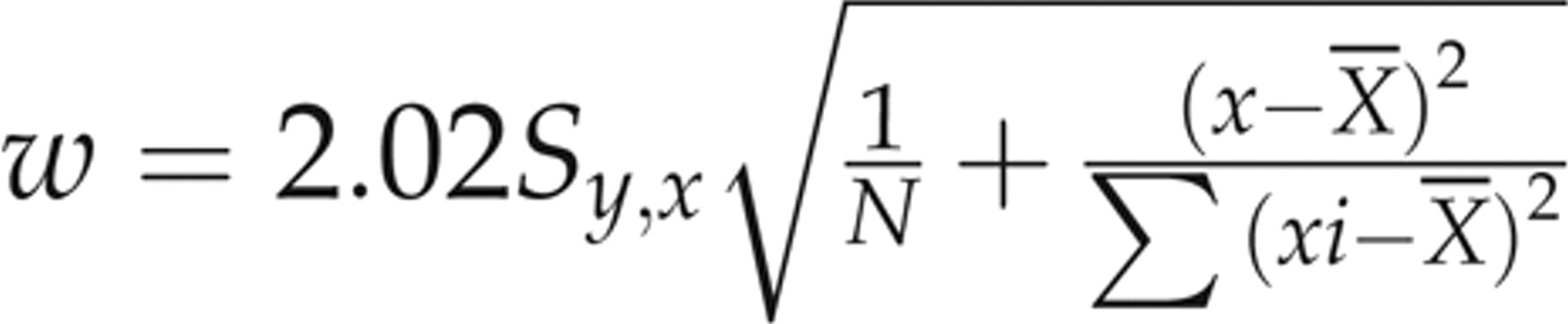
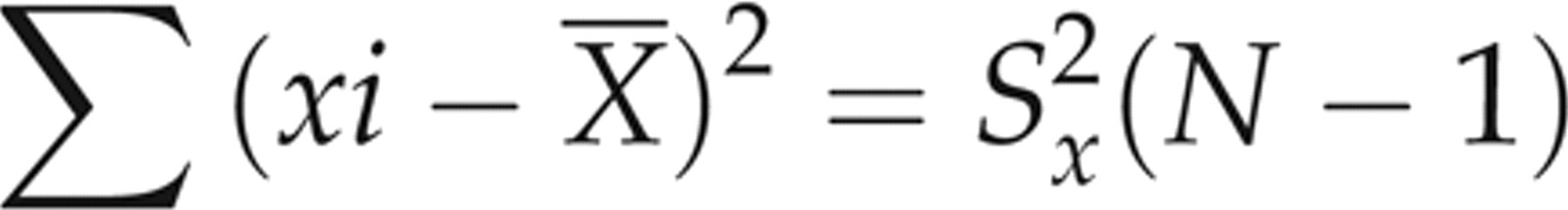
The residuals along the y-axis were calculated as the difference between the y value and the predicted value (ŷ). The normalized residuals were then calculated by dividing the residuals (y − ŷ) by the SD of the residuals from the patient specimen data (for a given evaluated assay). For each level of SRM 1955, the commutability was determined by assessing whether its normalized residual was within ±3.0 (in SD units). SRM 1955 samples that resulted in normalized residuals >±3.0 were deemed non-commutable with the given evaluated assay. 13
Results
Observed values of SRM 1955
In the measurement of the total folate concentrations of SRM Levels I, II and III by microbiological assay, the observed value was on average 16% higher than the ‘information value’ (Figure 1). While the total folate concentrations of SRM Levels I, II and III were measured by three automated methods, the observed values of SRM Level I were 44% lower by Access and 24% higher by Elecsys than the ‘information value’. The observed value of SRM Level II was 29% lower by Centaur and 13% higher by Elecsys, and that of SRM Level III was 43% and 21% lower by Access and Elecsys, respectively. In the regression analysis, the intercept of the y-axis was −0.2, −0.1 and 2.0 ng/mL by microbiological assay, Access and Elecsys, respectively. Because of the high cost of SRM 1955 and the small volume of these vials (1.0 mL), we could not determine the total folate concentrations by Access for Level II, and by Centaur both for Level I and Level III.
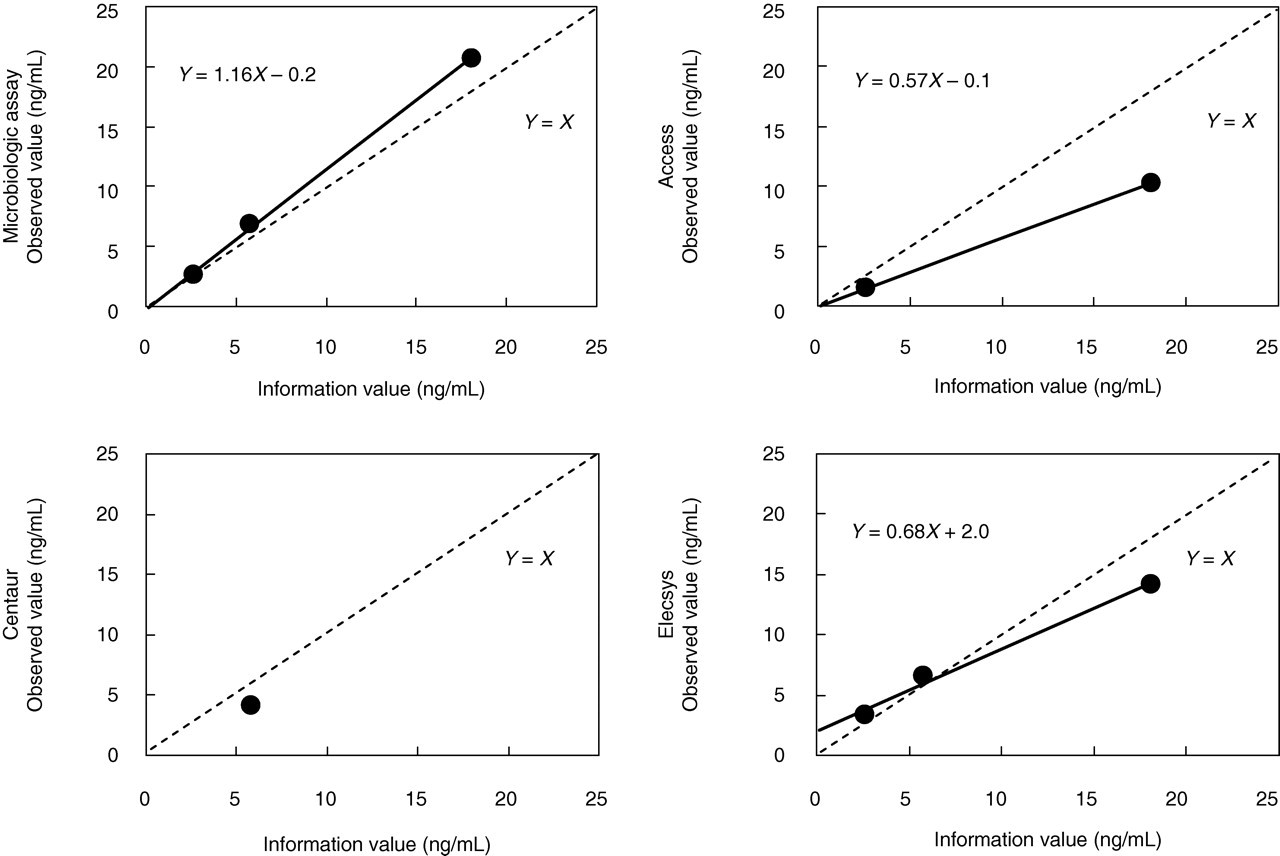
Observed values of SRM 1955 determined by microbiological assay and three automated methods. The ‘information values’ of the total folate concentrations were 2.65 ng/mL for Level I, 5.78 ng/mL for Level II and 18.08 ng/mL for Level III. SRM, standard reference material
Commutability
Patient specimen and SRM 1955 data generated each of the three evaluated assays (i.e. data by three automated methods not corrected by the ‘information values’ of SRM 1955) were plotted against the patient specimen and SRM 1955 data generated with the comparative assay. In this study, we selected the microbiological assay which was calibrated against the ‘information value’ of SRM 1955 as a comparative assay. Because the data from 50 patient specimens by the three automated methods were significantly correlated with those by the comparative microbiological assay, the regression line through the patient specimen results and the corresponding two-tailed 95% PI were calculated. Data points for SRM Level III as determined by Access and Elecsys were within the 95% PI generated from the 50 patient specimens (Figure 2). Data points for SRM Level II as determined by Elecsys were within the 95% PI, but that by Centaur was 1.0 ng/mL outside the lower limit of the 95% PI (1.0 ng/mL bias was higher than the measurement imprecision). Data points for SRM Level I determined by Access were within the 95% PI, but that by Centaur was 0.4 ng/mL outside the lower limit of the 95% PI (0.4 ng/mL bias was within the measurement imprecision).
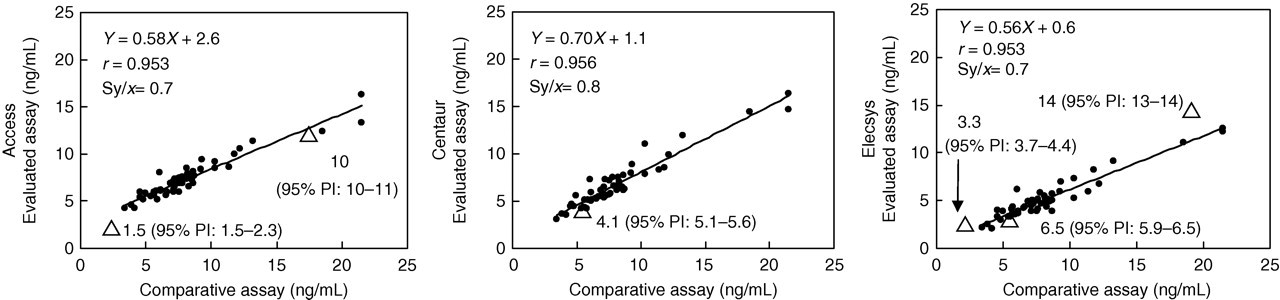
Linear regression plots of the evaluated assay data of automated methods versus the comparative assay data for the determination of the total folate concentration. (•) patient specimens; (▵) SRM 1955 (Levels I, II and III). PI, prediction interval
The plots of all of the normalized residuals including patient sera and SRM 1955 Levels I, II and III versus the comparative microbiological assay are shown in Figure 3. All of the evaluated assays generated normalized residuals (in SD units) within the ±3.0 interval for SRM 1955 Levels I, II and III. The normalized residuals of SRM 1955 Level I and Level III by Access were −0.64 and −0.52, respectively. The normalized residuals of Level II by Centaur were −1.52, and Level I, Level II and Level III by Elecsys were −1.06, 0.56 and 1.61, respectively.
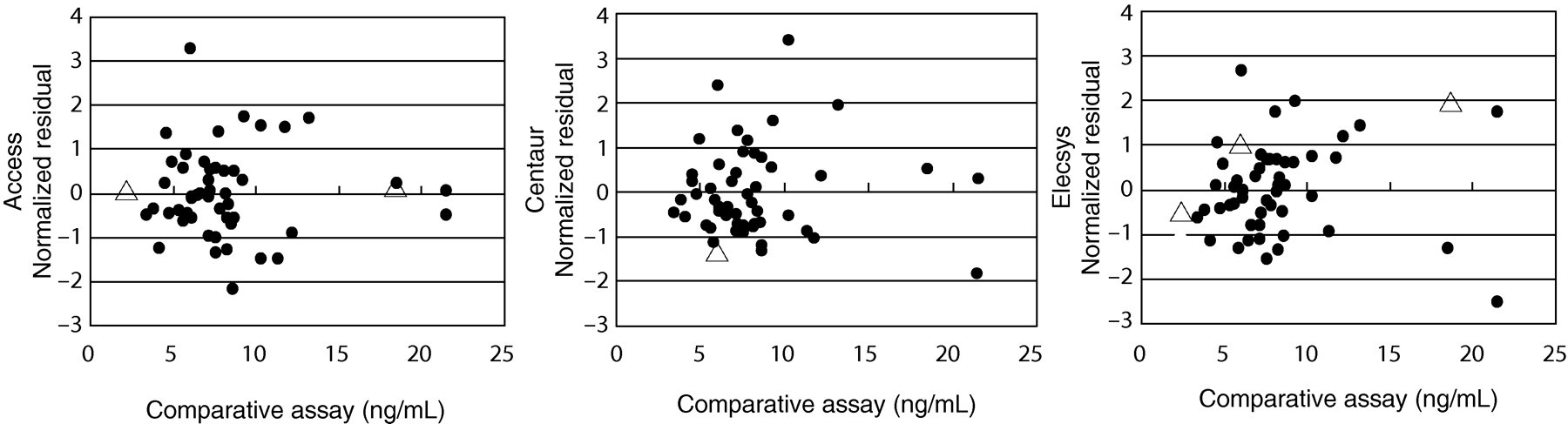
Plots of the normalized residuals versus the comparative microbiological assay. (•) patient specimens: (▵) SRM 1955 (Levels I, II and III). SRM, standard reference material
Standardization of assay values
Observed values of the total folate concentrations of the sera from 50 hospitalized patients measured by microbiological assay and the three automated methods were corrected by the ‘information values’ of SRM 1955. The observed values (Figure 4, left) and their corrected values (Figure 4, right) of the three automated methods were compared with a comparative assay. In addition, we nutritionally assessed the patients' data based on the criteria 2 as follows: <3.0 ng/mL was deficient, 3.0–5.9 ng/mL was low and ≥6.0 ng/mL was normal. The results of the nutritional assessments between the comparative assay and the automated methods disagreed in 33 of 50 patients by Access, 10 of 50 patients by Centaur and six of 50 patients by Elecsys before correction (Figure 4, left). After the observed values were corrected by SRM 1955, the nutritional assessments disagreed in five of 50 patients by Access, seven of 50 patients by Centaur and six of 50 patients by Elecsys (Figure 4, left).
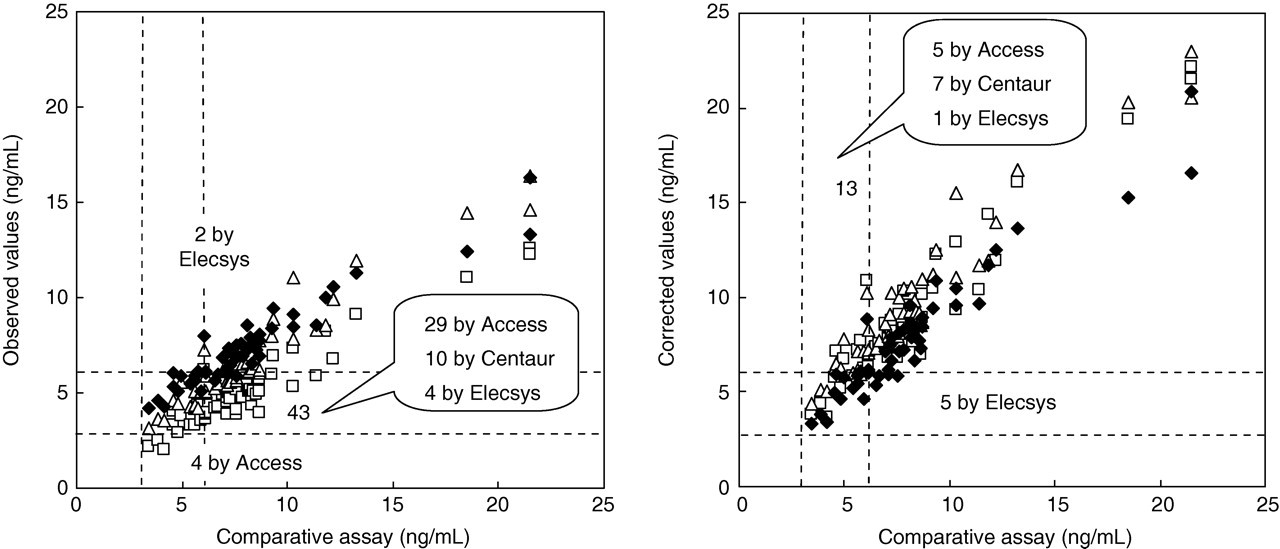
Assay values of the total serum folate concentrations in the three automated methods, all plotted both before (left, observed values) and after correction by SRM 1955 (right, corrected values). Numbers indicate cases that disagreed in the nutritional assessment between the comparative assay and automated methods: (□) Access, (▵) Centaur, (⧫) Elecsys. Corrected values (X: dependent variable) were obtained by putting the observed value of each assay into the ‘Y: explanatory variable’ of the regression equation as shown in Fig. 1. As mentioned in the ‘Materials and methods’, we utilized the corrected values of the microbiological assay as the comparative assay. SRM, standard reference material
Discussion
SRM 1955 was specifically designed as a primary control material for serum-based clinical assays of homocysteine and three folate vitamers; however, each concentration level of SRM 1955 has slightly different analytical specifications. The base serum pool, containing only endogenous folate forms, was developed into Level II of SRM 1955. Level I of SRM 1955 was prepared by diluting the base serum pool with PBS. Level III of SRM 1955 was prepared by spiking a known amount of 5MeTHF primary standard into the base serum pool. Thus, in comparison to Level II of SRM 1955, Level I of SRM 1955 has a reduced overall protein concentration and Level III of SRM 1955 contained both natural and synthetic forms of folate vitamers. In our previous study, a two-fold dilution with distilled water did not affect the assay values of the total folate concentration in the serum (unpublished data). We also reported that the automated methods gave a different reactivity between the serum folate and 5MeTHF solution prepared in the absence of serum protein. 3 However, in the present study, data points for SRM 1955 Levels I, II and III were within or outside (but near) the 95% PI. Although the observed value of Level II by Centaur was 1.0 ng/mL lower than 95% PI, the behaviour of SRM 1955 with the automated methods was quite similar to the behaviour observed in the patient specimens.
In this study, 95% PI was obtained against the comparative microbiological assay, since the National Health and Nutrition Examination Survey (NHANES) in the USA 14,16 decided to use LC/MS/MS (reference method) or a microbiological assay. The microbiological assay almost completely recovered the folate vitamers added to serum samples, except for folic acid (69%) and THF (36%). 14 These unrecovered biases were negligible, because, in usual serum, the serum concentration of folic acid and THF is <10% (<2 ng/mL) and <5% (<1 ng/mL) total folate concentration. 14,17 Furthermore, an elevated THF was observed in red cell folate exclusively in individuals with the wild-type genotype on the methylentetrahydrofolate reductase (MTHFR). 17,18 Fazili et al. 19 reported that the total folate concentration by either LC/MS/MS or microbiological assay was not affected by the MTHFR genotype. Bio-Rad Quantaphase II, the traditional reference method, was not used for the NHANES, because of the under recovery of 5MeTHF (61%) and 5FoTHF (38%). 14 As the LS/MS/MS is not always usable in most clinical laboratories, we used the values of a microbiological assay corrected with the ‘information values’ of SRM 1955 as the comparative values. The quality of Lactobacillus rhamnosus ATCC 27773 (L. casei), assay organisms for the folate measurement, was completely standardized by the ATCC.
To more critically investigate the commutability of SRM 1955 with the evaluated assays, plots of the normalized residuals were constructed. Figure 3 graphically demonstrates the commutable behaviour of SRM 1955 Levels I, II and III with all of the evaluated assays. Each of the evaluated assays gave normalized residuals (in SD units) within the ±3.0 boundary for SRM 1955. Thus, the data provided strong evidence suggesting that SRM 1955 Levels I, II and III may be commutable with patient samples for all of the evaluated assays.
Hence, owing to the findings of this preliminary study, SRM 1955 could potentially serve as a useful control or calibrator material for these assays. The total folate concentrations of 50 sera from 50 hospitalized patients measured by a microbiological assay and the three automated methods were compared before and after correction with SRM 1955. Overall, discordant data in the nutritional assessment 2 were reduced after correction by SRM 1955 (Figure 4). In the 50 patients, 12 patients were assessed as hypofolataemia (<6.0 ng/mL) and 38 patients as normal (≥6.0 ng/mL) as determined by a comparative microbiological assay. Before correction, serum folate levels in six of 12 patients were lower than 6.0 ng/mL, and those in seven of 38 patients were higher than 6.0 ng/mL as determined by the three automated methods. After correction, patients with folate levels lower than 6.0 ng/mL were found in four of 12 patients, and higher than ≥6.0 ng/mL were found in 33 of 38 patients. However, 13 patients remained discordant in the nutritional assessment among the microbiological assay and automated methods, which was probably due to limitations in the commutability study archived here. For example, the 95% PI was obtained from 50 data of patients, but no patients provided total folate concentrations less than 3.0 ng/mL. Furthermore, we failed to measure SRM 1955 Levels I and III by Centaur.
In addition, we considered that the discordance observed here would be likely to arise from the different calibration processes (Figure 5) employed in each automated method. Before automated methods (i.e. Access, Centaur and Elecsys) were developed, the serum folate concentration was determined by Bio-Rad Quantaphase II. Centaur and Elecsys adjusted their values to those of the Quantaphase II. Roche Diagnostics measured serum specimens simultaneously by Elecsys (calibrated against folic acid) and Quantaphase II (calibrated against 5MeTHF), and then the observed regression equation was stored in the reagent bar code. Siemens Healthcare Diagnostics measured their calibrators of 5MeTHF by Quantaphase II to assign their calibrator values. Access was calibrated against known concentrations of folic acid solutions. 3 In Quantaphase II, 5MeTHF was used as a calibrator. Nowadays, manufacturer's procedure calibrators in these automated methods are not calibrated against SRM 1955. Hence, the utility of SRM 1955 is limited to standardize the observed values of total folate concentrations arising from different methods. Instead of this traditional traceability system (Figure 5), we propose a new traceability chain (Figure 6), wherein the manufacturer's standard measurement procedures, in which LC/MS/MS is used as a reference measurement procedure, and their manufacturer's procedure calibrator should be directly traceable to SRM 1955. Furthermore, a microbiological assay also required to calibrate against SRM 1955 as was done in the present study. The use of SRM 1955 in the standardization of total folate measurement would help to reduce the intermethod variability present in different assay methods, including automated methods and microbiological assay.
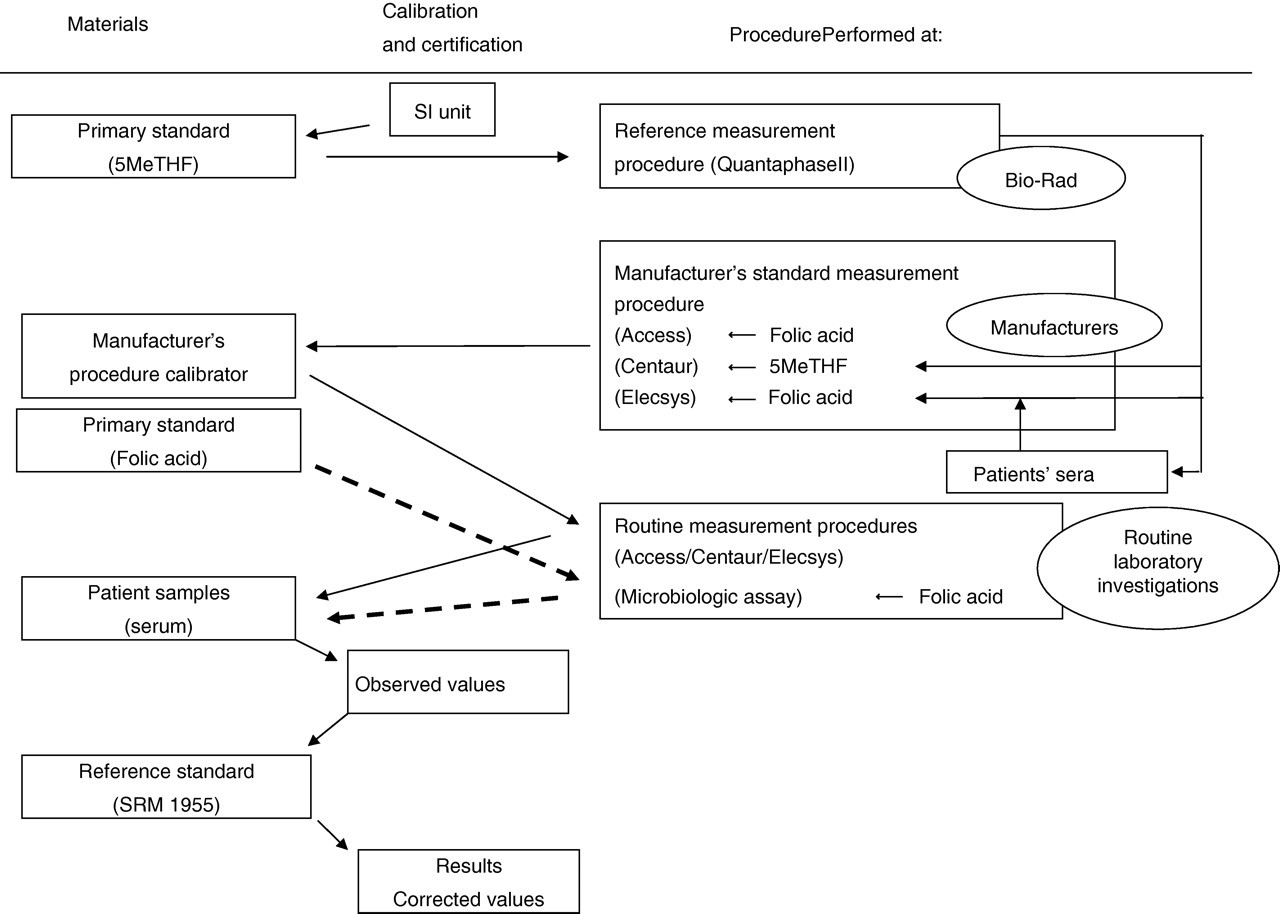
Traditional traceability system for total folate concentration, in which Bio-Rad Quantaphase II is used as a reference measurement procedure. SRM, standard reference material; 5MeTHF, 5-methyltetrahydrofolic acid
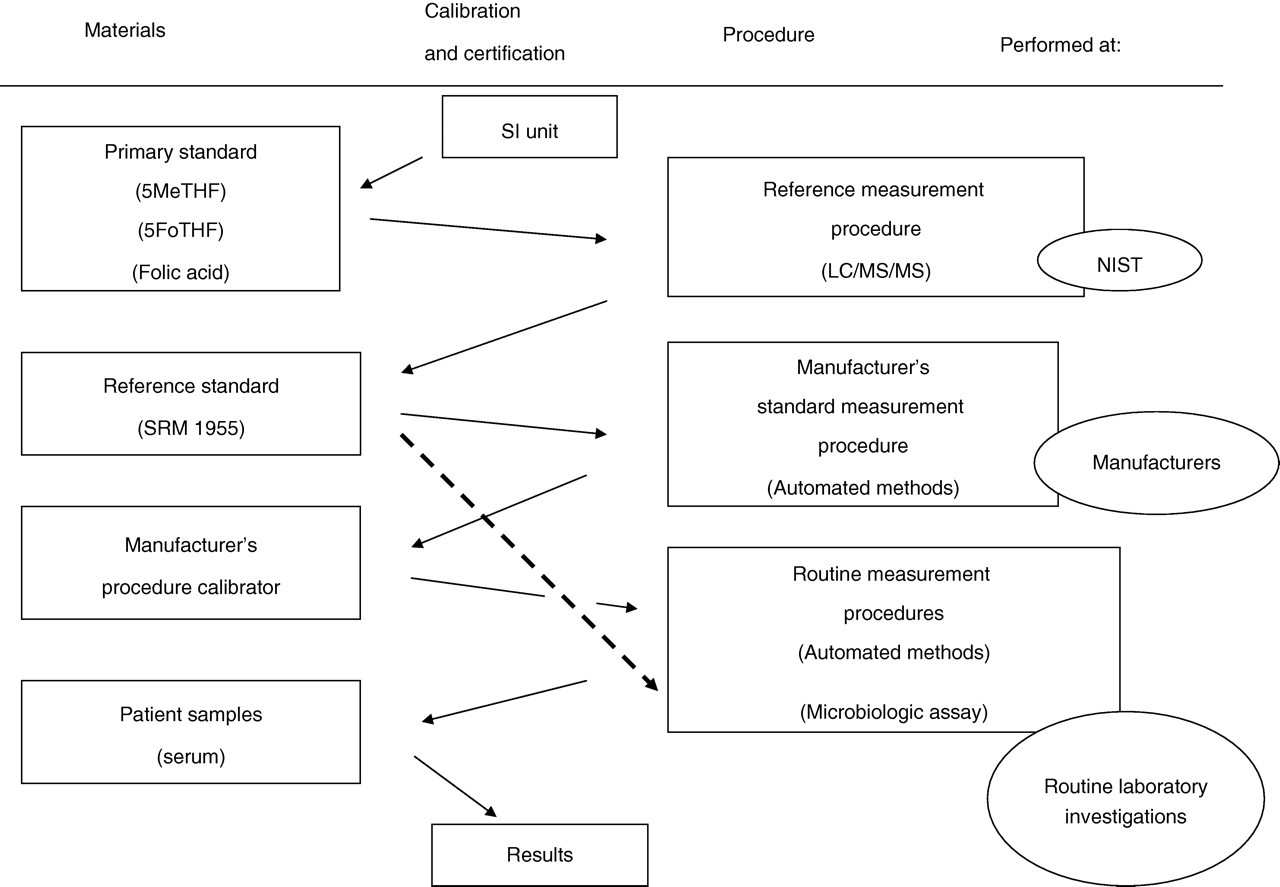
Proposed traceability chain for total folate concentration, in which LC/MS/MS is used as a reference measurement procedure. SRM, standard reference material; 5MeTHF, 5-methyltetrahydrofolic acid; 5FoTHF, 5-formyltetrahydrofolic acid; LC/MS/MS, liquid chromatography tandem mass spectrometry
DECLARATIONS
TW: folate measurement by microbiological assay; NH: planned the research study; MT: planned the research study; KK: folate measurement by Centaur; KO: folate measurement by Centaur; SS: folate measurement by Centaur; TS: folate measurement by Access; MI: folate measurement by Access; YA: search references, and read submitted English manuscript; MI: search references, and read submitted English manuscript; SI: search references, and read submitted English manuscript; KO: search references, and read submitted English manuscript; TE: read submitted manuscript (in Japanese); KS: read submitted manuscript (in Japanese); KS: read submitted manuscript (in Japanese); YN: read submitted manuscript (in Japanese); TN: search references, and read submitted English manuscript; KH: search references, and read submitted English manuscript; KF: read submitted manuscript (in Japanese); MM: search references, and read submitted English manuscript; MM: search references, and read submitted English manuscript; YO: search references, and read submitted English manuscript.
