Abstract
The diagnosis and prognosis of acute kidney injury (AKI) by current clinical means is inadequate. Biomarkers of kidney injury that are easily measured and unaffected by physiological variables could revolutionize the management of AKI. Our objective was to systematically review the diagnostic and prognostic utility of urine and serum biomarkers of AKI in humans. We searched MEDLINE, PubMed and EMBASE databases (January 2000–August 2009) for biomarker studies that could be classified into the following categories: (a) confirmation of the diagnosis of established AKI, (b) early prediction of AKI, and (c) prognostication of AKI. We identified 54 manuscripts published since 2000 that met our inclusion and exclusion criteria. Urinary interleukin-18 (IL-18), kidney injury molecule-1 (KIM-1), neutrophil gelatinase-associated lipocalin (NGAL) and N-acetyl-β-
Introduction
The clinical syndrome of acute kidney injury
The incidence of acute kidney injury (AKI, previously referred to as acute renal failure) has reached epidemic proportions worldwide, and is now estimated at 5–7% of all hospitalized patients. 1 In the critical care setting, AKI affects 5–25% of patients 2,3 and accounts for an overall mortality rate of 50–80%. 3–6 Once established, the treatment of AKI is largely supportive, unsatisfactory and associated with a poor prognosis. In a recent multinational study of AKI in nearly 30,000 critically ill subjects, the overall prevalence of AKI requiring supportive renal replacement therapy was 6% with a mortality rate of 60%. 7 The increase in morbidity and mortality associated with AKI has now been established in a wide variety of clinical situations, 8–11 including administration of radiocontrast dye, 12 cardiopulmonary bypass, 13–18 mechanical ventilation 19 and sepsis. 20,21 The dramatic negative influence of AKI on overall patient outcomes is also well documented. 22–27 AKI has emerged as a major risk factor for the development of non-renal complications, and as a powerful independent predictor of mortality. 12,28 Furthermore, the treatment of AKI represents an enormous financial burden to society, with annual expenses now estimated at $10 billion in the USA alone. 29,30
Tremendous progress has been made in our understanding of the molecular and cellular mechanisms of AKI, which has yielded novel therapeutic targets that work well in experimental models. 31–34 The translation of these findings to humans has been disappointing, and several of these agents have failed in clinical trials. 35–38 This disparity has been attributed, at least in part, to the paucity of early biomarkers for the prediction of AKI in the clinical setting. In the field of cardiology, the measurement of serum biomarkers such as cardiac troponins, that reflect early myocyte damage rather than diminished cardiac function, has facilitated the development and implementation of novel strategies for the management of coronary insufficiency, which have significantly reduced the morbidity and mortality from acute myocardial infarction. In stark contrast, AKI prevention and treatment studies using variables such as serum creatinine, urine output and serum and urine chemistries, that reflect the delayed diminution in kidney function and not the early structural tubule damage, have predictably not yielded interventions that decrease dialysis requirement or mortality. 39,40 Of those interventions that have been successful in small phase II studies of AKI, 41,42 none were efficacious at reducing dialysis requirement or mortality in larger phase III trials. 36,37,43 Regrettably, these studies were confounded by the use of serum creatinine concentration as an inclusion criterion, resulting in both delayed recognition of AKI and initiation of therapy. Possibly, these and other promising interventions would have been successful if they could be initiated at the onset of structural AKI rather than waiting several days for its functional sequelae such as the increase in serum creatinine.
The flawed ‘gold standard’ for the diagnosis of acute kidney injury
The diagnosis of AKI currently depends on detection of reduced kidney function by the increase in serum creatinine concentration. This is a notoriously delayed and unreliable measure in the acute setting, for a number of reasons. First, there are numerous non-renal factors influencing the serum creatinine concentration such as body weight, race, age, gender, hydration status, drugs, muscle mass, muscle metabolism and diet. Second, in AKI, serum creatinine is an even poorer reflection of kidney function, because the patients are not in steady state, and the increase in serum creatinine concentration lags far behind structural renal injury. 44,45 Third, significant renal disease can exist with no change in serum creatinine because of functional renal reserve, enhanced tubular secretion of creatinine and other factors. 44,46 Indeed, more than 50% of renal function may be lost before serum creatinine rises above the upper reference limit.
Other conventional biomarkers such as urinary casts (a late indicator of tubule damage) and fractional excretion of sodium (a highly variable measure that primarily differentiates volume-responsive prerenal injury from intrinsic AKI) have also proven to be insensitive and non-specific for the early recognition of AKI. 47–50 Thus, a troponin-like biomarker that is easily measured, unaffected by physiological variables, and capable of both early detection and risk stratification could revolutionize the management of AKI.
Tubular proteinuria for the diagnosis of AKI
Proteinuria can result from three basic mechanisms: glomerular, overflow and tubular. Glomerular proteinuria results from increased filtration of macromolecules (predominantly albumin) across the glomerulus. The implication of this type of proteinuria in the detection and management of chronic kidney disease has recently been published in this journal. 51 However, in the clinical setting, albuminuria can occur even in the absence of renal injury. Postulated mechanisms include acute changes in glomerular capillary permeability 52 and inflammatory insults. 53 In addition, clinical situations that lead to AKI may result in albuminuria due to diminished reabsorption by the injured proximal tubules. The potential utility of albuminuria as a test for the early diagnosis of AKI has not been systematically evaluated. Overflow proteinuria refers to the increased excretion of proteins that are markedly overproduced in specific disease states such that the filtered load exceeds the normal tubular reabsorptive capacity. Examples include immunoglobulin light chains in multiple myeloma and myoglobin in rhabdomyolysis.
Tubular proteinuria has traditionally referred to low molecular weight proteins that are normally produced in the body, freely filtered across the glomerulus and normally reabsorbed in the proximal tubule, but appear in the urine due to proximal tubular damage. Examples include α1-microglobulin, β2-microglobulin and retinol binding protein.
54–56
Tubular proteinuria can also result when proximal tubular enzymes and proteins, such as α-glutathione S-transferase (α-GST) and N-acetyl-β-
This review represents a systematic analysis of tubular proteins as biomarkers of AKI in humans, based on data published from January 2000 to July 2009. Both traditional as well as novel urinary biomarkers are critically evaluated. Also included are the most promising serum biomarkers for AKI, since some of the urinary biomarkers are also detected in, and may indeed originate from, the blood. Isolated glomerular proteinuria and biomarkers of chronic kidney disease are specifically excluded.
Methods
The most recent systematic analysis of biomarkers in AKI included studies from 2000 to 2006. 68 Since then, numerous new studies have substantially added to the field of knowledge. This review reproduces the systematic methods of the previous analysis, and updates the search to include all publications up to August 2009.
Study eligibility
Studies were eligible for this review if they were prospective or retrospective cohort studies, case-control studies or randomized controlled trials that investigated the use of tubular proteinuria or other biomarkers for the diagnosis or risk stratification of AKI. Studies that detected these proteins in either urine or blood samples were eligible. Only studies that examined 20 or more human subjects were included.
Search strategy
We searched MEDLINE, PubMed and EMBASE databases (January 2000–August 2009) using the MeSH terms ‘acute renal failure’ or ‘acute kidney injury’ or ‘creatinine blood level’ and cross referenced with the terms ‘biomarker’ or ‘biological marker’. The following limits were used: humans, 2000–2009 and diagnosis (sensitivity). We further increased the sensitivity of our search by manually searching through the references of eligible articles and by using the ‘related articles’ feature of PubMed. The search was restricted to the English language.
Data abstraction
Data were abstracted and studies were classified into three groups: (a) those evaluating biomarkers for confirming the diagnosis of established AKI, (b) those evaluating biomarkers for the prediction of AKI, and (c) those evaluating biomarkers for the prognosis of AKI. Within each group, studies were further divided by sample type – urine or blood. The primary endpoints abstracted from each study included the sensitivity, specificity, area under receiver operator curve (AUC) and positive likelihood ratios. In cases where the positive likelihood ratio was not reported, it was calculated using reported sensitivity and specificity.
Quality assessment
The methodological quality of each study was assessed using a modified scoring system based on the STARD criteria. 69,70 The 10 criteria used in our quality assessment were identical to those proposed by Coca et al. 68
Results
We have previously reported the results of our search strategy covering publications from 2000 to 2006. 68 For the present review, we updated the search to include all publications from January 2007 to August 2009. Our search identified 344 new citations published since January 2007, of which 317 were excluded upon title and abstract review (Figure 1). The primary reason for exclusion was the subject matter of studies pertaining to neither AKI nor biomarkers. Of the remaining articles, a further four were excluded after full article review; two articles defined AKI with the very biomarkers whose utility we are investigating, 71,72 one article had less than 20 subjects 73 and one was a subjective letter to the editor. 74 The remaining 23 studies, 75–97 in addition to the 31 studies previously found by Coca et al. 98–128 constitute a total of 54 manuscripts published since 2000 that examine biomarkers in the diagnosis and prognosis of AKI. The studies published since January 2007 examine a further 16 unique biomarkers in addition to the 21 identified by Coca et al. 68 Quality assessment revealed deficiencies in reporting of blinding (52% of studies were deficient), participant sampling (28% used convenience sample) and including a representative distribution of disease in the cohort (19% were deficient), as shown in Table 1.
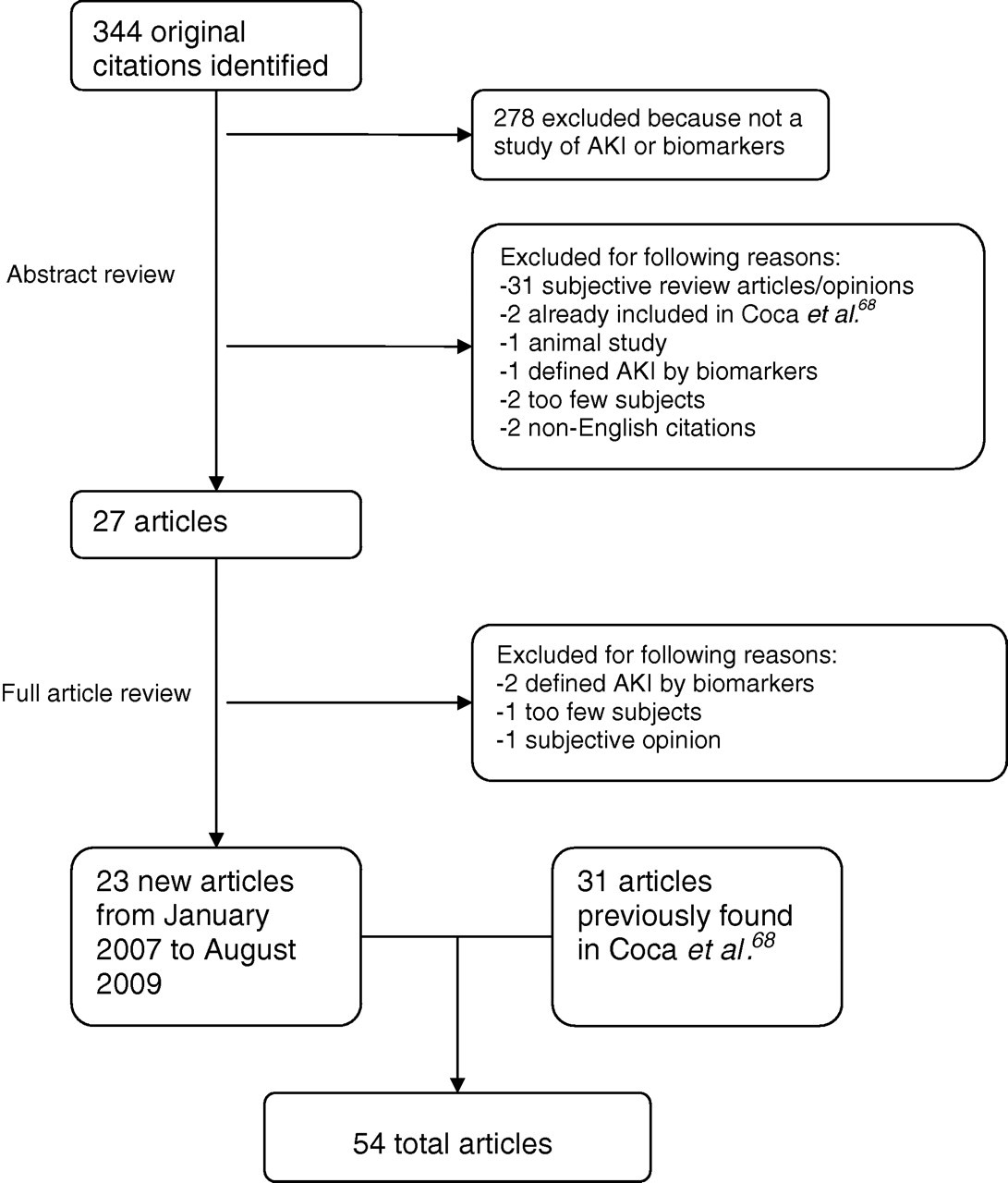
Study flowchart. AKI, acute kidney injury
Scoring system for validity
Biomarkers for the diagnosis of established AKI
Urinary biomarkers that have been quantitatively evaluated in subjects with established AKI include interleukin-18 (IL-18), kidney injury molecule-1 (KIM-1), neutrophil gelatinase-associated lipocalin (NGAL), NAG, matrix metalloproteinase-9 (MMP-9) and sodium–hydrogen exchanger isoform-3 (Table 2). Concentrations of urine IL-18 were significantly increased in patients with established AKI in comparison to those with prerenal azotemia, urinary tract infection or chronic kidney disease, with an AUC of 0.95. 115 Increased urine KIM-1 has also been documented in subjects with established AKI in comparison to those with urinary tract infection or chronic kidney disease, with an AUC of 0.90. 106,107 In this cohort, urinary NAG was also an excellent biomarker for the identification of established AKI (AUC 0.97), whereas MMP-9 performed only fairly (AUC 0.74). Finally, urinary NGAL was found to be increased greater than 25-fold in patients with established AKI when compared with controls or those with chronic kidney disease, but the biomarker properties were not reported. 113
Biomarkers for the diagnosis of established AKI
AUC-ROC (CI), area under the receiver operating characteristic curve (95% confidence interval); NGAL, neutrophil gelatinase-associated lipocalin; IL, interleukin; GST, glutathione S-transferase; NAG, N-acetyl-β-
*This value represents the odds ratio reported rather than the positive likelihood ratio
In the serum, cystatin C is emerging as the best performing biomarker for identifying established AKI, with reported AUCs of 0.88–0.97. 98,101,102,120,122,128 Serum NGAL also correlates highly with the degree of established AKI, but the sensitivities and specificities were not reported. 100,113
Biomarkers for the early prediction of AKI
The quest for biomarkers that can provide an early diagnosis of AKI is an area of intense scholarly interest, with 15 new studies published since 2007 (Table 3). In the urine, NGAL and IL-18 continue to be the most extensively studied biomarkers. Overall, urine NGAL is emerging as an excellent standalone urinary biomarker for the early prediction of AKI, but the results have been mixed depending on the clinical setting and type of population studied. In children undergoing cardiopulmonary bypass surgery, urine NGAL is an outstanding biomarker, with reported AUCs in the 0.93–1 range. 84,112 In adults undergoing percutaneous coronary intervention or cardiopulmonary bypass surgery, the AUCs have ranged widely from 0.61 to 0.96. 76,82,83,90,97,123 In a heterogeneous population of critically ill children, urine NGAL was predictive of subsequent AKI with an AUC of 0.78. 127 In general, the predictive value of urine NGAL appears to be enhanced in children in comparison with adults, with five recent studies reporting AUCs of 0.78–1.00. 84,86,87,112,123 In the setting of adult and paediatric kidney transplantation, urine NGAL was highly successful at predicting delayed graft function with an AUC of 0.9. 116 Finally, a large study of 635 adults admitted to the emergency department illustrated the value of urine NGAL as an initial screening tool for the subsequent development of AKI, with high sensitivity (0.90), high specificity (0.995) and an outstanding AUC of 0.95. 91 This study also examined several other urinary biomarkers, which also performed well but proved to be inferior to NGAL (AUCs of 0.89, 0.83 and 0.71 for α-1 microglobulin, α-1 acid glycoprotein and NAG, respectively).
Biomarkers for the early prediction of AKI
AUC-ROC (CI), area under the receiver operating characteristic curve (95% confidence interrval); NGAL, neutrophil gelatinase-associated lipocalin; IL, interleukin; L-FABP, liver-type fatty acid binding protein; A1M, alpha-1 microglobulin; A1AGP, alpha-1 acid glycoprotein; KIM-1, kidney injury molecule 1; GST, glutathione S-transferase; NAG, N-acetyl-β-
*This value represents the odds ratio reported rather than the positive likelihood ratio
Seven studies of urine IL-18 as an early predictor of AKI have been published to date. 75,83,90,114,116,117,124 The AUCs have ranged from 0.54 to 0.90. In the setting of cardiac surgery, three studies reported very good to excellent AUCs in the 0.75–0.90 range 83,90,117 whereas one study reported poor performance with an AUC of 0.55. 75 Urine IL-18 was an excellent predictor of delayed graft function in subjects undergoing kidney transplantation, with an AUC of 0.90. 116 In general, increases in urinary IL-18 were rarely false-positive (i.e. high specificity), but many patients who develop AKI do not have elevations in urine IL-18 (i.e. low sensitivity).
The ability of urinary KIM-1 to predict AKI has only been evaluated in one published study to date. 107 In a small cohort of children undergoing cardiac surgery, urine KIM-1 predicted subsequent AKI with an AUC of 0.83, which was superior to that of NAG (AUC 0.69) or MMP-9 (AUC 0.50).
Liver-type fatty acid binding protein (L-FABP) has recently been described as another promising urinary biomarker for the prediction of AKI. Urine L-FABP concentrations correlated with serum creatinine in subjects undergoing percutaneous coronary intervention. 79 In children undergoing cardiopulmonary bypass, early urine L-FABP measurements predicted AKI with an AUC of 0.81. 87 Several additional promising urinary biomarkers that predict AKI have been reported in critically ill subjects in a single publication, 125 including γ-glutamyl transpeptidase (AUC 0.95), α-GST (AUC 0.89) and π-glutathione S-transferase (AUC 0.93).
Among serum/plasma biomarkers, NGAL and cystatin C are the most promising for the early prediction of AKI. Four studies in the cardiac surgery setting 85,93,97,112 and one in subjects who developed contrast nephropathy 86 have illustrated the excellent predictive value of early plasma NGAL measurements, with AUCs in the 0.80–0.96 range. In a more heterogeneous critical care setting, the results were less robust, with a reported AUC of 0.68. 88 Two studies in the intensive care setting demonstrated excellent accuracy of plasma cystatin C for the early diagnosis of AKI, with an AUC of 0.90. 99,108 However, a recent publication showed plasma cystatin C to be a poor predictor of AKI in subjects undergoing cardiac surgery, with an AUC of 0.62. 76 In a large study of 876 patients with acute lung injury, a panel of plasma biomarkers that included interleukin-6 (IL-6), soluble tumour necrosis factor -1 and -2 and plasminogen activator inhibitor-1 were found to have significant associations with AKI, but biomarker characteristics were not reported. 77
Biomarkers for the prognosis of AKI
Urinary NGAL within the first five days of hospitalization demonstrated high sensitivity but low specificity for the subsequent need for dialysis in children with haemolytic uraemic syndrome 121 (Table 4). In patients undergoing cardiac surgery, urine NGAL correlated weakly with the duration of AKI (r 2 = 0.22; P = 0.005), and early urine NGAL levels were predictive of the need for dialysis as well as mortality, but the number of events were very small. 84,117 In a heterogeneous critically ill cohort, 127 urine NGAL was only minimally useful for prediction of AKI persistence (AUC 0.63) or for worsening of AKI (AUC 0.61). Similar weakly positive correlations have been reported for urine IL-18 to predict AKI severity. 117,124 Other tubular proteins and enzymes may be of greater utility for prognostication after AKI. Urinary cystatin C (AUC 0.92), α1-microglobulin (AUC 0.86), NAG (AUC 0.81) and retinol-binding protein (AUC 0.80) were all found to predict dialysis requirement in the intensive care setting. 109 In another study of hospitalized patients, there was a dose-dependent association between urinary NAG and the composite endpoint of dialysis or death. 110
Biomarkers for the prediction of AKI severity
AUC-ROC (CI), area under the receiver operating characteristic curve (95% confidence interval); NGAL, neutrophil gelatinase-associated lipocalin; IL, interleukin; L-FABP, liver-type fatty acid binding protein; A1M, alpha-1 microglobulin; CRP, C-reactive protein; KIM-1, kidney injury molecule 1; GST, glutathione S-transferase; GGT, gamma-glutamyl transpeptidase; NAG, N-acetyl-β-D-glucosaminidase; LDH, lactate dehydrogenase; HUS, haemolytic uraemic syndrome; NR, not reported; N/A, not applicable; Paed., pediatric; RRT, renal replacement therapy
*This value represents the odds ratio reported rather than the positive likelihood ratio
†This value represents the hazard ratio of death reported rather than the positive likelihood ratio
Few recent studies have examined serum biomarkers as prognosticators of AKI (Table 4). In critically ill patients with AKI, elevated serum cystatin C levels predicted the need for dialysis with an AUC of 0.76. 108 Other studies of patients with AKI revealed that both low serum prealbumin and increased serum C-reactive protein concentrations were associated with an increased risk of death, with a hazard ratio of 2.1. 80,81
Discussion
This review represents a systematic analysis and critical appraisal of the current status of tubular proteinuria (and the corresponding proteins in the serum) as biomarkers of AKI in humans. This has been an area of very active contemporary research, with 54 high quality manuscripts published since January 2000. These publications describe several promising biomarkers that have been identified, undergone initial clinical testing and are being systematically validated, thereby recapitulating the recently elucidated phases of the diagnostic test development process. 129,130 The biological sources and functions of these biomarkers are shown in Table 5.
Characteristics of promising biomarkers of acute kidney injury
NGAL, neutrophil gelatinase-associated lipocalin; IL, interleukin; L-FABP, liver-type fatty acid binding protein; A1M, alpha-1 microglobulin; KIM-1, kidney injury molecule 1; GGT, gamma-glutamyl transpeptidase; NAG, N-acetyl-β-
A pressing need in the field has been for biomarkers that predict AKI early, well before the increase in serum creatinine concentration, so that clinical trials with timely interventions can be initiated. The evidence suggests that a number of urinary (best exemplified by NGAL and IL-18) and plasma (most prominently cystatin C and NGAL) proteins could serve this purpose, and can now be considered candidates for larger phase 3 and 4 validation studies. NGAL has been the most extensively investigated biomarker for the early diagnosis of AKI. A recent meta-analysis of data from 19 publications involving 2538 patients out of whom 487 developed AKI revealed a diagnostic odds ratio of 18.6 (95% confidence interval [CI] 9.0–38.1) and an AUC of 0.82 (95% CI 0.73–0.89) of NGAL to predict AKI in all clinical settings, 131 suggesting its utility as an excellent standalone marker. It is likely that a combination of NGAL with other strategically selected markers will further enhance this performance, but this is yet to be tested. Panels of biomarkers may also assist in determining the timing of the original insult and duration of the ongoing injury, since their appearance follows a temporal pattern determined by the kidney's response to the noxious stimulus. This concept is similar to the panel used for the prediction of acute myocardial infarction (composed of myoglobin, creatine phosphokinase and troponin), and is illustrated in Figure 2. The data are from previously published manuscripts examining urinary biomarkers following cardiac surgery. NGAL 112 and L-FABP 87 displayed a pattern of rapid increase and subsequent decline, whereas IL-18 117 and KIM-1 107 showed a slightly later rise in concentration. However, all these biomarkers preceded the increase in serum creatinine, which was usually noted 1–3 days after cardiac surgery. Thus, a panel of these novel biomarkers could be useful to determine the timing and duration of AKI, which in turn could assist in determining the primarily aetiology and the most effective treatment strategy.
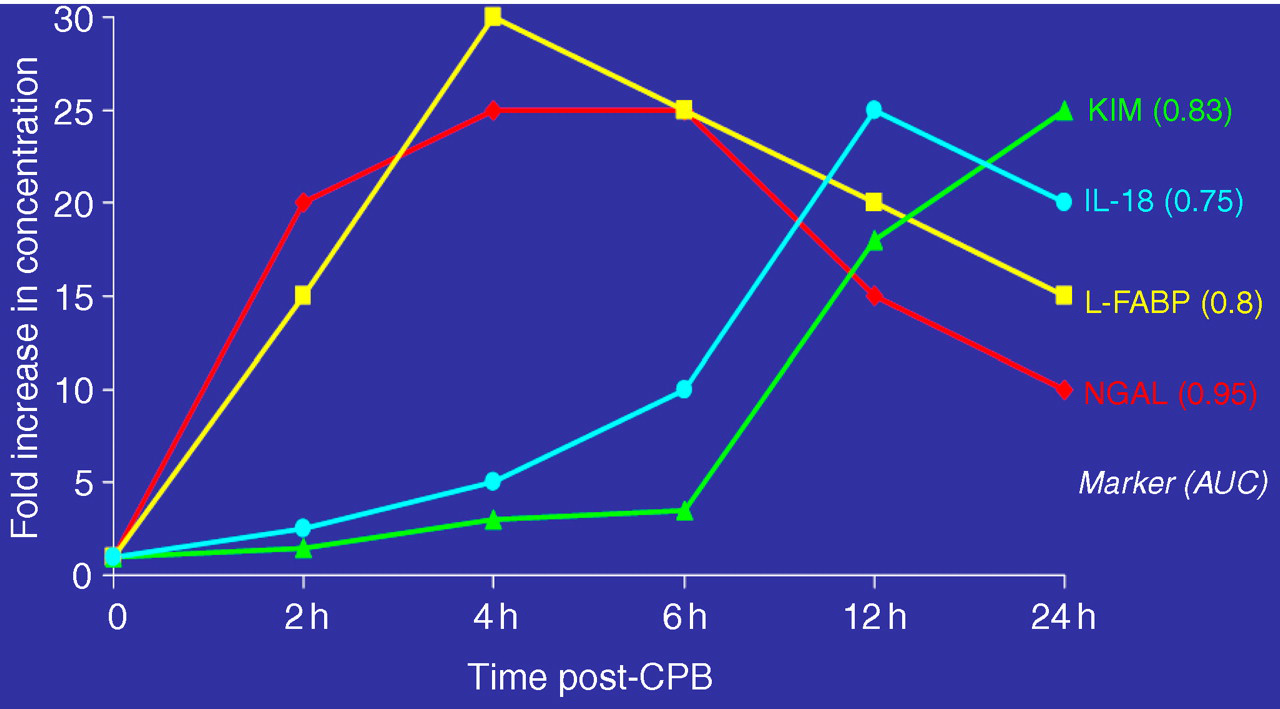
Temporal patterns of increases in urinary biomarker concentrations in subjects who developed acute kidney injury after cardio-pulmonary bypass (CPB). NGAL, neutrophil gelatinase-associated lipocalin 112 ; L-FABP, liver-type fatty acid binding protein 87 ; IL-18 interleukin-18 117 ; KIM, kidney injury molecule-1 107
Such panels may also enable us to determine the principal nephron segment that is injured in any given subject, as shown in Table 5.
Despite the optimism in the field, there are important limitations that exist in the published literature that must be acknowledged. First, this systematic review found that the majority of studies reported were from single centres that enrolled small numbers of subjects. Validation of the published results in large multicentre studies will be essential. Second, most studies reported to date did not include patients with chronic kidney disease. This is problematic, not only because it excludes a large proportion of subjects who frequently develop AKI in clinical practice, but also because chronic kidney disease in itself can result in increased concentrations of most of the biomarkers examined, thereby representing a confounding variable. Third, many studies reported only statistical associations (odds ratio or relative risk), but did not report sensitivity, specificity and AUCs for the diagnosis of AKI, which are essential to determine the accuracy of the biomarkers. Fourth, the majority of biomarkers discovered and investigated to date rely on research-based ELISA-type assays, which are impractical from the clinical utility standpoint. However, assays for cystatin C 108 and NGAL 84,85 are now widely available on standardized clinical platforms, which could facilitate their rapid validation and widespread adoption in the scientific and clinical communities. Fifth, only a few studies with a small number of cases have investigated biomarkers for the prediction of AKI severity, morbidity and mortality – results of testing these biomarkers as predictors of clinical outcomes in large multicentre studies that are already under way are keenly awaited.
Finally, the definition of AKI in the published studies varied widely, but was based largely on increases in serum creatinine, which raises the conundrum of using a flawed outcome variable to analyse the performance of a novel assay. The studies of biomarkers for the diagnosis of AKI may have yielded different results had there been a true ‘gold standard’ for AKI. Instead, using AKI as defined by a change in serum creatinine concentration sets up the biomarker assay for lack of accuracy due to either false-positives (true tubular injury but no significant change in serum creatinine) or false-negatives (absence of true tubular injury, but elevations in serum creatinine due to prerenal causes or any of a number of confounding variables that haunt this measurement). It will be crucial in future studies to understand the clinical outcomes of subjects who may be prone to AKI and are ‘biomarker-positive’ but ‘creatinine-negative’, since this will determine whether the biomarkers are sufficiently sensitive. Since the gold standard for true AKI (tissue biopsy) is highly unlikely to be feasible in a research setting, it is vital that large enough future studies demonstrate (a) the association between biomarkers and hard outcomes such as dialysis, cardiovascular events and death, and (b) that randomization to a treatment for AKI based on high biomarker concentrations results in an improvement in kidney function and reduction of clinical outcomes. This should be the next quest for the field. Additional studies that merely examine the association between biomarkers and clinical AKI will likely not lead to significant progress.
DECLARATION
