Abstract
Background
In an evaluation of androstenedione results from patient serum samples using the Siemens Immulite 2500 analyser and manual Coat-A-Count (CAC) methods, three outliers were evident with grossly elevated results in the CAC assay.
Methods
The clinic notes of three patients with apparently high serum androstenedione concentrations by the CAC assay were checked for medications. The samples were all from patients with polycystic ovary syndrome taking 100–200 mg/d of a steroidal antiandrogen (spironolactone). Two other patients on 50 mg spironolactone per day had less markedly higher androstendione results with the CAC assay. In a further five patients who were selected since they were on spironolactone and had high androstenedione results by the CAC method, spironolactone was temporarily withdrawn and fresh blood samples obtained for analysis.
Results
Spironolactone treatment was associated with higher androstenedione concentrations measured by the CAC assay that reverted to normal on treatment withdrawal. Based on a single test with spironolactone at 1000 ng/mL, the manufacturer reported only 0.109% interference in the CAC assay.
Conclusions
Spironolactone (and/or its metabolites) may interfere in the Siemens CAC assay for androstenedione but not in the Immulite 2500 assay. This experience highlights the need for information from clinicians on drug treatment when laboratory investigations are requested. Drug interferences in immunoassay are common and need evaluation beyond tests performed to certify laboratory reagents.
Introduction
As part of the process of changing a laboratory method, it is usual to compare results from a range of patients with those obtained by the method in current use. While such comparisons often reveal consistent differences in results between the methods, it is sometimes possible to identify one or more outliers that cannot be attributed to error. Collaboration with clinical colleagues to identify the unifying features of such outliers can provide previously unrecognized insights into potential interferences. We describe such a situation here.
Methods
During a preliminary evaluation of a competitive immunoassay for serum androstenedione on the Immulite 2500, we compared results from patient samples previously assayed with the manual Coat-A-Count (CAC) radioimmunoassay (both from Siemens Healthcare Diagnostics Ltd, Camberley, Surrey, UK).
Results
Three outliers with results that were four-fold higher by CAC (33–44 versus 9.2–10.6 nmol/L) were evident among the cohort of 86 patients analysed (Figures 1a and b). These were from female patients with polycystic ovary syndrome on treatment with spironolactone 100–200 mg daily. No previous androstenedione results were available on these patients. Among the cohort we identified two more patients on a lower dose of spironolactone (50 mg daily) whose results were two- to three-fold higher by CAC, a difference that was at the extreme of the range seen among the rest of the cohort (Figures 1a and b).
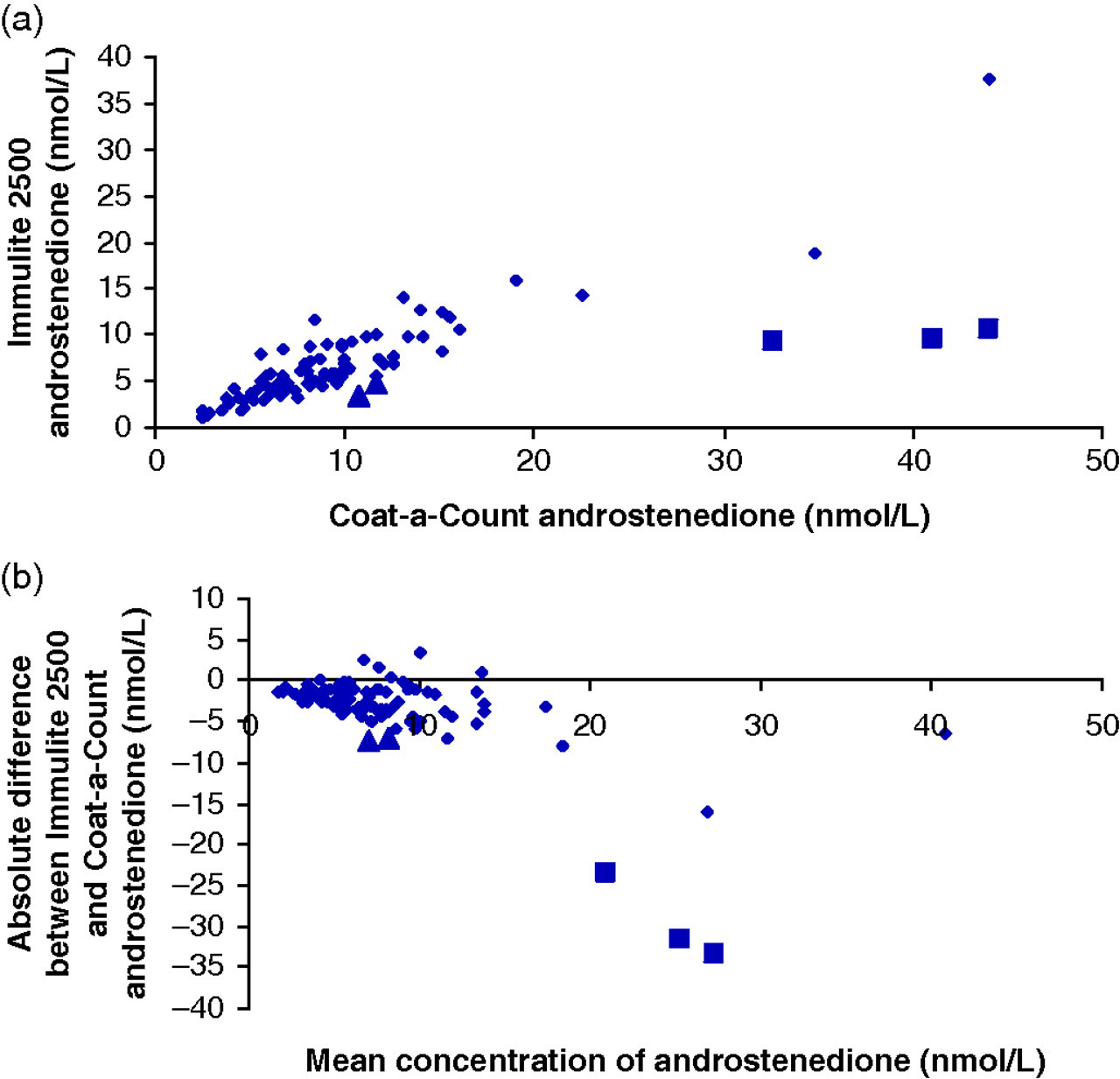
Serum androstenedione results in 86 samples measured on Immulite 2500 and by Coat-a-Count immunoassay. Three outliers (solid squares) are evident on both (a) regression analysis and (b) a difference plot. These patients were all on spironolactone 100–200 mg/d. Two patients on 50 mg/d (triangles) did not show as marked a difference
On historical review, elevated androstenedione concentrations with the CAC method were seen in a number of female patients including five with polycystic ovary syndrome in whom previous results had been normal. The increase in androstenedione was out of proportion to other hormone changes. The patients had been prescribed spironolactone up to 150 mg per day. When spironolactone was withdrawn for one month, serum androstenedione concentrations fell (Table 1). These studies were not standardized for sampling time, drug dose or time of last medication, which may have contributed to variability among patients in the apparent effects of spironolactone.
Endocrine results in five patients with polycystic ovary syndrome off and on treatment with spironolactone
LH, luteinizing hormone; FSH, follicle-stimulating hormone; Testo, testosterone; DHAS, dehydroepiandrosterone sulphate; n/r, not requested
Discussion
The cross-reaction of spironolactone in the CAC assay is not anticipated to be significant. The manufacturer reports cross-reaction to be 0.109% based on an apparent concentration of androstenedione of 1.09 ng/mL (3.8 nmol/L) when 1000 ng/mL of spironolactone was included in the assay. For the Immulite 2500 method, spironolactone cross-reaction was reported to be 0.03% (apparent androstenedione concentration 0.3 ng/mL or 1.045 nmol/L). Note the water solubility of spironolactone is low at 22 mg/L. The detail of the cross-reactivity experiments conducted by the manufacturers is not known.
Spironolactone is an aldosterone antagonist that is traditionally used in the treatment of hypertension, but it also has antiandrogenic effects and is used in the treatment of hirsutism. Spironolactone is rapidly absorbed from the gastrointestinal tract and in the circulation is largely bound to albumin and α 1-acid glycoprotein. Spironolactone gives rise to a large number of metabolites 1 with 7α-thiomethylspironolactone, 6β-hydroxy-7α-thiomethylspironolactone and canrenone as major products in the circulation with half-lives up to 36 h. Interferences in immunoassays for digoxin, 2 aldosterone 3 and 17-hydroxyprogesterone 4 have been reported. Looking at the structures of spironolactone and canrenone (Figure 2), the androstenedione antibodies could be recognizing the C/D ring structure of the drug and or the 7-modified thiol depending on the hapten used for antisera generation that includes a bridge to the protein in either of these areas. Manufacturers of in vitro diagnostics in general are not willing to describe the nature of the hapten and ligand used for assay development. There were insufficient samples to send for analysis by a more specific assay such as liquid chromatography-tandem mass spectrometry. However, it is important to note that for this analysis to be acceptable it would not be sufficient to only measure the concentrations of androstenedione in the samples. A check for ion suppression/enhancement would also be required using a continuous postcolumn infusion of androstenedione into the eluting sample extracts. 5 Testing spironolactone for interference would also have had the problem of low water solubility referred to above.
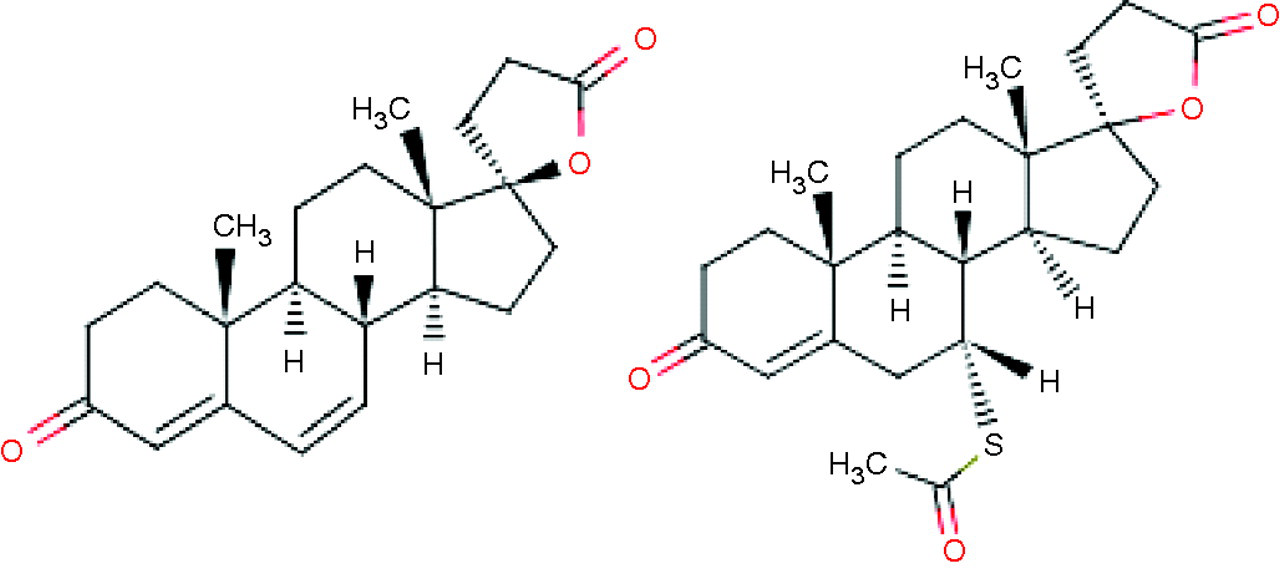
Canrenone (left) and spironolactone (right)
Conclusion
In patients on spironolactone, high concentrations of serum androstenedione were observed when measured by the CAC assay but not by the Immulite 2500 method. Off treatment, the results for androstenedione by CAC were lower. This highlights the possibility that even when very low cross-reactivity of a parent drug can be demonstrated in the assay, this does not preclude clinically significant interference. This study illustrates how drugs and/or metabolites may interfere in immunoassays through binding of related structures to the antibodies. Cross-reactivity data based on single point tests do not always identify compounds at risk of assay interference. Laboratories should have strategies to investigate interferences when suspected. 6 We were able to adopt two strategies using routinely available clinical samples – comparison of different methods and drug withdrawal. Tandem mass spectrometry is seen to be a more specific analysis of steroids but drug interferences can arise unless evaluation excludes co-elution and ion-suppression/enhancement experiments. Based on these findings, requests for androstenedione to be measured with the Siemens CAC assay should be rejected from patients on spironolactone unless clinically relevant interference in the assay has been excluded. While monitoring of androstenedione during treatment with spironolactone may rarely be indicated clinically, our experience is that this request is often made in routine practice (and falsely raised results have caused alarm in our service). Tandem mass spectrometry is seen to be a way to have more specific analysis of steroids and other analytes but even with this new technology, drug interferences can arise unless evaluation excludes co-elution and ion suppression studies.
DECLARATIONS
