Abstract
Background
Short-chain fatty acids such as lactic acid produced by the intestinal bacterial flora have various physiological actions involved in health, and it is important to determine the concentrations of faecal short-chain fatty acids and evaluate their relationship with large intestinal diseases. In this study, we evaluated the highly selective and sensitive simultaneous measurement of both volatile and non-volatile short-chain fatty acid hydrazides using high-performance liquid chromatography (HPLC).
Materials and methods
Faeces treated with ethanol were used as analytic samples. Short-chain fatty acids were measured as fatty acid hydrazides by HPLC.
Results
For 12 types of short-chain fatty acid, the results regarding linearity, recovery tests and reproducibility were favourable. Faeces treated with ethanol could be stored at room temperature.
Discussion
The stability of short-chain fatty acids in faeces at room temperature was statistically analysed. Faeces stored without treatment with ethanol showed increases/decreases in the concentrations of short-chain fatty acids, which may be due to assimilation by intestinal bacteria. However, specimen in 70% ethanol and stored in room temperature exhibited no substantial changes in concentrations of short-chain fatty acids up to seven days.
Introduction
Short-chain fatty acids are produced due to the fermentation of food fibres and indigestible oligosaccharides by a vast number (1014) of 500 types of indigenous microorganism in the large intestine, called the intestinal bacterial flora. The produced short-chain fatty acids are readily absorbed by large intestinal mucosal cells, and metabolized and utilized as energy sources in the liver and muscles. It has been reported that short-chain fatty acids in human faeces are present in a ratio of acetic:propionic:butyric acid of about 60:24:16. 1 Butyric acid as a type of short-chain fatty acid is an essential nutrient for large intestinal mucosal cells, 2 and butyric acid deficiency leads to large intestinal dysfunction, showing that it is essential for human health. Short-chain fatty acids not only serve as an energy source but also exert many physiological actions to maintain health, such as promoting the absorption of water and minerals likely to be deficient, including calcium, magnesium and iron, and inhibiting cholesterol synthesis. Butyric acid also helps to remove mutant cells by inducing their apoptosis. 3,4
In Japan, the incidence of large intestinal diseases has been increasing with Westernization of the dietary lifestyle. It was reported that faecal lactate (non-volatile short-chain fatty acid) was increased in patients with active ulcerative colits, reaching very high concentrations in severe cases, whereas concentrations of other short-chain fatty acids, especially butyric acid (volatile short-chain fatty acid), were reduced. 5,6
Therefore, it is important to measure short-chain fatty acids in human faeces and clarify the relationship between the concentrations of acetic, propionic and butyric, and non-volatile short-chain fatty acids and large intestinal diseases.
Gas chromatography (GC) and high-performance liquid chromatography (HPLC) are used for measurement of short-chain fatty acids, but the sample clean-up procedure for GC is complex in many cases, and derivatization is necessary for the detection of non-volatile short-chain fatty acids. For HPLC, specific derivatization methods are employed for various types of detectors, such as Mass spectroscopy (MS), fluorescence and ultraviolet–visible light (UV–VIS) detectors, but MS and fluorescence detectors are expensive. We employed HPLC using the standard UV–VIS detector in the HPLC system.
Miwa et al. 7 reported an HPLC method in which organic acids were reacted in the presence of 2-nitrophenylhydrazine hydrochloride (2-NPH-HCl) and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (1-EDC-HCl), and detected them as acid hydrazides at UV (230 nm) and VIS (400 nm) light wavelengths. However they did not explore the applicability of the method to measure biological samples such as faeces and serum.
Generally, faeces are stored at −20 to −80°C to avoid changes with time, but the freeze–thaw procedure is complex, causing the samples to deteriorate.
In this study, we investigated highly selective and sensitive simultaneous measurement of non-volatile and volatile short-fatty acid hydrazides based on the study reported by Miwa et al. as well as the faeces storage method.
Materials and methods
Reagents
Thirteen short-chain fatty acids were used: succinic, lactic, acetic, propionic, crotonic, isobutyric, n-butyric, 2-methylbutyric, isovaleric, n-valeric, 2-ethylbutyric (internal standard), isocaproic and n-caproic acids. All were purchased from Wako Pure Chemical Industries (Osaka, Japan).
Stock solutions (200 mmol/L) were prepared by dissolving these acids with 50% aqueous methanol solution and used after dilution when necessary. Pyridine (dehydrated pyridine, Wako Pure Chemical Industries) was adjusted with ethanol to 3% (V/V) (Reagent (1)).
1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (1-EDC) hydrochloride (N-(3-dimethylaminopropyl)-N-ethylcarbodiimide hydrochloride (Sigma-Aldrich, St Louis, MO, USA) and 2-NPH-HCl (Tokyo Chemical Industry, Tokyo, Japan) were adjusted with ethanol to 250 and 20 mmol/L, respectively (Reagents [2] and [3], respectively). Potassium hydroxide (Wako Pure Chemical Industries) was adjusted to 15% (W/V) with water, and used at a potassium hydroxide solution/methanol ratio of 80/20 (Reagent [4]). Phosphoric acid (Wako Pure Chemical Industries) was used as 0.5 mol/L aqueous solution (Reagent [5]). Ethanol, methanol and other solvents in mobile phases were of reagent grade for HPLC (Wako Pure Chemical Industries), and ether was ether 300 for pesticide residue analysis (Wako Pure Chemical Industries).
Derivatization
The internal standard was 2-ethylbutyric acid, and 200 μL of stock solution (200 mmol/L) was diluted with ethanol to 50 mL and used.
Each sample (300 μL) was mixed with 50 μL of the internal standard and 300 μL each of pyridine (Reagent-[1]), 1-EDC-HCl (Reagent [2]) and 2-NPH-HCl (Reagent [3]) solutions as reaction-assistive agents, reacted at 60°C for 20 min, and then mixed with 200 μL of potassium hydroxide solution (Reagent [4]) as a reaction stopper and reacted at 60° for 20 min. After cooling, the mixture was shaken with 3.0 mL of phosphoric acid aqueous solution (Reagent [5]) and 4.0 mL of ether for three minutes for extraction, and centrifuged. The obtained ether layer was shaken with 4.0 mL of water for three minutes and centrifuged. The ether layer was obtained, and ether was eliminated using nitrogen gas. The obtained fatty acid hydrazide was dissolved in 100 μL of methanol, and 30 μL was subjected to HPLC.
Conditions for HPLC analysis
HPLC was performed using Agilent 1100 of HEWLETT PACKARD (Santaclara, CA, USA) and a YMC-Pack FA 250 × 6 mm ID column (YMC, Kyoto, Japan).
The mobile phase was composed of acetonitrile–methanol–water (30:16:54), and the pH was adjusted to 4.5 with 0.1% trifluoro acetic acid (TFA: Wako Pure Chemical Industries, Special Grade). The column temperature was 50°C; flow rate, 1.1 mL/min; and measurement wavelength, 400 nm.
Preparation of analytical faecal samples
Faeces were collected from three healthy adults (2 men and 1 women) after obtaining consent to the study objective from the subjects and approval by the Ethics Committee of BIKEN. Since Reagents (1), (2) and (3) were ethanol solutions, the samples were prepared with ethanol. Faeces immediately after defaecation (about 0.2–0.5 g) was added to 5.0 mL of 70% ethanol contained in a weighed glass centrifuge tube with a screw cap, and the tube as weighed to determine the faecal weight, followed by mixing well using a glass stick and centrifugation at 20°C, 2500 rpm for 10 min, and the supernatant was collected as the analytical sample.
Results
Calibration curves
Twelve standard substances were used: succinic, lactic, acetic, propionic, crotonic, isobutyric, n-butyric, 2-methylbutyric, isovaleric, n-valeric, isocaproic and n-caproic acids. For the preparation of standard solutions, 125 μL each of 200 mmol/L stock solutions were added into a 50-mL measuring flask and mixed with ethanol to obtain standard mixture containing the standards at 500 μmol/L each, and 4, 20, 100 and 500 μmol/L standard mixture solutions were prepared by five-fold serial dilution. Calibration curves (n = 3) produced using the standard mixture solutions showed a good linearity from 4–500 μmol/L for all standards. The calibration curves of the representative fatty acids are shown in Figure 1. The correlation coefficient was ≥0.999 for all standards, and regression analysis showed significant correlations with the regression line (P < 0.05).
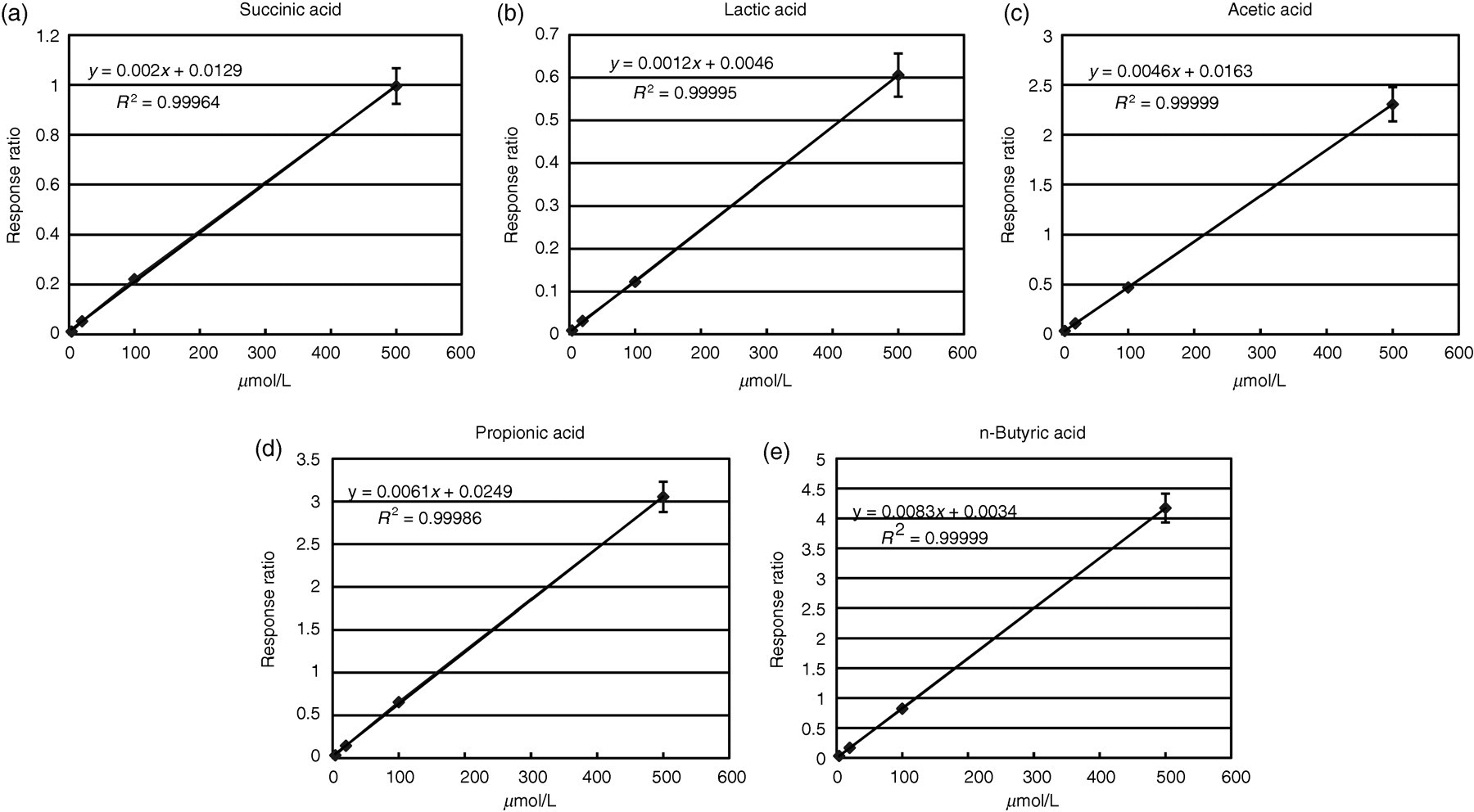
Standard curves of 2-nitrophenylhydrazine delivatization of short-chain fatty acids (succinic, lactic, acetic, propionic and n-butyric acids). Each point represents the mean of three measurements. Error bars show standard error
Recovery test
A blank was prepared by adding 150 μL of water to 150 μL of the faecal sample. Recovery tests (n = 3) were performed after adding 150 μL of 20, 100 and 500 μmol/L standard mixture solutions prepared in ‘Calibration curves’ to 150 μL of the faecal sample.
Figure 2 shows chromatograms of the blank and sample mixed with 150 μL of the mixture containing the standards at 50 μmol/L each.
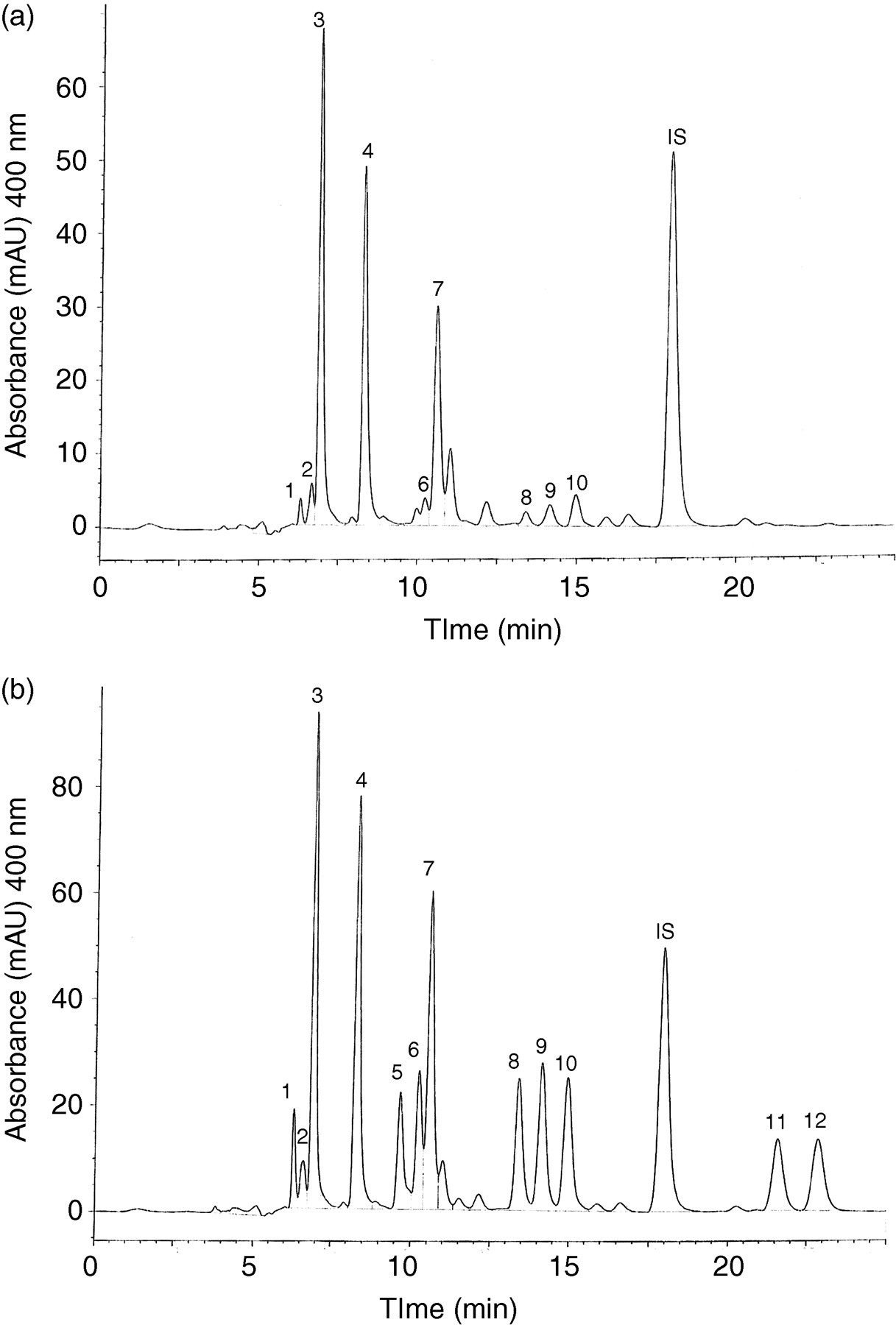
High-performance liquid chromatography chromatograms of the 2-nitrophenylhydrazides of faeces (a) standard mixture of 12 short-chain fatty acids for addition (b) obtained at 400 nm detection. Concentrations of standards are 50 μmol/L and addition volumes are 150 μL. Peaks: 1 = succinic, 2 = lactic, 3 = acetic, 4 = propionic, 5 = crotonic, 6 = iso-butyric, 7 = n-butyric, 8 = 2methylbutyric, 9 = iso-valeric, 10 = n-valeric, 11 = iso-caproic and 12 = n-caproic acid hydrazides
The mean recovery rate was 90–130%. Marked variations (standard deviation, 47–59%) were observed for lactic, acetic and n-butyric acids added at 10 μmol/L. Favourable results (standard deviation, 1–33%) were obtained after addition at 50 and 250 μmol/L.
Serial changes in short-chain fatty acids in human faeces
To avoid changes with time in faeces, the freezing of faeces immediately after defaecation is necessary. However, in the sampling and collection of faeces in the future, transport and storage at home in a freezer may be difficult.
Therefore, stability at room temperature was evaluated using faecal samples processed by the procedure shown in ‘Preparation of analytical faecal samples’ and intact faecal samples were stored at room temperature.
About 2 g of faeces from three healthy subjects was placed in test tubes with a plastic cap. A portion was obtained, and analytical samples were immediately prepared by the procedure shown in ‘Preparation of analytical faecal samples’. The remaining portions were stored at room temperature.
The analytical samples prepared immediately after defaecation were stored at room temperature, and stability was evaluated by measuring the fatty acids immediately after analytical sample preparation (0 h), and 1 (24 h), 2 (48 h), 5 and 7 d. The results are shown in Table 1. From the remaining faeces stored at room temperature, samples were obtained after 10, 24 and 48 h, and 5 and 7 d, prepared by the procedure shown in ‘Preparation of analytical faecal samples’, and subjected to measurement. Changes with time in intact faeces during storage at room temperature were evaluated. The results are shown in Figure 3.
Time changes in succinic, lactic, acetic, propionic and, n-butyric acid concentrations in faeces from three normal individuals (A, B, C). Other short-chain fatty acids are present in trace amounts. The data are the mean ± SEM, * shows a significant difference (P < 0.05) compared with 0 h. Statistical comparison was made using the paired t-test Intra- and inter-day precision for short-chain fatty acids determination in pooled human faecal extract CV, coefficient of variation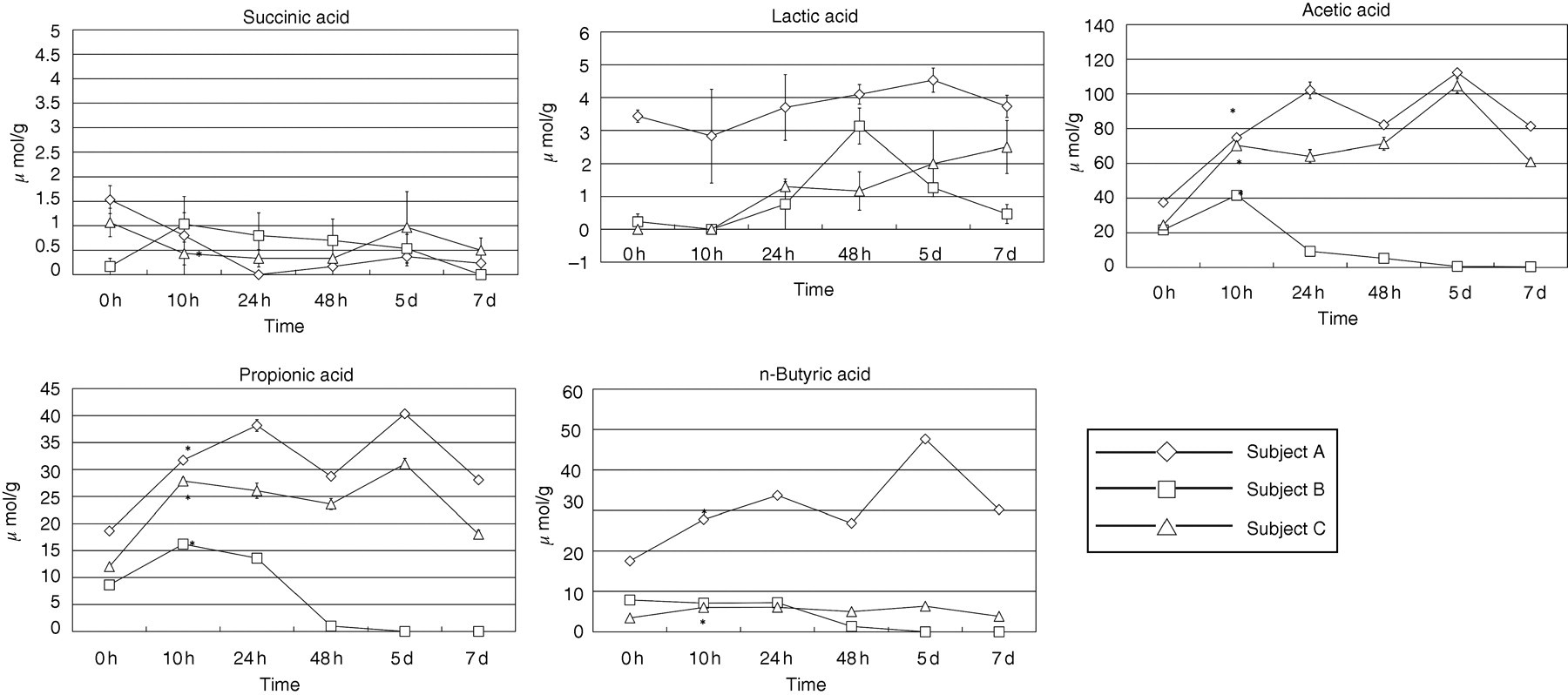
Reproducibility
Analytical samples prepared in ‘Preparation of analytical faecal samples’ were pooled, and the standard mixture prepared in ‘Calibration curves’ was added to this pool sample, followed by dilution with ethanol to low, medium and high concentrations, and they were stored at 4°C as precision control samples.
Using the precision control samples, intra-day (number of samples, n = 10) and inter-day (number of samples, n = 2; 10 days) reproducibility tests were performed. The coefficient of variation (CV) of the measurement values in the precision control samples was 1.5–5.3% in the intra-day and 3.4–8.8% in the inter-day reproducibility tests. The CVs of each short-chain fatty acid are shown in Table 1.
Discussion
Short-chain fatty acids produced by the intestinal bacterial flora through fermentation of food fibres and indigestible oligosaccharides play a vital role in maintenance of colonic integrity and metabolism. However, some short-chain fatty acids, such as lactate, may be deleterious to the colonic mucosa. Thus, the comprehensive short-chain fatty acid profile of human faeces should be useful for understanding the physiological and pathophysiological roles of both volatile and non-volatile short-chain fatty acids, as well as for diagnosing and monitoring various diseases such as ulcerative colits, radiation proctitis, pouchitis and antibiotic-associated diarrhoea. 6
Miwa et al. 7 developed a method to measure concentrations of short-chain fatty acids as acid hydrazides. This method can measure concentrations of both volatile and non-volatile short-chain fatty acids simultaneously with standard HPLC equipment. However, they did not explore the applicability of the method to measure biological samples such as faeces and serum. To utilize this method in the measurement of biological samples, we examined the linearity, reproducibility and interference of this method in faecal samples. Furthermore, to evaluate applicability of this method to mass screening, specimen stability up to seven days was assessed.
Good linearity was obtained from 4 to 500 μmol/L for all fatty acids evaluated. Thus we set this range as our measurement range. The lowest level (4 μmol/L) was determined from s/n ratios of 50–200 at 0.4 μmol/L with a safety margin of 10.
As for the reproducibility, both intra-day (number of samples, n = 10) and inter-day (number of samples, n = 2; 10 d) reproducibility tests produced satisfactory CVs.
In the recovery test, the addition of higher concentrations of short-chain fatty acids (50 and 250 μmol/L) produced favourable results. However, the variation in the recovery rate of lactic, acetic and n-butyric acid added at 10 μmol/L may be due to poor peak separation (lactic acid: 107 ± 34%; n-Butyric acid: 107 ± 29%). To overcome this problem, an increase in the retention time by reducing the flow rate can be considered. However, a long analysis time per sample is necessary, and so it is not practical.
Concerning the stability of faeces at room temperature, a comparison with analytical samples immediately after defaecation (0 h) was performed using the paired t-test and one-way analysis of variance (one-way ANOVA) (Table 2). The paired t-test showed no significant difference from the 0-h analytical sample (P > 0.05, *P > 0.01). However, as a result of one-way ANOVA, one (subject C) of the three subjects showed no significant difference (P > 0.019) in propionic and isobutyric acids but revealed a significant difference (P < 0.0009) in isovaleric acid. Tukey's multiple comparison (significance level, 0.05) was performed for subject C, and significant differences were observed between the values after 24 h and 5 d, between 24 h and 7 d, between 48 h and 5 d, and between 48 h and 7 d (P = 0.008, 0.008, 0.0057 and 0.0057, respectively). The mean values ± SEM after 24 and 48 h were 0.63 ± 0.15 and 0.67 ± 0.15, respectively, being close to the lower measurement limit. It is also possible that the above significant differences were due to problems with the calculation. In the other two subjects, no significant differences were noted in isovaleric acid (P > 0.16), suggesting its stability. The analytical samples prepared as described in ‘Preparation of analytical faecal samples’ were stable at room temperature for at least seven days.
Stability of short-chain fatty acids in 70% ethanol of three human faecal samples at room temperature
Differences among each point were analysed using the paired t-test and one-way ANOVA. Results are expressed as the mean ± SEM (n = 3). Values at each point compared with 0 h are non-significantly different by the paired t–test. P > 0.05, *P > 0.01**shows P < 0.0009 and ***P > 0.019 by one-way ANOVA. Other short-chain fatty acids are present in trace amounts
When the intact faeces were stored at room temperature, and analytical samples were subsequently prepared using the procedure described in ‘Preparation of analytical faecal samples’, changes over time after the storage of intact faeces were evaluated using the paired t-test by a comparison of the fatty acid levels after 0 and 10 h. Regarding volatile fatty acids, the acetic, propionic and n-butyric acid levels were nearly 0 μmol/g after 48 h in one subject (subject B), but the acetic and propionic acid levels after excluding the n-butyric acid level significantly increased after 10 h in the three subjects (P < 0.05, * in Figure 3). Regarding non-volatile fatty acids, the succinic and lactic acid levels tended to decrease and increase, respectively. The succinic acid level significantly decreased in one subject (subject C), but no significant changes were noted in any other fatty acid level (P > 0.05). Degradation by intestinal bacteria was considered for the decreased succinic acid level, while the assimilation of undigested substances in faeces by intestinal bacteria was considered for the increase in the acetic and propionic acid levels, but the details were unclear. For the measurement of short-chain fatty acids in faeces, faecal samples should be processed using the procedure shown in ‘Preparation of analytical faecal samples’ immediately after defecation.
Kotani et al. 8 reported that short-chain fatty acids in faeces can be measured by HPLC without derivatization using an electrochemical detector. We employed HPLC using the standard UV–VIS detector in the HPLC system. Our measurement system required no special detectors like an electrochemical detector. The maintenance of the instrument was easy, and short-chain fatty acids could be simply and accurately measured as hydrazides at a low cost.
In addition, faeces treated with 70% ethanol could be stored at room temperature up to seven days with acceptable stability for the measurements of both volatile and non-volatile short-chain fatty acids. Furthermore, processing faecal samples with ethanol lessened the microbiological risks of microbiological hazards to the laboratory staff. Thus, this finding might be useful for processing many samples, such as those collected on mass screening.
DECLARATIONS
