Abstract
Background
Substance P (SP) and neurokinin A (NKA) are neuropeptides that have been researched as pain markers in adults, as they are involved in transmission and modulation of pain signals. There is a potential role for them as neurochemical markers of pain in neonates, but this has never previously been investigated.
Aim
To establish normative values of SP and NKA in neonates.
Methods
Longitudinal once-daily morning blood samples were collected over two weeks from 142 neonates, gestation 23–40 weeks. Peptides were extracted, and then quantified using an in-house radioimmunoassay. Infants with presumed painful conditions were excluded.
Results
SP concentrations ranged from <0.98 to 11.2 pmol/L (median 1.7 pmol/L) and NKA concentrations from <1.95 to 74.6 pmol/L (median 6.0 pmol/L). Gestation and birth weight had no significant correlation with peptide concentrations. Postnatally, there was a gradual rise in median SP during the first three days, which decreased again by day 14. Median NKA showed a similar rise, but was not statistically significant. This postnatal rise and fall were more apparent in preterm infants ≤32 weeks gestation.
Conclusions
This is the first description of normative values of SP and NKA in neonates. SP and NKA show changes with postnatal age, which are more marked in preterm infants.
Introduction
The diagnosis of pain in an infant remains a challenge in neonatal intensive care. Physiological and behavioural measures exist, such as the Neonatal Facial Coding System, 1 Pain Assessment Tool 2 and Premature Infant Pain Profile, 3 but have limitations in the sickest and most premature infants. They have also been validated for use in acute pain, but not for chronic or persistent pain. Neurochemical measures such as cortisol, catecholamines and beta-endorphin have largely been used in research. Finding a reliable indicator of pain would greatly facilitate its management in neonates.
Substance P (SP) and neurokinin A (NKA) are short peptides of 11 and 10 amino acids, respectively. They are both classified as members of the tachykinin family, 4 which are peptides that have a similar amino acid sequence at the carboxyl-terminus and share a common spectrum of biological activity. Due to high concentrations of these tachykinins in the dorsal horn of the spinal cord, 5,6 it has been postulated that a major role of SP and NKA in vivo is the transmission or modulation of pain signals. Morphine and related analgesics were found to inhibit SP release from sensory terminals in vitro. 7 Later, it was reported that cells that responded most sensitively to SP also responded most strongly to painful stimuli. 8 It appeared that SP has more to do with chronic pain associated with inflammation than with acute pain.
SP and NKA therefore appeared to be potentially useful neurochemical markers of chronic or persistent pain. Prior to this study, only one brief paper on plasma SP concentrations in healthy infants has been produced. 9 However, only ‘healthy’ infants older than three weeks were studied and the samples were random in time and age. SP and NKA have never before been studied in term and preterm infants receiving intensive care.
The initial aims of this study were to establish whether SP and NKA could be detected in samples from neonates, including preterm infants, and to examine whether SP and NKA concentrations were affected by gestation, postnatal age or other antenatal factors. These had to be determined before the tachykinins could be investigated as possible markers of pain.
Methods
Cohort selection
Infants who were born in the Royal Infirmary of Edinburgh, Scotland, were eligible for the study. As there were no previously published data on tachykinin values in neonates, infants of all gestations were included over the study period of 18 months so as to establish ‘normal’ values. Infants with presumed painful conditions, such as necrotizing enterocolitis, meningitis, severe intraventricular haemorrhage and surgery, were excluded. All recruited infants were also scored on the Premature Infant Pain Profile with each sample taken. The study was approved by the Lothian Research Ethics Committee and written informed parental consent was obtained.
Sample handling and analysis
Once-daily morning blood samples were obtained opportunistically when infants were undergoing routine venepuncture. They were collected on days 1, 2, 3, 7 and 14. If an infant did not require any routine blood tests on a study day, then no sample was taken solely for the purposes of the study. All blood samples were taken by two principal investigators (CMW and EMB) into polypropylene microtubes containing 50 μL each of freeze-dried 10,000 KIU/mL aprotinin and 5 mmol/L EDTA. Samples were centrifuged, plasma separated and snap-frozen, all within 10 m of collection. This protocol was based on preliminary studies showing that tachykinin concentrations were stable for only 10 m after collection (CMW, data published in PhD thesis).
Plasma samples were stored at −70°C until analysis. A modified solid phase peptide extraction procedure was essential as there was significant cross-reactivity with unextracted samples (CMW, data published in PhD thesis). Samples were acidified with an equal volume of 1% trifluoroacetic acid (TFA), and centrifuged at 3900
Peptide analysis was by in-house radioimmunoassays performed in polypropylene tubes and specifically developed to detect the small tachykinin concentrations anticipated in neonatal samples. The lower detection limit, defined as the concentration two standard deviations below the average counts at zero binding, was 0.98 pmol/L for SP and 1.95 pmol/L for NKA.
Data and statistical analysis
Data and statistical analyses were performed using SPSS Version 11.0 and analysed using the Mann-Whitney U test, Spearman correlation coefficients (r) or analysis of variance (ANOVA).
Results
Blood samples were obtained from 142 neonates with gestation at birth ranging from 23 to 40 weeks. In neonates, plasma SP concentrations ranged from <0.98 to 11.2 pmol/L (median 1.7 pmol/L) and NKA concentrations from <1.95 to 74.6 pmol/L (median 6.0 pmol/L). For SP, 55 (11%) of 485 samples yielded a result of 0.0 pmol/L. There was a small positive correlation between plasma SP and NKA concentrations (r = 0.13, P = 0.003).
There was no significant correlation between plasma SP concentrations and gestation (r = −0.12, P = 0.2) or birth weight (r = −0.09, P = 0.3). There was also no correlation between plasma NKA concentrations and gestation (r = 0.06, P = 0.5) or birth weight (r = 0.03, P = 0.7).
Postnatally, there was a gradual rise in median plasma SP and NKA over the first three days, which then decreased again by day 14 (Figures 1 and 2). Using a one-way between-subjects ANOVA, this postnatal variation of plasma SP was found to be statistically significant (F[4, 480] = 5.189, P < 0.001). This pattern was more apparent in preterm infants less than 32 weeks gestation (F[4, 314] = 6.146, P < 0.001). The postnatal rise and fall in NKA was not statistically significant.
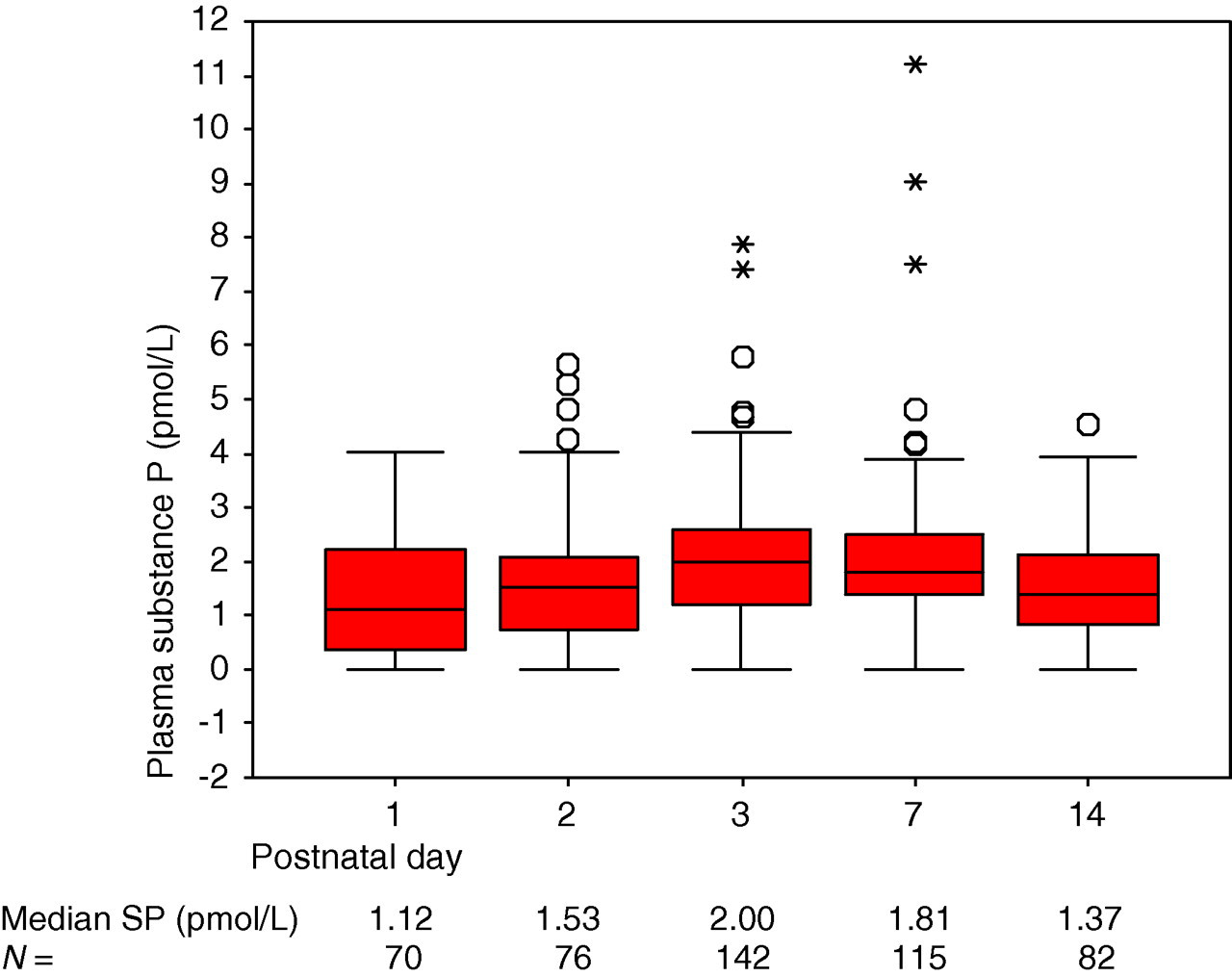
Variation of plasma substance P with postnatal age. The boxes represent interquartile ranges and whiskers represent the full range, apart from outliers and extreme values. O = outlier (a value more than 1.5 box-lengths away from the box); *Extreme value (a value more than 3 box-lengths away from the box)
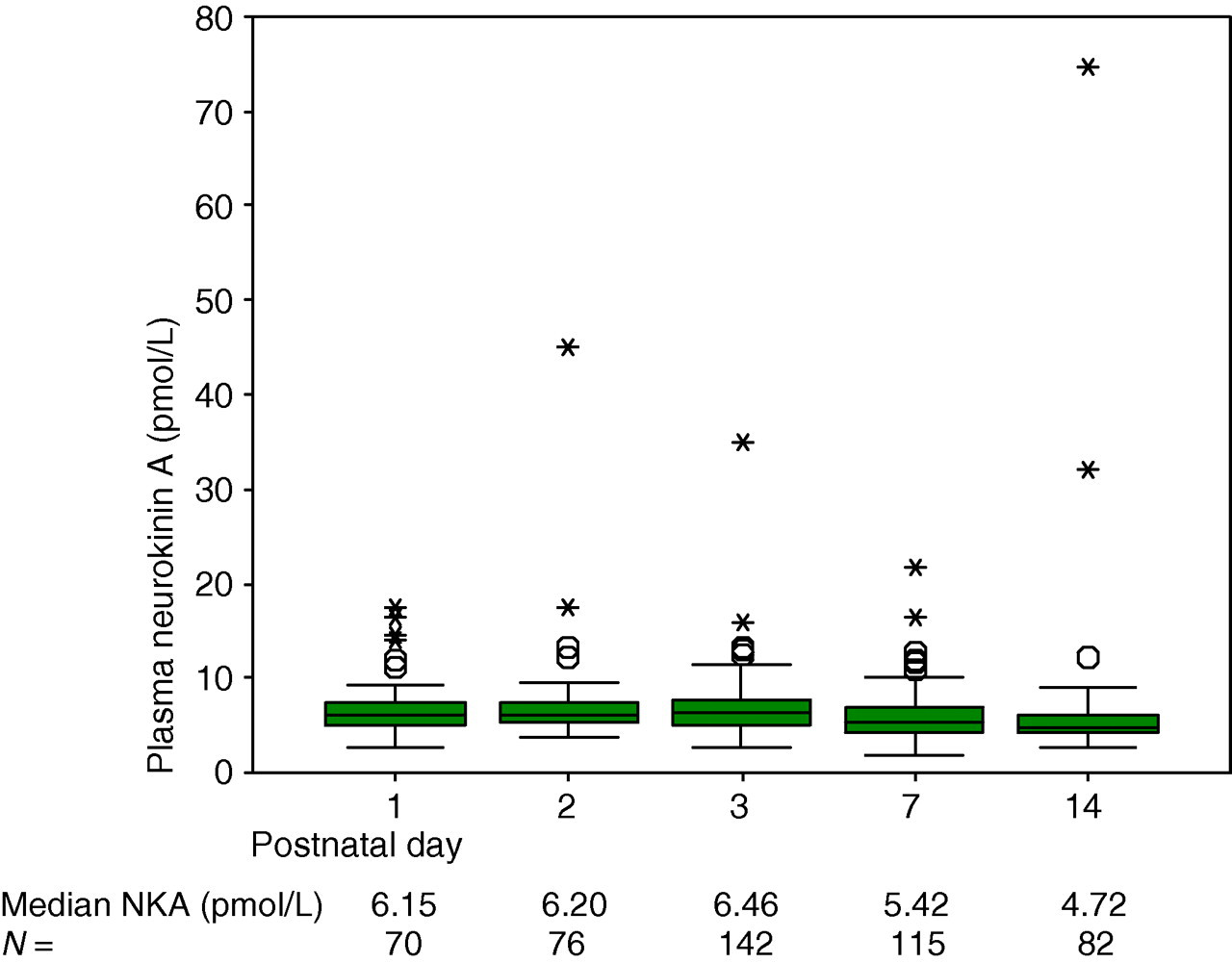
Variation of plasma neurokinin A with postnatal age
Perinatal factors were also examined and found to have no effect on day 1 plasma SP concentrations. These are listed in Table 1. However, plasma NKA concentrations were significantly higher in infants born by caesarean section than by spontaneous vertex delivery (Table 1). Concentrations were also significantly higher in the absence of labour and with the use of maternal epidural analgesia. Other perinatal factors such as use of antenatal steroids and maternal general anaesthesia had no effect, although maternal opiate administration showed a non-statistically significant trend towards lower neonatal plasma NKA concentrations.
Effect of perinatal factors on plasma NKA
CS, caesarean section; NKA, neurokinin A; SVD, spontaneous vaginal delivery
Discussion
The plasma SP concentrations found in neonates in this study were lower than those published elsewhere. Scholle et al. 9 reported mean (standard error) plasma SP concentrations of 20.2 (4.6) pmol/L in 32 preterm infants, and 28.4 (5.7) pmol/L in 19 term infants. 9 However, the results in Scholle's study were from unextracted plasma samples, which can yield higher concentrations of SP due to antibody cross-reactivity. There was also no description of sample handling and preservation procedures, which has been shown to be of importance (CMW, data published in PhD thesis). Furthermore, despite claiming that their study was ‘designed to determine the plasma concentration pattern of SP during the first year of life in full- and preterm infants’, Scholle et al. 9 studied only a small population of infants, and the investigations were performed any time after the third week of life, with results plotted according to the infants' corrected gestational age (which ranged from –4 to 63 weeks [0 being term]). This gave rise to a very heterogeneous group of infants, with a small number of infants having different numbers of plasma samples contributing to the final results.
Within the setting of an observational study utilizing opportunistic blood sampling, it is difficult to obtain blood samples from truly healthy term infants, as this group of infants are likely not to need any blood samples at all. This study therefore seeks to report values from a large group of infants of varying gestational ages who appear free from pain, but not necessarily to establish reference ranges, as this would require multiple blood samples from full-term infants and hence is not ethically possible. There is also the argument of how ‘normal’ a preterm infant is anyway. It would be impossible to find an extremely low birth weight preterm baby who was truly free of all pain, as all these infants require invasive procedures as part of their postnatal stabilization.
Eleven per cent of the plasma SP results from the current study yielded a value of 0.0 pmol/L, but these were probably genuinely undetectable concentrations as NKA remained detectable in all the same samples, with concentrations ranging from 2.6 to 45.0 pmol/L (median 6.6 pmol/L). Moreover, other investigators have also reported undetectable concentrations of plasma SP not only in infants, 9 but also in adults. 10–12 In addition, we now know that unextracted plasma gives higher concentration measurements than extracted samples, and hence the accuracy of other published reports on high plasma SP concentrations (300–500 pmol/L) where sample extraction had not been performed 13–15 may be open to question.
Plasma NKA concentrations were found to be higher than those of plasma SP, and no sample yielded an undetectable result. This is the first study of plasma NKA in newborn infants, hence no comparisons are available. Most of the in vivo research into NKA and pain has been in synovial fluid rather than plasma. 16–19
First day samples from each infant showed no correlation between plasma neuropeptide concentrations, and gestation or birth weight. However, day 1 samples can also be a reflection of perinatal circumstances. Plasma SP concentrations were not affected by perinatal factors, but unexpectedly plasma NKA concentrations were higher in the absence of labour, in infants born by caesarean section and if maternal epidural analgesia was administered (but not general anaesthesia). This was contrary to expectation, as NKA concentrations would be predicted to rise with pain. Nevertheless, although labour is painful to women, it is not known to cause pain to the infant. Also, a normal delivery may be stressful to the infant, but is not known to be painful. It is recognized that labour induces endogenous beta-endorphin production in women 20,21 as well as fetal beta-endorphin levels as determined from cord blood after delivery. 22,23 The fetal beta-endorphins may act as a natural analgesic to the infant. Epidural analgesia reduces the pain of labour to the mother, and has been shown to attenuate maternal beta-endorphin production, but does not affect fetal beta-endorphin levels. 20,21 Maternal opiate administration was associated with a trend towards lower neonatal plasma NKA concentrations, which would be as anticipated.
With postnatal age, both plasma SP and NKA showed a gradual rise over the first three days, followed by a subsequent decline, but these trends were statistically significant only for SP. This pattern was more marked in preterm infants less than 32 weeks gestation. Infants receiving intensive care are more likely to undergo multiple painful or stressful procedures during the first days of initial stabilization, which could explain this finding. Also, the more premature the infant, the more invasive were the procedures required for stabilization.
The current study shows that a weak correlation exists between plasma SP and NKA. The correlation between the neuropeptides is unlikely to be due to cross-reactivity in the radioimmunoassays, as this was <2%. It is more likely to be due to the fact that the same gene codes for both neuropeptides (PPT-A gene) and hence upregulation of the gene for one peptide could result in increased production of the other peptide. NKA is co-synthesized and co-secreted with SP, but has a different regional distribution and asserts its effects via a different receptor to result in different biological functions.
This study's findings with regard to NKA suggest further investigation is warranted. SP appears to be detected in plasma at concentrations too low to ascertain any variation, and perhaps should be measured at a local rather than systemic level. The unexpected variations of NKA with labour, caesarean section and maternal epidural analgesia may best be examined by comparing cord blood samples of neuropeptide concentrations with day 1 samples. The day 1 samples in this study were taken anytime in the first 24 h of an infant's life, hence could have been influenced by postnatal treatments already. It is not clear how NKA concentrations would reflect such perinatal factors and this should be further investigated by studying neuropeptide concentrations in cord blood. It would also be useful to measure concomitant beta-endorphin and cortisol concentrations in the same cord blood samples.
Because they have an immature nervous system, newborn infants experience pain via functional signalling pathways that are not found in mature individuals. Pain signalling occurs via a complex interaction of neurotransmitters and neuromodulators at different levels in the nervous system. As such, it is unlikely that any one neurotransmitter can serve as a ‘marker’ for pain, and that perhaps a measure of variations in a selected set of neurotransmitters may be more informative. As the study of the developmental neurobiology of pain signal processing advances, there is hope that analgesic therapies that are more specific to newborn infants will be developed in the future.
DECLARATIONS
