Abstract
Background
Chlorophosphonazo-III (2,7-bis[4-chloro-2-phosphonophenylazo]-1,8-dihydroxy-3,6-naphthalenedisulphonic acid, disodium salt; CPZ-III) reacts with calcium and magnesium in a sample under acidic to neutral conditions. However, the specific method of measuring calcium in serum using CPZ-III has not been established because of the difficulty of avoiding the interaction between CPZ-III and albumin.
Methods
In this study, we found that the non-specific reaction between CPZ-III and albumin could be controlled and calcium in serum could be specifically detected using CPZ-III combined with vanadate. On the basis of this finding, we evaluated a novel method of serum calcium determination using CPZ-III.
Results
This CPZ-III vanadate method gave linear results from 0 to 7.0 mmol/L. The coefficient of variation was 0.63–0.76%. There was no interference except with Omniscan. There was no change in control performance during 60 d under open-air conditions. The assay results correlated well with those of the Arsenazo-III (2,7-bis(2-arsonophenylazo)-1,8-dihydroxy-3,6-naphthalenedisulphonic acid) method (slope = 1.067; intercept = −0.120; r = 0.989; Sy/x = 0.036 mmol/L), o-cresolphthalein complexone method (slope = 0.911; intercept = 0.186; r = 0.988; Sy/x = 0.035 mmol/L), amylase enzymatic method (slope = 0.981; intercept = 0.072; r = 0.989; Sy/x = 0.036 mmol/L) and inductively coupled plasma emission spectroscopy method (slope = 0.955; intercept = −0.001; r = 0.979; Sy/x = 0.048 mmol/L).
Conclusions
These results suggested that the present method has great clinical potential for measuring calcium.
Introduction
The analysis of calcium in a sample is very important for clinical diagnostics and current methods employ various reagents and devices. The o-cresolphthalein complexone (OCPC) method is most commonly used in both clinical laboratory tests and in the field of general chemical analysis. 1,2 The reagent used in OCPC has a high pH, but this is markedly lowered at the time of use owing to the absorption of carbon dioxide from the air. Moreover, storing the reagent in the form of a solution for a long time decreases the pH, a phenomenon that causes inaccurate measurements in some cases. A reagent based on 2,7-bis(2-arsonophenylazo)-1,8-dihydroxy-3,6-naphthalenedisulphonic acid (Arsenazo-III), which has an improved pH range and a lower pH, has been developed. 3 However, the reagent used in Arsenazo-III is problematic in terms of environmental pollution caused by its disposal because Arsenazo-III is an organic arsenic compound. There are several alternative approaches to calcium determination, such as atomic absorption analysis, 4 enzymatic methods based on amylase, 5 phospholipase D 6,7 or ATPase, 8 and the methylxyleneblue (MXB) method. However, each method has drawbacks. For atomic absorption analysis, for example, the sample must be diluted and thus technical errors may be introduced. The enzymatic method reagent is costly, while MXB has a high pH, which, similar to OCPC, is markedly lowered by carbon dioxide.
In the present situation, it would be extremely useful to develop a reagent for calcium determination that is inexpensive and stable, which depends on a chelating method, and that gives satisfactory performance. We considered that chlorophosphonazo-III (2,7-bis(4-chloro-2-phosphonophenylazo)-1,8-dihydroxy-3,6-naphthalenedisulphonic acid, disodium salt; CPZ-III), 9–11 which is a chelating compound used for the analysis of dried diagnostic reagents, 12 might be a suitable substance to fulfil these criteria. Fergusons et al. 13 has reported that the CPZ-III spectrophotometric procedure can be used to determine additively calcium and magnesium concentrations in the same sample. However, it seems that it is impossible to use CPZ-III in a liquid form because it non-specifically reacts with albumin in samples under acidic to neutral conditions. This reaction is termed ‘metachromasis’, and the absorbance produced can interfere with the absorbance generated by the reaction of CPZ-III with a specific metal compound.
In this study, we considered whether CPZ-III could be used in a new liquid-type reagent as a colorimetric chelating agent that avoids the problems associated with existing methods.
As a result, we found that an intermediate that was formed by a prereaction between CPZ-III and vanadate could be used to avoid the reaction with albumin and specifically detect calcium in serum. We then evaluated the present method of serum calcium determination and compared it with routine calcium measurement methods and the inductively coupled plasma emission spectroscopy (ICP-AES) method.
Materials and methods
Instrumentation and reagents
Instruments
We used the Hitachi 7180 automatic analyser (Hitachi High-Technologies, Tokyo, Japan) for the present calcium measurement assay and other routinely used methods. For the comparison, we used ICP-AES (JY238ULTRACE spectroscope; Horiba, Kyoto, Japan).
Reagents
Malonic acid, sodium dihydrogenphosphate, hydrogenphosphate disodium salt, manganese sulphate, copper sulphate, zinc diacetate, iron(III) chloride, ammonium vanadate, sodium tungstate dihydrate, 8-hydroxyquinoline-5-sulphonic acid, EDTA 2Na, citric acid, sodium fluoride, heparin sodium and heparin lithium were obtained from Wako Pure Chemical Industries Ltd. (Osaka, Japan). Calcium chloride dihydrate, human serum albumin (HSA) and Triton X-100 were obtained from Sigma-Aldrich (St Louis, MO, USA). CPZ-III was obtained from Dojin Labs (Kumamoto, Japan). The control serums used were Consera and Consera A, which were obtained from Nissuiseiyaku (Tokyo, Japan). Interference check A Plus was obtained from Sysmex Corporation (Kobe, Japan). Intralipid 10% intravenous injection was obtained from Terumo (Tokyo, Japan). For non-onic magnetic resonance imaging contrast medium, gadodiamide hydrate: Omniscan was obtained from Daiichi Pharmaceuticals (Tokyo, Japan). Meglumine gadopentetate: Magnevist Syringe was obtained from Nihon Schering (Osaka, Japan). Gadoteridol: ProHance was obtained from Bracco-Eisai (Tokyo, Japan). The Arsenazo-III reagent kit was obtained from NIPRO (Osaka, Japan). The amylase enzymatic reagent kit was obtained from Toyobo Co, Ltd (Osaka, Japan). The OCPC reagent kit and calcium standard solution were obtained from Nittobo Co, Ltd (Tokyo, Japan).
Specimens
Serum samples (130 samples) were collected from patients who provided informed consent at Nitto Hospital. These samples were then analysed anonymously by the person in charge of the tests. All samples were stored at −80°C until used for analysis.
Calibration
The present method, the OCPC method and the ICP-AES method were calibrated with calcium standard solution. Other methods were calibrated with each manufacture's calibrator.
Procedure
Analytical conditions
This assay was carried out with the Hitachi 7180 automatic analyser using the following established procedure for this machine. In this assay, 2.5 μL of sample was mixed with 200 μL of reagent 1 in an assay cuvette. After five minutes, 50 μL of reagent 2 was added. A two-point end assay was used to measure the absorbance change between before and after the addition of reagent 2 at a primary wavelength of 660 nm and a secondary wavelength of 800 nm.
Reagent compositions
In this assay, we considered that an intermediate compound that was formed by a prereaction between CPZ-III and a metal ion could avoid a reaction with albumin. Therefore, we investigated the addition of some metal ions (manganese sulphate, copper sulphate, ammonium vanadate, sodium tungstate dihydrate) added to reagent 2 including CPZ-III.
Each added reagent and reagent without metal ion were evaluated by measuring with a calcium standard solution and a control serum. In addition, the obtained measurement values were compared with measurement values obtained using the OCPC method.
Reagent 1 contained 200 mmol/L malonic acid buffer (pH 5.0), 150 mmol/L sodium chloride, 1% Triton X-100 and 2.5 mmol/L 8-hydroxyquinoline-5-sulphonic acid. Reagent 2 contained 20 mmol/L sodium dihydrogenphosphate/hydrogenphosphate disodium salt buffer (pH 8.0), 150 mmol/L sodium chloride, 1% Triton X-100, 1.25 mmol/L CPZ-III and 40 mmol/L each of metal ion. 8-Hydroxyquinoline-5-sulphonic acid was added to reagent 2 as a magnesium blocker. 14 For reagent 1, pH 5.0 was selected to avoid the effects of carbon dioxide. For reagent 2, pH 8.0 was selected to stabilize the substrate in storage. The final pH in the reaction mixture was pH 5.2, which was optimal for sensitivity, specificity and low reagent blank.
Technical characterization of the CPZ-III vanadate method
Within- and between-run precision
The precision of the present method was determined using two kinds of known concentration control serums (Consera and Consera A). Within-assay coefficient of variations (CVs) were determined with 20 replicates of each sample. Between-assay CVs were determined from assays performed on nine different days.
Linearity and detection limit
To prepare a set of samples that would provide linear results, 7.0 mmol/L calcium chloride dihydrate was added to deionized water. This high-calcium sample was serially diluted 10-fold with saline. We estimated the detection limit of the presented method by assaying zero concentration (saline) 10 times.
Accuracy evaluation
Accuracy evaluation used SRM909b-1 and SRM909b-2 (National Institute of Standards and Technology, Gaithersburg, MD, USA). These serum samples were assayed on five separate occasions.
Recovery test
The three original samples used for recovery testing were serum samples that were drawn from in-house volunteers who provided informed consent. Three concentrations of calcium chloride solution were added to the each sample. The samples to be analysed were then mixed with the original samples at a ratio of 1:9 (v/v).
Interferences
Potential interference materials were added to the control serum (Consera) up to the following concentrations: haemoglobin (up to 5000 mg/L), free bilirubin (up to 400 mg/L), ditauro-bilirubin (up to 400 mg/L), chyle (up to 3000 formazine turbidity units), intralipid (up to 5%), ascorbic acid (up to 500 mg/L), Mg2+ (up to 32.9 mmol/L), Fe2+ (up to 0.1 mmol/L), Cu2+ (up to 0.1 mmol/L), Zn2+ (up to 0.1 mmol/L), EDTA 2Na (up to 15 mmol/L), citric acid (up to 10,000 mg/L), sodium fluoride (up to 5000 mg/L), heparin sodium (up to 2000 mg/L) and heparin lithium (up to 2000 mg/L); Omniscan (up to 0.2 mmol/L), Magnevist (up to 0.2 mmol/L) and ProHance (up to 0.2 mmol/L) were also tested. In addition, HSA was dissolved at various concentrations (0–9.1 g/dL) using 2.5 mmol/L calcium chloride solution.
Stability under open-air conditions
Control serums were measured over 60 d (0, 1, 2, 7, 14, 21, 28, 45 and 60 d) with reagents 1 and 2 analysed using the Hitachi 7180 automatic analyser under open-air conditions. The absorbances of saline (reagent blank) and calcium standard solution were also measured at the same time.
Correlation with other methods
Correlations were measured by comparing our results with those obtained using the Arsenazo-III method, the amylase enzymatic method, the OCPC method and the ICP-AES method. We performed regression analysis and produced Bland–Altman difference plots.
Statistical analysis
All statistical analyses were performed using Microsoft Excel.
Results
Reagent compositions
The initial investigation demonstrated that the most suitable metal ion to use is vanadate. On the other hand, reagent 2 samples containing other metals and without metal ions could not be used, for example, because the calibration curve did not work or positive error was obtained. Additionally, we examined vanadate added to reagent 2 at concentrations from 0 to 60 mmol/L. Applying over 40 mmol/L of vanadate to reagent 2 provided measurement values approximately equal to those obtained with the OCPC method (Figure 1a). Reagent 2 containing 40 mmol/L of vanadate completely inhibited the reaction with albumin (Figure 1b). Owing to the solubility of vanadate in reagent 2, we decided to add 40 mmol/L vanadate to reagent 2.
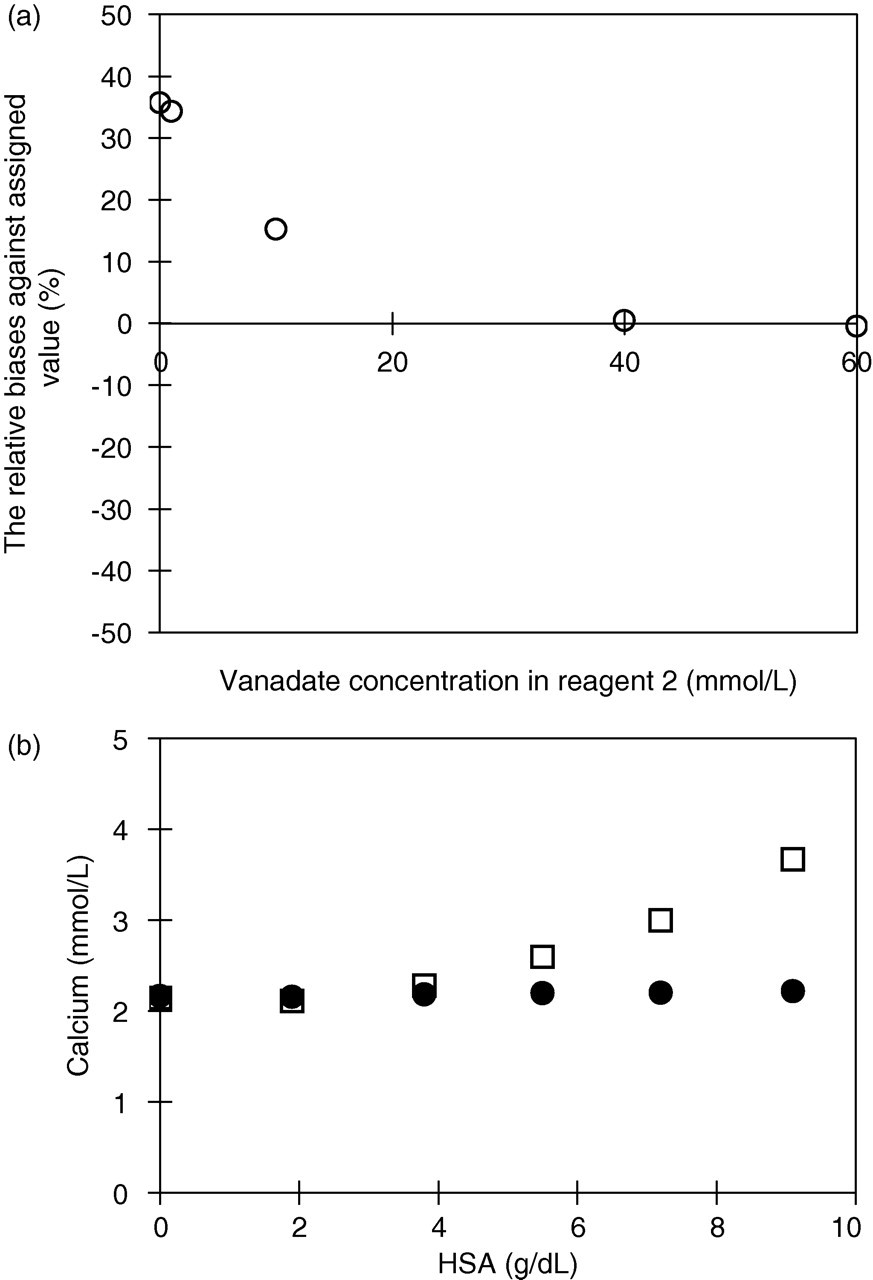
(a) Optimum vanadate concentration in reagent 2. Applying over 40 mmol/L of vanadate to reagent 2, the relative bias levels of these serum samples against assigned value was less than 0.5%. (b) Interference of human serum albumin. (•) Reagent 2 containing 40 mmol/L vanadate; (□) reagent 2 without metal ion
Within- and between-run precision
The within-run CV was 0.63–0.76% and the between-run CV was 0.79–0.94% (Table 1).
Within- and between-run precision
CV, coefficient of variation
Linearity and detection limit
The linearity of the results of the assay is shown in Figure 2. The present method gave linear results from 0 to 7.0 mmol/L (Y = 0.704X + 0.107, r = 0.999). The detection limit, defined as the calcium ion concentration for zero concentration + 3 standard deviation, was 0.025 mmol/L.
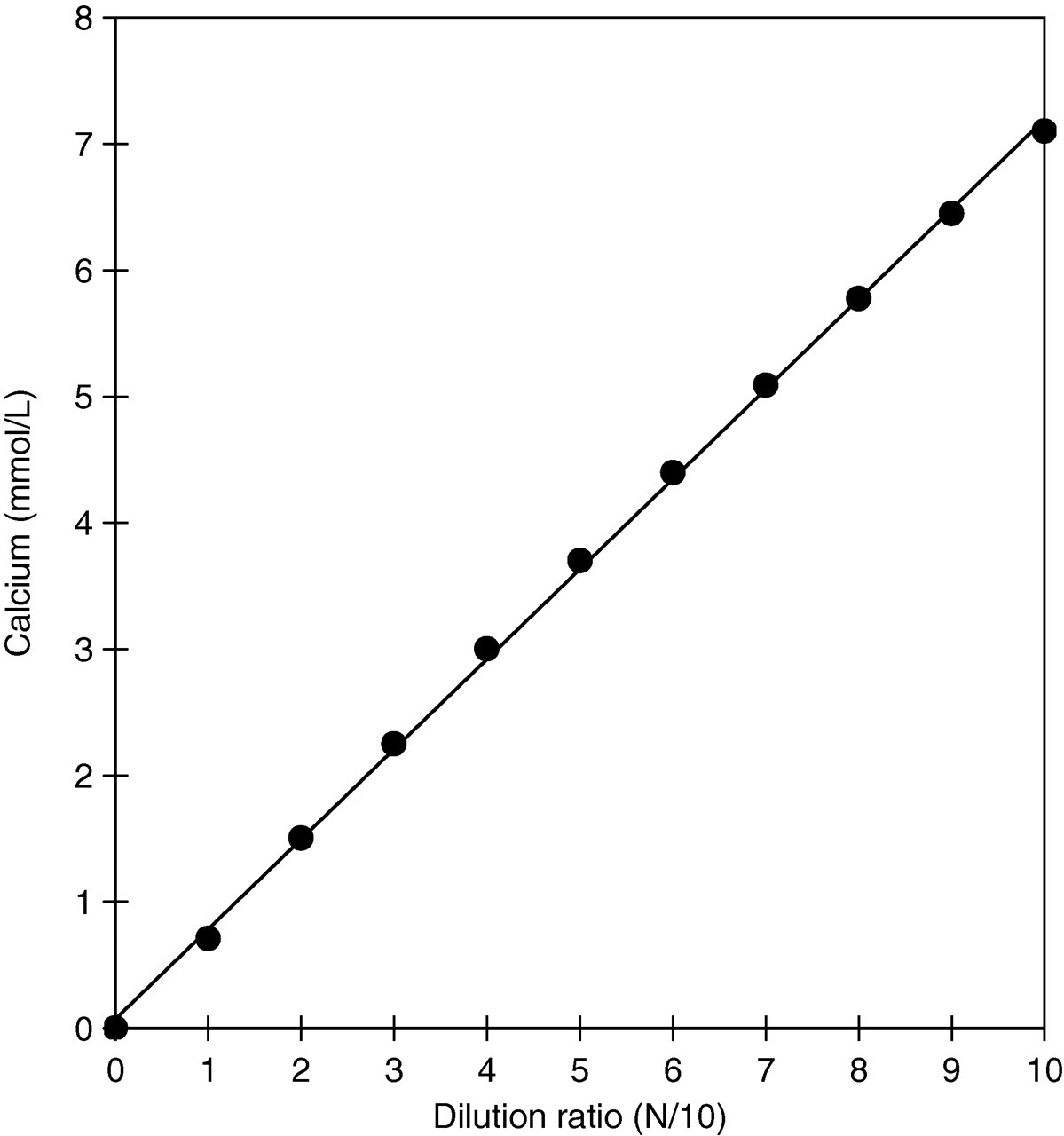
Linearity of results from the CPZ-III vanadate method. This high-calcium sample was diluted 10-fold with saline. The results showed that the linearity was Y = 0.704X + 0.107, r = 0.999. CPZ-III, chlorophosphonazo-III (2,7-bis[4-chloro-2-phosphonophenylazo]-1,8-dihydroxy-3,6-naphthalenedisulphonic acid, disodium salt
Accuracy evaluation
The mean results of SRM909b-1 and SRM909b-2 by the present method were 2.19 and 3.52 mmol/L, respectively. The relative bias levels of these serum samples for each labelled value were −0.70 to −1.4%.
Recovery test
Recovery was 97.6–103.2% (Table 2).
Recovery of calcium in serum
Interferences
No interference was observed in the assay (less than 5% change in calcium concentration) at normal administration levels, except with Omniscan. Omniscan showed slightly positive interference (greater than 5% change in calcium concentration).
Stability under open-air conditions
There was no change in the measured values of controls or reagent blanks, or in sensitivity over 60 d under open-air conditions (Figure 3).
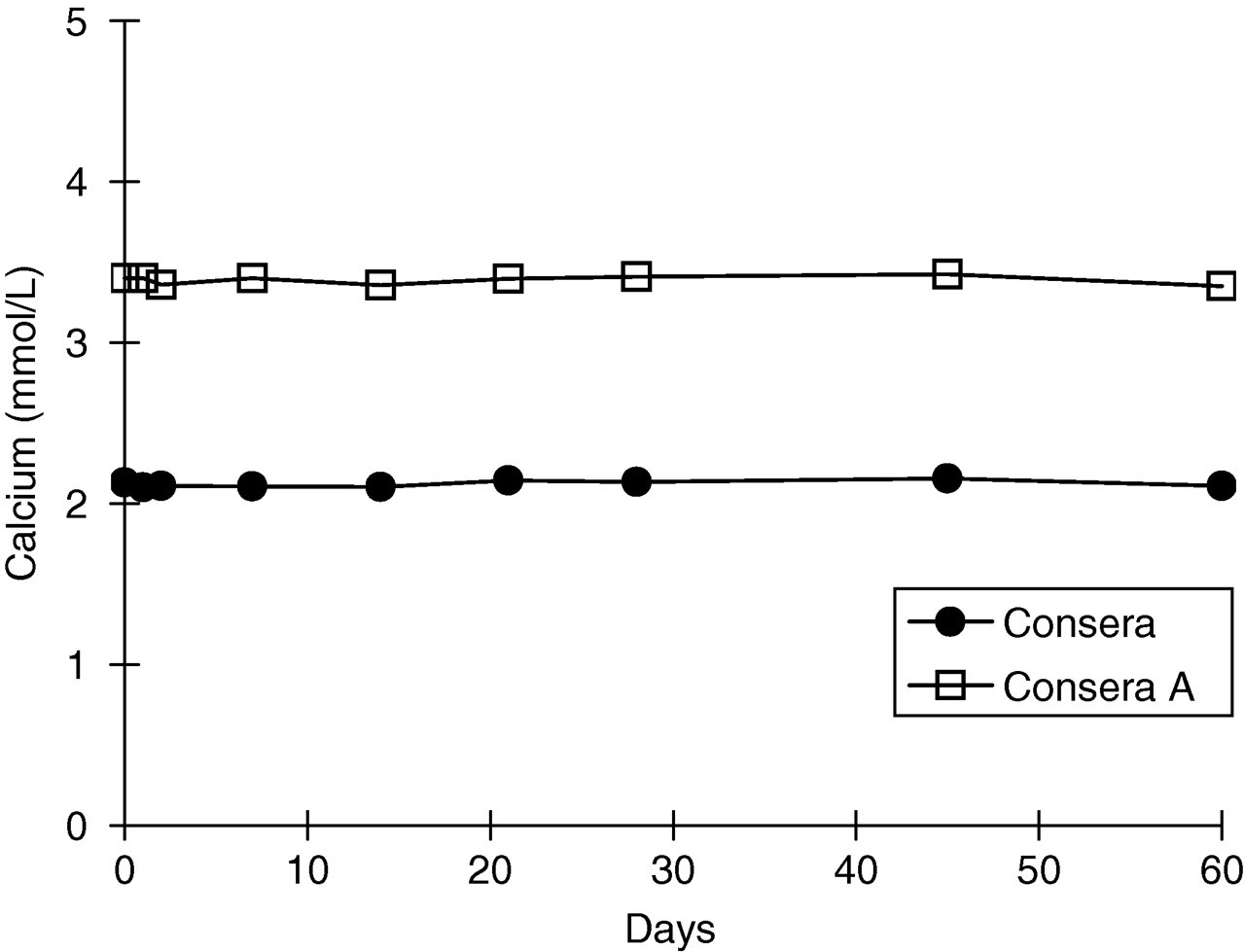
Stability under open-air conditions. The figure describes the performance of the assay in the analysis of control serum for 60 d
Correlation study
In the correlation study, the CPZ-III method results agreed with those of the Arsenazo-III method (slope = 1.067; intercept = −0.120; r = 0.989; Sy/x = 0.036 mmol/L), the OCPC method (slope = 0.911; intercept = 0.186; r = 0.988; Sy/x = 0.035 mmol/L), the amylase enzymatic method (slope = 0.981; intercept = 0.072; r = 0.989; Sy/x = 0.036 mmol/L) and the ICP-AES method (slope = 0.955; intercept = −0.001; r = 0.979; Sy/x = 0.048 mmol/L), as shown in Figure 4. Results showed good correlation with the present method.
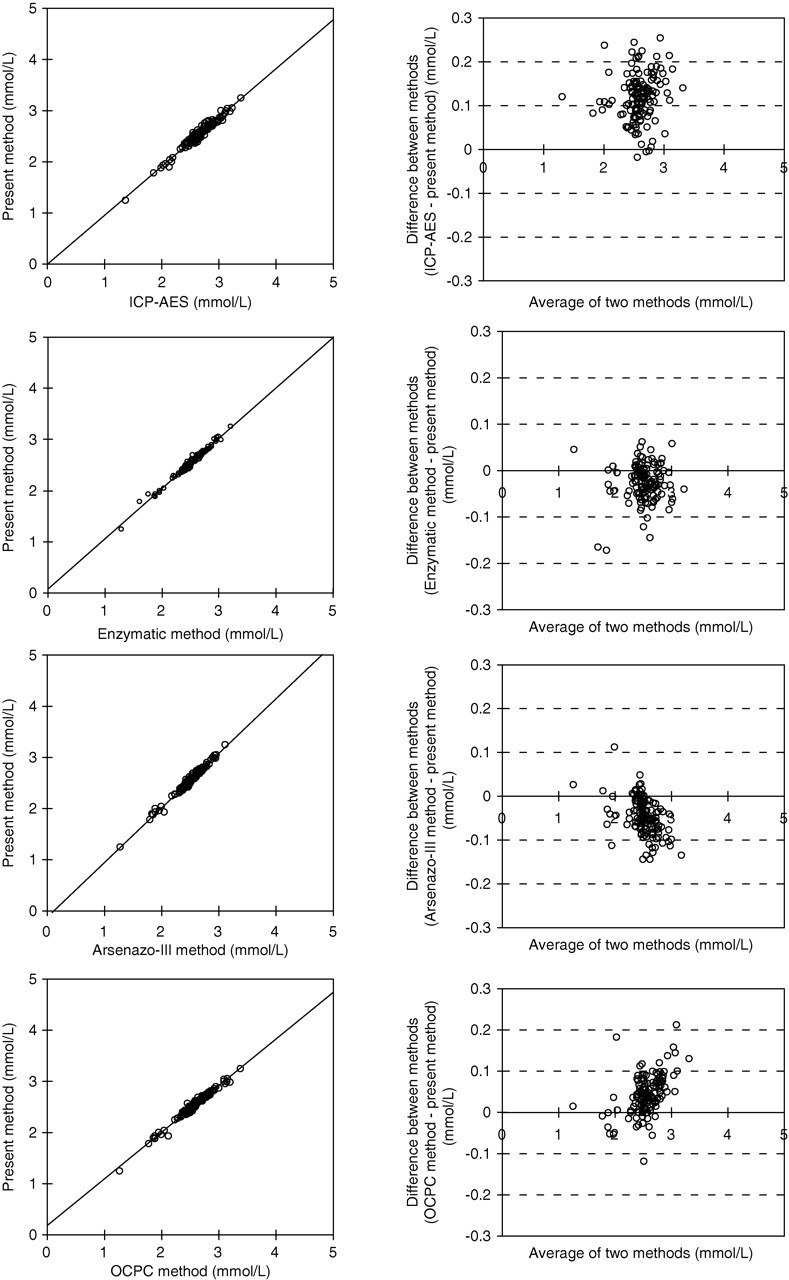
Correlation study and Bland–Altman difference plots. Correlation between the results of CPZ-III vanadate method and those of the ICP-AES method (Y = 0.955X − 0.001; r = 0.979; Sy/x = 0.048 mmol/L), the Arsenazo-III method (Y = 1.067X − 0.120; r = 0.989; Sy/x = 0.036 mmol/L), the amylase enzymatic method (Y = 0.981X + 0.072; r = 0.989; Sy/x = 0.036 mmol/L) and the OCPC method (Y = 0.911X + 0.186; r = 0.988; Sy/x = 0.035 mmol/L). CPZ-III, chlorophosphonazo-III (2,7-bis[4-chloro-2-phosphonophenylazo]-1,8-dihydroxy-3,6-naphthalenedisulphonic acid, disodium salt; ICP-AES, inductively coupled plasma emission spectroscopy; Arsenazo-III, 2,7-bis(2-arsonophenylazo)-1,8-dihydroxy-3,6-naphthalenedisulphonic acid; OCPC, o-cresolphthalein complexone
Discussion
CPZ-III is a compound that contains no arsenic atoms and has been previously used in the dry calcium analysis method. CPZ-III is also reported to be a chelating agent that can react with calcium under acidic to neutral conditions. Furthermore, on the basis of a comparative study of the binding constants of various chelating agents with calcium, Matsuda have reported that CPZ-III can react with calcium more sensitively and can form a chelating compound with calcium more stably than Arsenazo-III. 15 CPZ-III is hardly affected by carbon dioxide in the air and also performs adequately for practical use. However, in our experience, the CPZ-III method has a major drawback in that it reacts non-specifically with albumin in samples under acidic to neutral conditions, forming a coloured product that has the same maximum absorption (669 nm) as that of the chelating compound formed with calcium.
In the case of the dry method, this problem seems to be solved by incorporating a protein removal step during the sample spreading process in the dry testing medium. In order to develop a calcium measurement method based on CPZ-III in solution, it is necessary to avoid this non-specific reaction between CPZ-III and albumin in the sample. We found that the non-specific colour-forming reaction between CPZ-III and albumin was completely inhibited when vanadate ions were prereacted with CPZ-III. The association between CPZ-III and vanadate is stronger than that between CPZ-III and albumin, and is weaker than that between CPZ-III and calcium. Therefore, the vanadate is removed from CPZ-III in accordance with the concentration of calcium, and the CPZ-III then combines with calcium to produce a colour-forming compound.
According to the recovery test and the accuracy evaluation, the present method has good accuracy. Furthermore, there was no interference with HSA. These findings mean that there is no influence of serum proteins or of other metals including magnesium. In addition, interference from coloured molecules, such as haemoglobin, bilirubin or chyle, did not affect the present method because the measurement wavelength is 660 nm, which differs from the absorption wavelength of these compounds. In a plasma sample, some chelating agents have the effect of masking calcium. 16 The present method is not influenced by chelating agents such as EDTA 2Na and citric acid. If a plasma sample containing a chelating agent were mixed with a reagent containing vanadate, the calcium bound to the chelating agent would be substituted by the vanadate in the reagent. As a result of the reaction between CPZ-III and substituted calcium, the correct calcium concentration could be obtained even if other chelating agents were added to the samples. This is why almost all chelating agents usually have a larger stability constant with vanadate than with calcium. For all the contrast agents except Omniscan, there was no interference in the present method. For Omniscan, CPZ-III binds with gadolinium and gives a colour reaction 17,18 because vanadate combines with the diethylenetriaminepenta-acetic-acid-bis-methylamide included in Omniscan. Therefore, it is necessary to be aware of whether a patient has been given Omniscan before any measurements of serum calcium. The results of the present method exhibited good linearity from 0 to 7.0 mmol/L. There was no change in the results of controls, reagent blanks or in the sensitivity over 60 days of open-air testing. In other words, there was no influence by the surroundings because this method involves acidic reaction conditions that are negligibly influenced by the carbon dioxide in the air. The results of the present method correlated well with those of routine calcium measurement methods and the ICP-AES method. 19–21 The ICP-AES method was developed as an advanced technique of atomic absorption (NIST reference method). 22,23 Our data showed that the present method has good accuracy. For the OCPC method, a negative slope and a positive intercept were observed owing to the lower estimations at low calcium concentration ranges, as extensively reported elsewhere.
Fergusons reported that CPZ-III can react with alkaline earths (particularly calcium and magnesium) in biological fluid samples and form a coloured chelation compound with a maximum absorption at 669 nm under acidic to neutral conditions. CPZ-III also reacts with albumin in serum and forms a coloured product with a maximum absorption at 669 nm under the same conditions. We have shown that the formation of the coloured product with albumin can be avoided if CPZ-III is used with vanadate. This new reagent for measuring calcium does not show changes in the measured values on open-air testing. The present method is reasonably inexpensive and performs adequately for practical use. Thus, it should be useful in routine calcium measurements in clinical laboratories.
DECLARATIONS
