Abstract
Background
Short synacthen tests (SSTs) are frequently performed in medical inpatients with suspected adrenocortical insufficiency. The utility of a random or baseline serum cortisol in this setting is unclear. We determined random cortisol thresholds that safely preclude SSTs in acute medical admissions.
Methods
We analysed SSTs in acute non-critically ill general medical patients (n = 166, median age 66, range 15–94 y; men 48%, women 52%). The SST was defined according to the 30-min cortisol as ‘pass’ (>550 nmol/L) or ‘fail’ (≤550 nmol/L). Receiver operating characteristics (ROC) curves were generated to determine the predictive value of the basal cortisol for a failed SST.
Results
Of 166 SSTs, a pass was seen in 127 (76.5%) tests, while 39 (23.5%) tests failed the SST. ROC curves showed that no single cut-off point of the baseline cortisol was adequately both sensitive and specific for failing the SST despite a good overall predictive value (area under curve 0.94; 95% confidence interval 0.89–0.98). A basal cortisol <420 nmol/L had 100% sensitivity and 54% specificity for failing the SST, while a basal cortisol <142 nmol/L had 100% specificity and 35% sensitivity. Restricting the SST to patients with a basal cortisol <420 nmol/L would have prevented 44% of SSTs while correctly identifying all patients who failed the SST.
Conclusion
A baseline serum cortisol may prevent unnecessary SSTs in medical inpatients with suspected adrenocortical insufficiency. However, SSTs are still indicated in patients with random cortisol <420 nmol/L, or where the suspicion of adrenal insufficiency is compelling.
Introduction
The short synacthen test (SST) is commonly used as a screening test of adrenocortical insufficiency in medical inpatients with acute non-critical illness. In this setting the insulin tolerance test (ITT), the gold standard investigation of the hypothalamo-pituitary-adrenal (HPA) axis, is impractical and potentially harmful. Thus, clinical decisions often rest on the outcome of the SST. The conventional SST is based on the adrenal response to synthetic adrenocorticotropic hormone (250 μg, intramuscularly or intravenously) and most laboratories would consider a 30-min postsynacthen cortisol >550 nmol/L as indicative of normal adrenocortical reserve. 1,2 Its excellent safety profile notwithstanding, the SST has the additional requirements of parenteral injections and precisely timed sampling and is also contraindicated in patients with a history of atopy. Thus, unnecessary requests will pose added demands on busy inpatient units and increase the potential for serious adverse reactions. Accordingly, some authors have proposed that a baseline cortisol may circumvent the need for the SST in a proportion of patients. 3
However, evaluations of the basal cortisol have mostly been undertaken in controlled outpatient environments 3 or postpituitary surgery settings. 4 In contrast, the utility of the baseline cortisol in medical inpatients has so far received scant attention. Basal cortisol thresholds that safely preclude further testing for adrenal insufficiency in acute medical admissions may differ from those in non-stressed outpatient settings, since the stress of acute illness is associated with the activation of the HPA axis. 5 Such stresses, on the other hand, are unlikely to match those observed in critical illness where a more complex disturbance of adrenal function is seen. 5 Here we have analysed SSTs performed in medical inpatients with acute non-critical illnesses. Our objectives were to define basal cortisol thresholds that accurately predict the outcome of the SST and which could thus prevent unnecessary SSTs.
Materials and methods
We audited SSTs performed in acute medical admissions in our institution between January 2008 and July 2009 (n = 166, median age 66, range 15–94 y; men 48%, women 52%). All patients were managed on general medical or acute admissions wards and the decision to undertake SSTs and the timing of tests was at the discretion of individual admitting physicians. Forty-two per cent of SSTs were performed between 9:00 and 12:00, 37% between 12:00 and 15:00, 19% between 15:00 and 18:00 and 2% of tests between 18:00 and 21:00. The most common indications for SSTs were hyponatraemia (21%), hypotension (18%), lethargy (15%), gastrointestinal symptoms (11%), weight loss (10%) and long-term glucocorticoid use (9%). The indications for SSTs were unclear in 17% of requests. The admitting diagnoses included pneumonia (21%), chronic obstructive pulmonary disease (9%), urinary tract infection (14%), congestive cardiac failure or acute coronary syndromes (12%), gastroenteritis (7%), seizures (2%), metastatic malignancy (4%), cerebrovascular accidents (2%), hyperglycaemic states (7%), renal failure (2%), severe hypothyroidism (1%) and non-specific presentations (19%).
Serum cortisol was measured before (basal) and 30 min after intravenous injection of 250 μg of synacthen (Nycomed Austria GmbH, Linz, Austria). Cortisol assays were performed by an electrochemiluminescence assay and analysed on the Roche E170 analyser (Roche Diagnostics GmbH, Mannheim, Germany). The intra-assay coefficient of variation (CV) was 1.1% and 1.0% at 418 and 866 nmol/L, respectively. The interassay CV was 4.6%, 3.9% and 8.6% at 532, 896 and 64 nmol/L, respectively.
The SST was interpreted according to the 30-min cortisol level as ‘pass’ (>550 nmol/L) or ‘fail’ (≤550 nmol/L). The incremental cortisol value (30–0 min) was calculated as the difference between the 30- and 0-min cortisol concentrations. Receiver operating characteristics (ROC) curves were generated for the predictive value of the basal cortisol and incremental values for a failed SST. Correlations between the basal, incremental and 30-min cortisol concentrations were determined using the Pearson's correlation coefficient. The Kolmogorov–Smirnov–Z test was used to compare median cortisol concentrations in men and women. All analyses were performed using SPSS for windows, version 16 (SPSS Inc, Chicago, IL, USA). P < 0.05 was considered statistically significant.
Results
Of the 166 SSTs, a pass was seen in 127 (76.5%) tests, while 39 (23.5%) tests failed the SST. Median cortisol concentrations in patients who passed the SST was not significantly different in women and men for the 0- (431 versus 455 nmol/L), 30- (809 versus 818 nmol/L) and 30–0 min values (464 versus 374 nmol/L); women versus men, P > 0.05 for all values. In patients who failed the SST median cortisol concentrations was also not significantly different in women and men for the 0- (127 versus 124 nmol/L), 30- (445 versus 379 nmol/L) and 30–0 min values (265 versus 232 nmol/L); women versus men, P > 0.05 for all values.
ROC curves showed that no single cut-off point of the baseline cortisol was at the same time adequately sensitive and specific for failing the SST despite a good overall predictive value (area under curve [AUC] 0.94; 95% confidence interval 0.89–0.98). A basal cortisol <420 nmol/L had 100% (maximum) sensitivity and 54% specificity for failing the SST, while a basal cortisol <142 nmol/L had 100% (maximum) specificity and 35% sensitivity. Defining a fail as 30-min ≤600 nmol/L produced similar thresholds as the 550 nmol/L cut off. Figure 1 shows excellent correlation between basal and 30-min cortisol levels (r = 0.84; P < 0.0001).
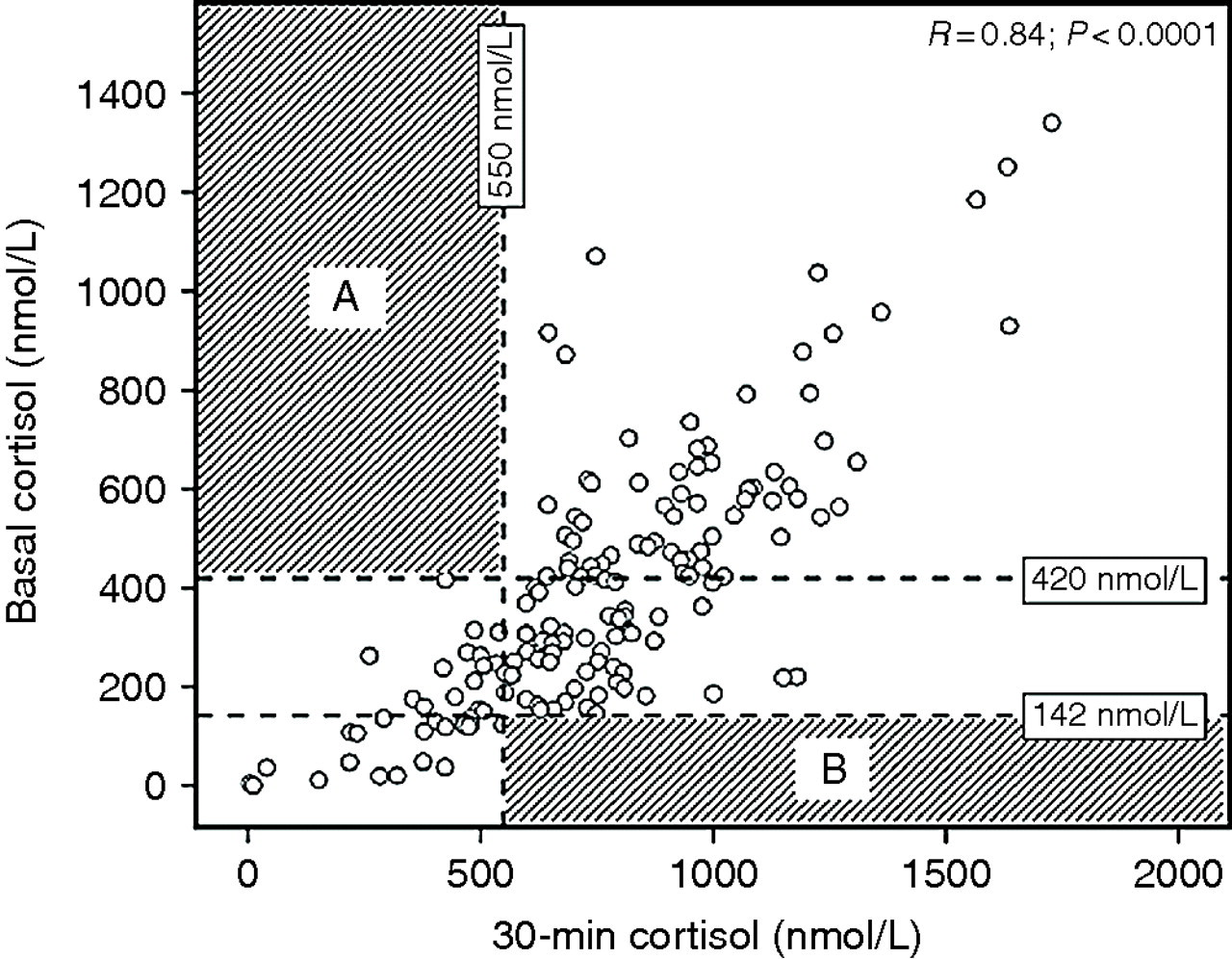
Relationship between basal and 30-min peak serum cortisol in acutely ill patients with suspected adrenocortical insufficiency. The horizontal dashed lines represent the basal cortisol thresholds with maximum sensitivity (420 nmol/L) and maximum specificity (142 nmol/L) for failing the SST. The vertical dashed line represents the 30-min peak cortisol cut-off point (550 nmol/L) used to define a ‘pass’ or ‘fail’ in the SST. The shaded quadrant A shows that no patient with a basal cortisol >420 nmol/L failed the SST, while the shaded quadrant B shows that no patient with a basal cortisol <142 nmol/L passed the SST. SST, short synacthen test
ROC curve for the incremental cortisol was inferior to the basal cortisol in predicting the SST (AUC 0.82; 95% confidence interval 0.75–0.88). Maximum sensitivity (100%) was seen at an incremental value of 426 nmol/L with corresponding specificity of 54% at this cut off. Specificity was poor and 92% specificity was only achievable at the cost of extremely poor sensitivity (19%) at an incremental cut off of 0 nmol/L. The correlation between the incremental and 30-min cortisol concentration was weak (r = 0.55; P < 0.001) and no correlation was observed between the incremental and basal cortisol concentrations (r = 0.04; P > 0.05).
Conclusion
Here we show that performing a random or baseline serum cortisol may preclude the need for SSTs in a proportion of acute medical admissions. No patient with a basal cortisol >420 nmol/L failed the SST, while a basal cortisol <142 nmol/L guaranteed a fail. Restricting the SST to patients with baseline cortisol <420 nmol/L would have safely avoided 44% of SSTs. These thresholds differ from those obtained in standard outpatient conditions. Le Roux et al. 3 showed that an outpatient 9:00 cortisol <100 nmol/L had maximum specificity for failing the SST, while optimal sensitivity was seen at <500 nmol/L. The higher maximum specificity threshold in our sample may be due to increased endogenous HPA axis activation in acute illness. 5 However, our 420 nmol/L cut-off point is close to the basal serum cortisol threshold of 450 nmol/L validated against the ITT as a normal adrenal response. 4
We acknowledge the limitations in defining an adequate adrenal response in acute illness. The SST occasionally gives false-negatives when set against the ITT. 4 Furthermore, the widely adopted normal peak serum cortisol response of >550 nmol/L has largely been validated in standardized settings only. 1 Its application in acute illness remains debatable and some have suggested that a 600 nmol/L limit may be more appropriate in acute medical admissions. 6 Nonetheless, the basal serum cortisol performed similarly against both peak points in our sample. One important caveat in the interpretation of the SST is that total serum cortisol levels may be affected by concentrations of cortisol binding globulins (CBG). 7 Hence, reduced CBG concentrations in critical illness may give false-positive results, whereas synthetic oestrogens may increase CBG concentrations and cause elevations in total cortisol concentrations. 1 The influence of these factors on our findings is unclear since free cortisol concentrations were unavailable. The performance of our thresholds against free cortisol indices will, however, be of interest and deserves further study.
We note that our protocol for the SST may differ in respects from practice elsewhere. Some have proposed the use of a low-dose SST (1 μg) as a more sensitive test of adrenal insufficiency in acute illness. However, the superiority of the low-dose test over the conventional SST is unproven and the low-dose test is currently not recommended in routine practice. 1 Incremental cortisol values have also been used in critical care settings to define adrenal insufficiency. 1 In our study, we found no correlation between the incremental value and the basal cortisol concentration. Furthermore, the incremental value was a poor predictor of the SST.
To conclude, a baseline cortisol >420 nmol/L guaranteed a pass in the SST and was sufficient to exclude adrenal insufficiency, although no single threshold of the baseline cortisol was at the same time adequately sensitive and specific to be reliable as a diagnostic test of adrenocortical insufficiency. Thus, a random serum cortisol may be useful as a rapid initial screening test of adrenocortical insufficiency in acute non-critically ill patients. However, the SST will still be indicated in patients with random serum cortisol <420 nmol/L or where the clinical suspicion of adrenal insufficiency is compelling. Lastly, it may be prudent to proceed to the SST rather than delay the diagnostic process in situations where timely processing of basal cortisol assays is impractical.
DECLARATIONS
