Abstract
Introduction
The purpose of this study was to evaluate the utility of imaging examinations in patients with elevated tumour markers when (a) the tumour marker is not validated for as a primary diagnostic test; (b) the patient had no personal history of cancer and (c) the patient had no other imaging indication.
Materials and methods
Patients without known cancer who had abnormal carcinoembryonic antigen, CA19-9, CA125 and/or CA15-3 serology over a one-year period were included. A retrospective medical record review was performed to assess the number of these cases who underwent imaging because of ‘elevated tumour marker’ in the absence of a clinical indication for imaging. The number and result of these imaging studies were evaluated.
Results
Eight hundred and nineteen patients were included. Of those, 25 patients (mean age: 67.8 [range 41–91] y), were imaged to evaluate: ‘elevated tumour marker’. They underwent 29 imaging studies (mean [±standard deviation (SD)] per patient = 1.2 [±0.4]), and had 42 elevated tumour marker serology tests (mean [±SD] per patient = 1.7 [±0.7]). Four patients had >1 imaging test. No patient had an imaging study which diagnosed a malignancy or explained the elevated tumour marker.
Conclusion
The non-judicious use of tumour markers can prompt further unnecessary investigations including imaging. In this study, there was no positive diagnostic yield for imaging performed for investigation of ‘elevated tumour marker’. ‘Elevated tumour marker’, in the absence of a known underlying malignancy, should not be considered an independent indication for imaging.
Introduction
Biochemical tumour marker serology is an important tool in the management of patients with cancer, allowing effective monitoring of response to therapy, detection of subclinical recurrent disease and in a limited number of specific situations providing an effective screening and diagnostic test. 1 As with any advanced diagnostic test, however, non-judicious use of these tests can lead to more questions than answers, potentially wasting resources and leading to further poorly indicated tests. While useful in the management of patients with known tumours, many tumour marker serology tests are not suitable for use as primary diagnostic tests in individual patients without known tumours, because of limited sensitivity and specificity in this setting. Tumour markers such as carcinoembryonic antigen (CEA), CA19-9, CA125 and CA15-3 are recommended as tools for evaluating response to therapy and post-therapy surveillance, but are not recommended in isolation as screening or diagnostic tests. 2–5 It is our experience that many clinicians order serum tumour markers (often multiple) in patients with non-specific or unrelated complaints in an effort to diagnose an occult malignancy. This pattern of investigation is also well documented in the literature. 5–8 In many cases, the tumour marker serology is ordered outside of institutional guidelines, 9 and there is a significant cost associated with these inappropriate laboratory tests. 10 Having inappropriately ordered the serum tumour markers, any resulting elevated serum tumour marker level will sometimes prompt clinicians to order further investigations to evaluate for a cause of this abnormal result. This pathway is pursued despite the lack of relevant patient symptoms and frequently, at our institution, the next investigation ordered is an imaging test.
The purpose of this study was to evaluate the utility of performing imaging examinations in patients with elevated tumour marker serology when: (a) the tumour marker in question is not validated as a primary diagnostic test; (b) the patient has no personal history of cancer and (c) the patient has no other clinical indication for the imaging study.
Materials and methods
The study was performed at St James' University Hospital, Dublin, a large university-affiliated teaching hospital, and was compliant with the Health Insurance Portability and Accountability Act. According to local policy, as the study was a retrospective medical record review, it was exempt from review by the local institutional review board.
Patient selection
A retrospective review of the hospital biochemistry database was carried out to include all patients who underwent testing with CEA, CA19-9, CA125 and CA15-3 serology over a one-year period, from 1 January to 31 December 2006.
Patients were included if all of the following applied:
The result of one or more of these tests (CEA, CA19-9, CA125 or CA15-3) was above the reference interval; The physician who ordered the tumour marker serology was not a clinical oncologist (as the vast majority of those patients would likely have had known tumours).
The picture archiving and communication system database was searched for imaging studies performed for each included patient. We searched for imaging studies where the stated imaging indication on the radiology requisition from the referring physician was entered as ‘elevated tumour marker’. The imaging requisition was also assessed for other clinical indications for the imaging study. For each patient who fulfilled the above criteria, a review of their medical notes was carried out.
Patients were excluded if:
They had a prior diagnosis of cancer; The patient had another clinical indication for the imaging study in addition to tumour marker elevation.
Imaging studies
The type and number of imaging examinations performed on each patient was recorded. The mean (±standard deviation [SD]) number of imaging examinations per patient was calculated. For each patient, the imaging test result was recorded and categorized as follows:
Diagnosis of cancer associated with elevation of the tumour marker in question; Diagnosis of alternative pathology which may explain the elevated tumour marker result; Diagnosis of incidental pathology without a documented association with the elevated tumour marker; Normal imaging study.
Tumour markers
CEA, CA19-9, CA125 and CA15-3 serology tests were all performed at the on-site clinical biochemistry laboratory. Each analysis was undertaken on the Elecsys 2010 (Roche Diagnostics Gmbh, Mannheim, Germany) in accordance with the manufacturer's protocols and the manufacturer's reference intervals were in operation. For each patient who underwent imaging for evaluation of elevated tumour markers, the number of different tumour marker tests performed was recorded. The mean (±SD) serum level of abnormal tumour marker in this group was calculated.
Statistical analysis
All data were recorded on an Excel spreadsheet (Microsoft Office Excel 2007, Microsoft Corporation) and sums, means and SDs of the mean were calculated.
Results
Patients
Figure 1 illustrates the inclusion and exclusion process and the derived study cohort. Eight hundred and nineteen included patients, referred by non-oncologists, had 1442 elevated tumour marker serology results during the one-year period of the study at our institution. Of those, 35 patients subsequently had imaging studies for ‘elevated tumour marker’, with no other indication stated on the radiology requisition for the imaging study. The case-notes of eight patients were unavailable for review. These patients were excluded from analysis. One patient was excluded as she had a personal history of breast cancer (T2N1M0), resected six years prior to the study period. One patient had a clinical indication for the imaging study performed, not stated on the imaging requisition, but evident from the clinical notes. A total of 25 patients met all inclusion and exclusion criteria. The mean age of included subjects was 67.8 (range 41–91), and the study cohort consisted of 12 male and 13 female patients.
Flowchart illustrates cohort of included patients for analysis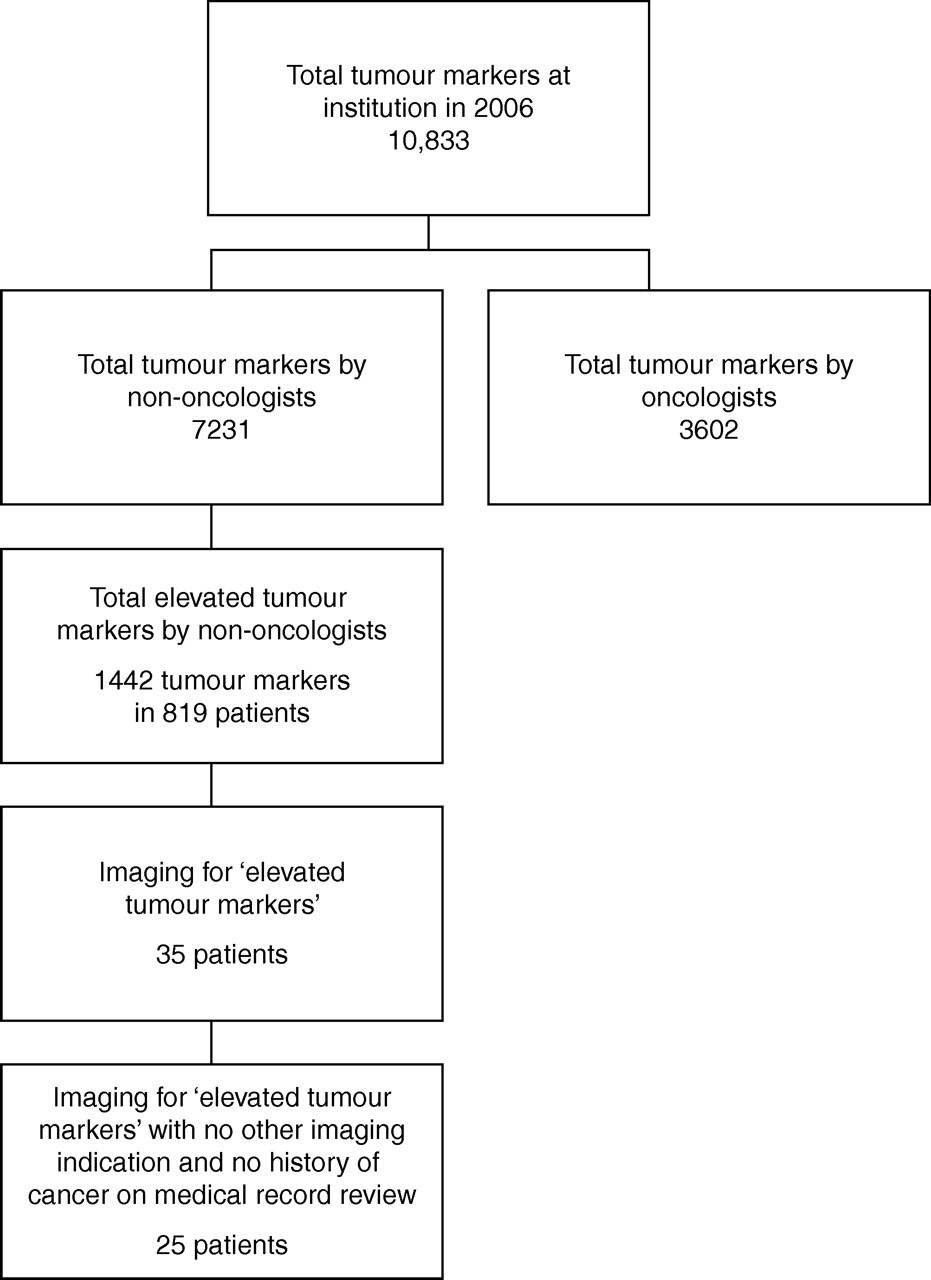
Tumour markers
The 25 patients who underwent imaging had a total of 42 elevated tumour marker serology tests. The mean (±SD) number of elevated tumour marker serology tests per patient was 1.7 (±0.7). Seventeen patients had elevated CEA, 15 patients had elevated CA19-9, seven patients had elevated CA125 and three patients had elevated CA15-3. The mean (±SD) CEA, CA19-9, CA125 and CA15-3 were 7.8 (±3.9) μg/L, 213.8 (±458.0) kU/L, 92.9 (±74.0) kU/L and 32.2 (±2.4) kU/L, respectively. The reference ranges were as follows: CEA: <3.4 μg/L; CA19-9: <27 kU/L; CA125: <35 kU/L; and CA15-3: <25 kU/L.
Imaging studies
The 25 included patients underwent a total of 29 imaging studies (mean [±SD] number of studies per patient = 1.2 [±0.4]). Twelve patients underwent ultrasound (11 abdominal, one pelvic study). Thirteen patients had CT abdomen and pelvis and one patient had CT thorax performed. Two magnetic resonance imaging (MRI) tests were performed (one magnetic resonance cholangiopancreatography, one MRI abdomen). Two mammograms were carried out. Four patients had more than one imaging test performed.
No patient had an imaging study which confirmed the diagnosis of a malignant neoplasm. No patient had an imaging study which explained the reason for the tumour marker result.
Discussion
Tumour markers are minimally invasive tests which can provide important perspectives of malignant biological processes. The optimum use of these diagnostic tests in the clinical setting is the subject of ongoing research and practice guideline development by groups including the American Society of Clinical Oncology, 11 the National Academy of Clinical Biochemistry 12 and the European Group on Tumour Markers. 13 It is widely acknowledged that the primary role of serum tumour markers is in evaluating the response of patients to therapeutic regimens and in the monitoring for disease relapse in patients with treated cancer. To date their use in screening for and the early diagnosis of malignancy is very limited.
Practice guidelines exist for each tumour marker based on validation to date. CEA is not recommended as a screening test for colorectal cancer. 4 In patients with known colorectal cancer, however, CEA is recommended for use in staging/treatment planning, postoperative surveillance of patients with Stage II or III disease, and in monitoring response to systemic chemotherapy in patients with metastatic disease. 3,4,14 Similarly, CA19-9 is not recommended for screening for pancreatic cancer. 4,15 For patients with pancreatic cancer, CA19-9 can be used in the monitoring of patients with locally advanced or metastatic disease receiving active therapy. 4 CA15-3 is not recommended as a screening test for breast cancer, 2 but may be used for therapy monitoring in advanced disease. 14 CA125 has a high false-positive rate and should not be used in isolation as a primary diagnostic or screening test. 5 CA125 can, however, be used in the investigation of postmenopausal women with suspicious pelvic masses. 14,15 CA125 is also recommended for monitoring response to therapy and detecting recurrence in patients with known ovarian cancer. 14
In spite of the fact that the tumour markers listed above are not recommended for use in screening, considerable inappropriate ordering of these tests persists. 5,6,8,16 Tumour markers are often inappropriately ordered in the elderly population. The mean age of the group in the current study who underwent imaging for abnormal tumour markers was 67.8 y. In this group, false-positives are particularly likely to occur; Lopez et al. 17 found that 40% of randomly selected patients aged between 66 and 99 years had elevation of at least one tumour marker. There are many reasons for false-positive tumour marker serology. As an example, CA19-9 can be elevated because of inflammatory hepatobiliary disease, thyroid disease and benign (as well as malignant) biliary obstruction. 4
There are several reasons why tumour markers can be inappropriate in screening. First, it is not an effective diagnostic strategy. In a study by Arioli et al. 6 only 5% of tumour markers (CEA, CA19-9, CA15-3 or CA125) ordered at a department of internal medicine in a six-month period were deemed ‘appropriate’ on the basis of international literature. In only 1/201 (0.5%) of cases where the tumour marker was ordered inappropriately was a diagnosis of cancer made. Second, the finding of an abnormal tumour marker result and the investigations that subsequently ensue can cause substantial patient anxiety and clinical uncertainty. 5 Third, this approach causes unnecessary additional cost. In a study by Petignat et al. 16 the estimated annual cost of inappropriate CA125 tests in Switzerland in 2000 is 223,399 euro/per year. Finally, as many of the investigations involve CT, an unnecessary radiation dose is incurred.
Tumour markers should be used to contribute to practice decisions in such a way as to improve clinical outcome including disease-free survival, quality of life and to optimize the cost of a patient's health care. 18 The inappropriate use of these tests, however, can only serve to increase health-care costs not only by virtue of the tumour marker itself but also because of additional tests ordered to search for the tumour which might have caused the abnormal blood test. The current study demonstrates that clinicians often order imaging tests in this manner and that this is a fruitless and costly exercise. The total cost of imaging studies performed at our institution in one year to evaluate the cause of abnormal tumour markers was 6815 euros. The total cost of these unnecessary imaging tests in a North American setting based on estimated national average cost per study 19 would have been in the region of $40,280.
In conclusion, the current study reinforces the importance of the judicious use of tumour marker serology in the investigation and management of patients. The use of many tumour markers as primary diagnostic tests in patients is inappropriate and this practice can also prompt further unnecessary investigations including diagnostic imaging tests. In our study, there was no positive diagnostic yield of imaging tests performed solely for the investigation of ‘elevated tumour markers’. ‘Elevated tumour marker’ (where the tumour marker in question is not validated as a primary diagnostic test) should not be considered an independent indication for imaging in the absence of additional clinical symptoms.
DECLARATIONS
