Abstract
Double gap metabolic acidosis occurs in the setting of unmeasured active osmoles in the serum (osmolal gap) and anion gap (AG) metabolic acidosis. We describe a 67-year-old woman with acute respiratory failure on mechanical ventilator from pneumonia and anuric acute on chronic renal failure (urea nitrogen 21.4 mmol/L, creatinine 530.4 μmol/L) requiring haemodialysis (HD). On hospital day 5, she was found to have progressive metabolic acidosis (serum pH 7.16, PCO2 4.38 kPa, HCO3
− 12.1 mmol/L and AG 21 mmol/L). There was no evidence of hypoxaemia, hypoperfusion or haemodynamic instability. Normal serum ketone and
Introduction
A high anion gap (AG) and osmolal gap (double gap) metabolic acidosis is an important clue of toxic alcohol intoxication, such as methanol and ethylene glycol. Propylene glycol (PG, 1,2-propanediol), a less-recognized alcohol, is believed to be innocuous and has been widely used as a solvent and preservative in cosmetics, foods and pharmaceuticals, including intravenous lorazepam. Although lorazepam is the drug of choice for maintenance sedation in intensive care units (ICU) due to its appreciated safety, high-dose continuous infusion (>0.1 mg/kg/h) have been implicated in the development of PG accumulation and toxicity. 1–4
PG is synthetically prepared from propylene and glycerol as a racemic mixture of
Case report
A 67-year-old woman presented to the emergency department with shortness of breath and fever for one day. She had poorly controlled hypertension for 10 y and a two-year history of stage 4 chronic renal insufficiency. On admission, her blood pressure was 118/76 mmHg, heart rate 102 beats/min, respiratory rate 24/min, body temperature 38.2°C and body weight 50 kg. Physical examination showed jugular venous distention, use of accessory muscles for respiration, diffuse inspiratory crackles and grade II pitting oedema. The remainder of the physical examination was unremarkable. Laboratory studies showed a white cell count of 14.0 × 109/L, haemoglobin 95 g/L and platelets 146 × 109/L. Serum biochemistries demonstrated acute renal failure (urea nitrogen 21.4 mmol/L, creatinine 530.4 μmol/L) and normal liver function (Table 1). Her chest X-ray showed bilateral pneumonia in the lower lobes and abdominal ultrasonography showed bilateral atrophic kidneys. The patient was intubated for acute respiratory failure and lorazepam continuous infusion uptitrated to 10 mg/h to achieve adequate sedation for synchronized ventilation. Ticarcillin/clavulanate 2.0 g every 12 h intravenously was begun for treatment of pneumonia. She also underwent haemodialysis (HD) on days 2 and 3 of hospitalization for acute renal failure.
Serial serum laboratory investigations
BUN, blood urea nitrogen
*Osmolal gap = measured plasma osmolality − calculated osmolality = measured osmolality − (2 × [Na+] + [BUN]/2.8 + [glucose]/18) 6
By the fifth hospital day, her pneumonia improved markedly, but high AG metabolic acidosis (pH 7.16, PO2 14.9 kPa, PCO2 4.4 kPa, HCO3
− 12.1 mmol/L, AG 21 mmol/L) (Table 1) was noticed without hypoxaemia, tissue ischaemia or severe sepsis. A work-up for the excessive anions including
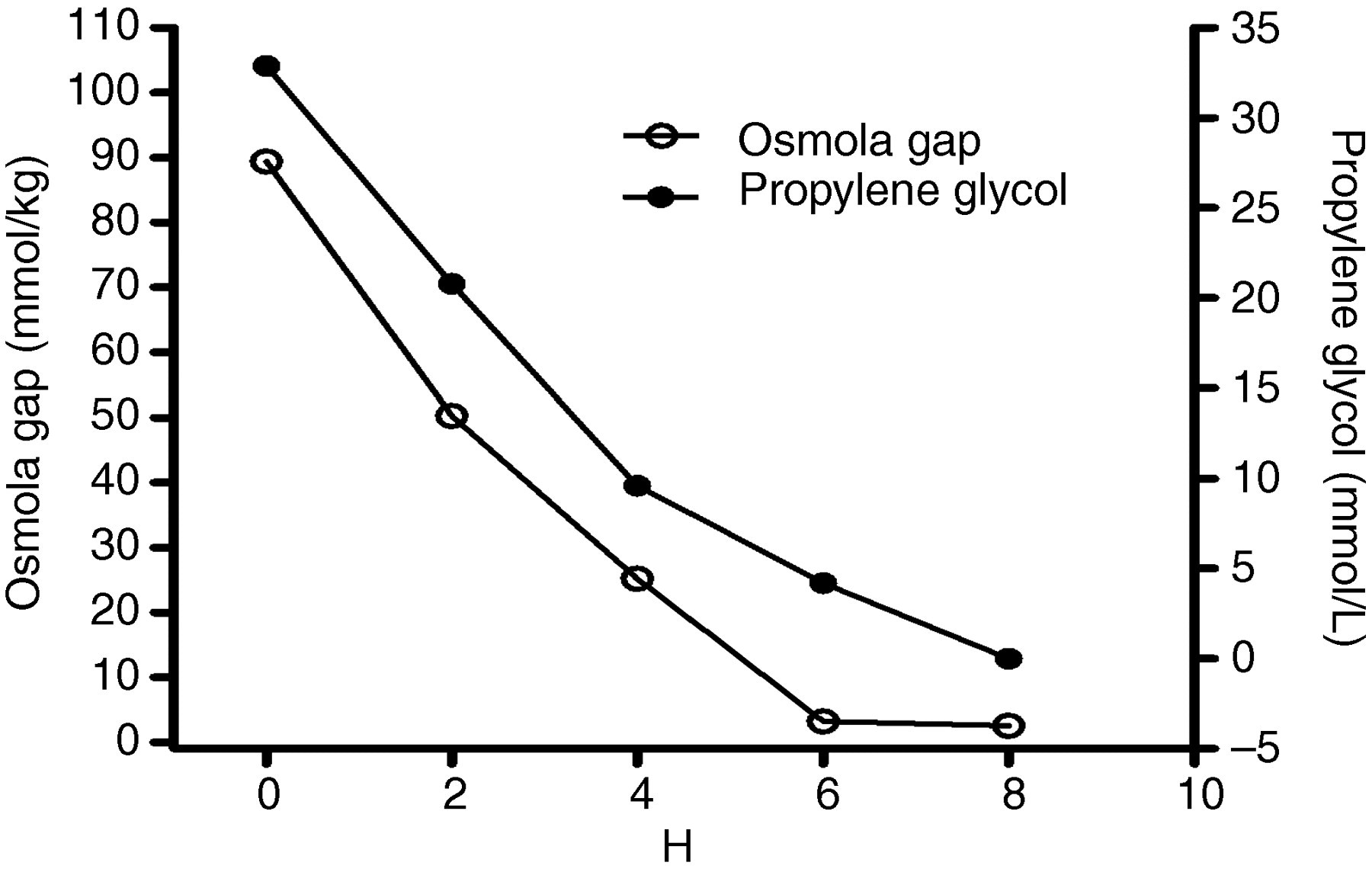
Efficacy on removal of propylene glycol by long-extended haemodialysis
Discussion
This patient developed double gap metabolic acidosis following the continuous administration of high-dose PG-containing lorazepam (0.2 mg/kg/h, containing PG 54.5 mmol/h) for five days. PG overdose was considered after the exclusion of ethanol, methanol and ethylene glycol intoxication. The total accumulated dose of PG was estimated to be at least 2616 mmol (54.5 mmol × 24 h × 2 d) within the two days from the last HD to the measurement of PG because HD presumably is effective in the clearance of PG.
7
Her serum PG of 32.9 mmol/L represented an osmolality of approximately 33 mmol/kg, which did not fully account for the osmolal gap 89.4 mmol/kg. In addition to PG, other solvents and unmeasured intermediate metabolites such as methylglyoxal or lactaldehyde may in part contribute to her osmolal gap.
8
Extended HD for eight hours effectively eliminated the serum PG and osmolal gap. However, another hidden cause was responsible for the worsening AG metabolic acidosis after dialysis because her serum
Intravenous lorazepam contains lorazepam at a concentration of 2 mg/mL suspended in PG (80%, molecular weight [MW] 76 Da), polyethylene glycol-400 (PEG-400, 18%, MW 400 Da) and benzyl alcohol (2%, MW 108 Da). PG contributes 23 and 57 times as many osmotically active particles as PEG-400 and benzyl alcohol and exerts toxicity at a concentration exceeding 2.36 mmol/L.
9
In general, a maximal PG dose of 1 g/kg/d (13.2 mmol/kg/d) is presumed to be safe in the absence of risk factors. The half-life of PG is 1.4–3.3 h in normal individuals. The elimination kinetics of PG is a saturable capacity-limited metabolism, indicating first-order elimination for low-to-moderate plasma concentration but zero-order for high concentration.
10
Approximately 12–45% of PG is excreted unchanged in urine. The rest is oxidized to
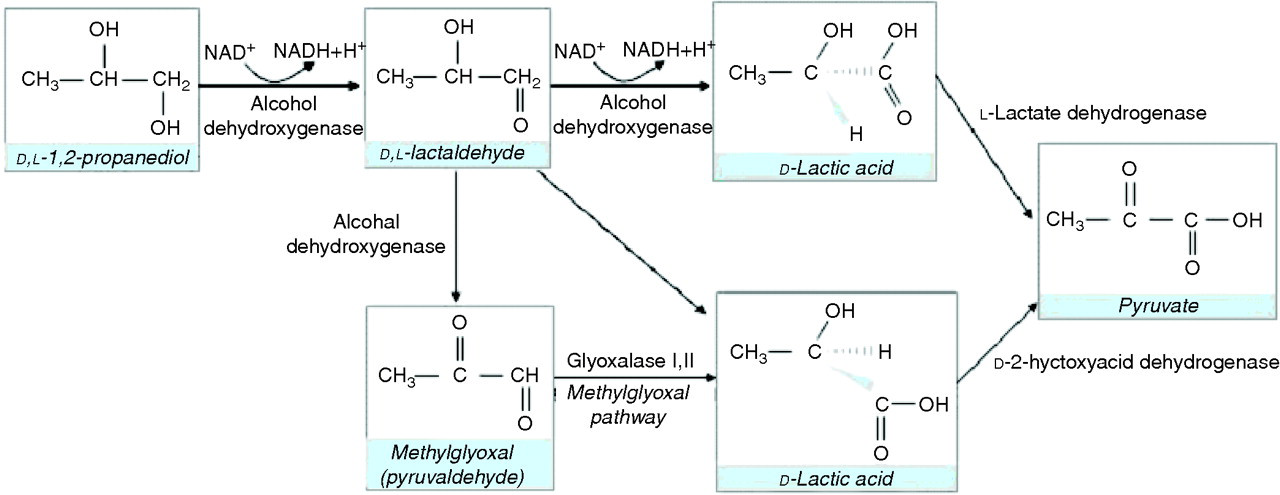
Metabolism of propylene glycol (propanediol)
PG toxicity has been hypothesized to result from direct cytotoxicity of the parent compound and its metabolites. A significant correlation between cumulative lorazepam dose and end-of-therapy PG concentration has been demonstrated in paediatric ICU and in adult patients on high-dose lorazepam infusion for more than 48 h. 11,12 Serum PG concentrations showed considerable interpatient variation with respect to the rate of infusion and accumulated dose in adults (Table 2). 9,13–22 We analysed the prior cases in the literature and found no significant correlation between the serum PG concentration and daily dose of PG, serum lactate concentration or AG. As seen in Figure 1, the serum osmolal gap trended together with PG concentration during HD and may be a sensitive and convenient index to assess the degree of PG intoxication.
Literature review of intravenous lorazepam-induced propylene glycol intoxication
CVVHD, continuous veno-venous hemodialysis; HD, hemodialysis; NS,not shown; PG, propylene glycol
The metabolic acidosis with normal serum
Other unmeasured anionic metabolites, like pyruvate, α-ketoglutarate, sulphate and phosphate, may contribute to the high AG metabolic acidosis in this patient. Because this patient was dialysed with conventional alkali dialysate, endogenous acid production from protein oxidation during the interdialytic period rather than intradialytic factors was a potentially important contributor for her AG metabolic acidosis. 24 Protein hypercatabolism in the face of severe critical illness may be a significant source of increased endogenous acid production in this patient who had no ability to excrete acid renally.
In conclusion, PG is an important cause of high osmolal gap but not necessarily high AG metabolic acidosis. The serum osmolal gap is a reliable surrogate for monitoring and assessing retained PG. Whether
DECLARATIONS
