Abstract
Background
Individuals who are unable to metabolize the short-acting muscle relaxant succinylcholine due to abnormal cholinesterase activity are currently investigated via spectrophotometry using artificial substrates and enzyme inhibitors. Methods have been described using succinylcholine as substrate but with measurement of the product choline. However, choline may be released from other endogenous substrates within the serum. Direct measurement of the in vitro metabolism of succinylcholine as substrate may provide a better indication of the in vivo situation with regard to cholinesterase status.
Methods
The rate of in vitro metabolism of succinylcholine by cholinesterase was measured using liquid chromatography linked to tandem mass spectrometry (LC-MS/MS). A comparison was made using serum samples in which cholinesterase activity had been measured using propionylthiocholine as substrate and phenotyped by enzyme inhibitor studies.
Results
A good correlation (r = 0.9, P < 0.0001) was found between cholinesterase activity measured by LC-MS/MS using succinylcholine as substrate compared with propionylthiocholine as substrate measured spectrophotometrically. All serum samples with a cholinesterase activity of <1 IU/L, as measured using succinylcholine as substrate, were considered to be at increased risk of succinylcholine sensitivity. These latter results correlated well to the atypical phenotypes.
Conclusions
A simple and fast LC-MS/MS technique for the measurement of cholinesterase activity using succinylcholine as substrate has been described. This method clearly identifies patients at risk of prolonged apnoea following succinylcholine administration and compares favourably with existing spectrophotometric methods using artificial substrates.
Introduction
Butyrylcholinesterase (acylcholine acylhydrolase EC 3.1.1.8), also known as serum cholinesterase and occasionally pseudocholinesterase, is clinically important for the hydrolysis of choline esters, such as the short-acting muscle relaxants succinylcholine and mivacurium. Succinylcholine is a depolarizing neuromuscular blocking agent favoured for both its fast onset of action and short duration. It is often used in emergency medicine and anaesthesia to induce muscle relaxation and to enable endotracheal intubation. However, shortly after its introduction some individuals were found to be unable to metabolize succinylcholine efficiently, resulting in a prolonged ‘scoline apnoea’ requiring artificial respiration for an extended period. 1 The most clinically significant causes are due to one or more inherited abnormal alleles that reduce cholinesterase activity either by reducing the amount of enzyme present or by reducing its ability to effectively hydrolyse succinylcholine. 2 Kalow and Gunn, 3 using the cholinesterase inhibitor dibucaine, subsequently found that the enzyme of affected individuals resisted inhibition. By using the percentage inhibition (of normal activity), three distinct phenotypes were ascribed to the enzyme, the ‘Usual’ (UU) and ‘atypical’ (AA) homozygotes and the intermediate (UA) heterozygotes. Fluoride-resistant (F) 4 forms of the enzyme were also found as were the additional variants, (J) 5 and (K) 6 that are only identifiable when they occur as heterozygotes with other forms. Unfortunately, there is no recommended reference method for the measurement of cholinesterase activity. Historically, a variety of methods employing different artificial substrates, e.g. propionylthiocholine, 7 butyrylthiocholine, 8 benzoylcholine 9 or acetylcholine at different temperatures have been used. 2 Enzyme inhibition studies using dibucaine, fluoride and Ro-02 0683 enable biochemical phenotyping.
The use of artificial substrates has also been criticized with regard to the poor correlation between enzyme activity and an individual's clinical response to succinylcholine. 10 Variants exist that can metabolize artificial substrates in vitro, and hence are classified as ‘Usual’, but do not metabolize succinylcholine in vivo 11,12 and so are susceptible to scoline apnoea. In these cases, it would be advantageous to use succinylcholine itself as the assay substrate.
The best methods to date using succinylcholine as substrate employ choline oxidase to measure the choline released following metabolism by cholinesterase. 11,13 Several authors using these methods have highlighted patients classified as ‘Usual’ with normal activity by the standard benzoylcholine method, but demonstrating impaired enzyme activity using succinylcholine as substrate. However, attempts to repeat this work has been difficult. 14 Assays for cholinesterase measuring the choline released may be hindered by the fact that endogenous choline present in plasma can increase independently of cholinesterase activity. 15 Problems with these methods therefore point to the benefits of measuring substrate depletion directly.
Recently, a method was developed and validated for the simultaneous determination of succinylcholine and its succinylmonocholine metabolite in serum and urine samples. 16 The liquid chromatography linked to tandem mass spectrometry (LC-MS/MS) method was used for forensic and clinical toxicology purposes in two cases investigated for potential succinylcholine intoxication. We have therefore applied tandem mass spectrometry to develop a simple and fast enzyme method for the direct measurement of succinylcholine hydrolysis by cholinesterase in serum.
Materials and methods
Samples
All serum samples used in this study had been referred to this laboratory for the analysis of cholinesterase activity and for enzyme phenotyping in relation to the investigation of possible scoline apnoea. Samples had been stored at −20°C and the stability of cholinesterase has been demonstrated previously. 17 Samples were grouped (Table 1) according to both their known activities, measured using propionylthiocholine as substrate, 7 and phenotype, determined by inhibition studies using dibucaine, 18 fluoride 5 and Ro-02 0683, 19 and benzoylcholine as substrate.
Patients samples were grouped for comparison with LC-MS/MS according to activity determined using propionylthiocholine and phenotype via inhibitor studies
LC-MS/MS, liquid chromatography linked to tandem mass spectrometry
Reagents
Succinylcholine chloride was supplied by Sigma (Sigma-Aldrich, Dorset, UK) and prepared as 5 mmol/L, 250 μmol/L and 180 μmol/L stock solutions in H2O and stored at −80°C. An eight-point calibration curve was prepared for each assay from the 250 μmol/L stock solution in H2O using serial dilutions to cover the range of 0.39–25 μmol/L. A stock internal standard consisting of deuterated (D8) succinylcholine (CDN Isotopes, Quebec, CA, USA) was prepared in water to 200 μmol/L and stored in 1 mL aliquots at −80°C. A working internal standard solution of 20 μmol/L D8-succinylcholine was prepared daily from the stock solution and discarded after use.
Assay buffer for enzyme studies consisted of phosphate-buffered saline (PBS, 50 mmol/L, pH 7.4). The mobile phase for LC-MS/MS was ammonium acetate 4 mmol/L (BDH, Lutterworth, UK) in water at pH 2.2 adjusted with formic acid (Scientific Laboratory Supplies, Nottingham, UK). Following pH adjustment methanol was added to a concentration of 5% (volume in volume). Methanol and acetonitrile used in this work were high-performance liquid chromatography grade (Sigma-Aldrich, Dorset, UK). SeronormTM control material at two levels of cholinesterase activity was purchased from BioStat Diagnostic Systems (Stockport, UK).
An ion exchange column was prepared in-house for LC-MS/MS using a strongly basic anion exchange resin (BioRad, Hertfordshire, UK) suspended in deionized water and packed into a 50 × 3 mm2 steel column using a column slurry packer (Alltech Associates Applied Science Ltd, Lancashire, UK).
Assay procedure
Kinetic studies were performed to establish the time course of the reaction and determine the K m of cholinesterase for succinylcholine. Reaction time course was studied using a serum sample from a patient demonstrating a cholinesterase activity of 3800 IU/L as measured by the propionylthiocholine method and a ‘Usual’ phenotype. Serum (500 μL) was added to 5 mL of assay buffer and preincubated at 37°C in a water bath. This was followed by the addition of 500 μL of substrate (succinylcholine, 360 μmol/L) and time recorded. At two-minute intervals 200 μL of the incubate was removed and added to 1 mL of cold acetonitrile to stop the reaction. The reaction was monitored over a time period of 14 min.
A zero-minute time point, where no metabolism of succinylcholine had occurred was also prepared using 50 μL of serum, 50 μL of succinylcholine substrate and 500 μL of PBS added directly to 1200 μL cold acetonitrile. All samples were centrifuged at 13,000
The results of this initial study (Figure 1) demonstrated the rapid metabolism of succinlycholine with a t ½ of 2.2 min and total substrate exhaustion by 14 min. Hence, all further experiments were carried out monitoring the time course of the rate of reaction over the first two minutes.
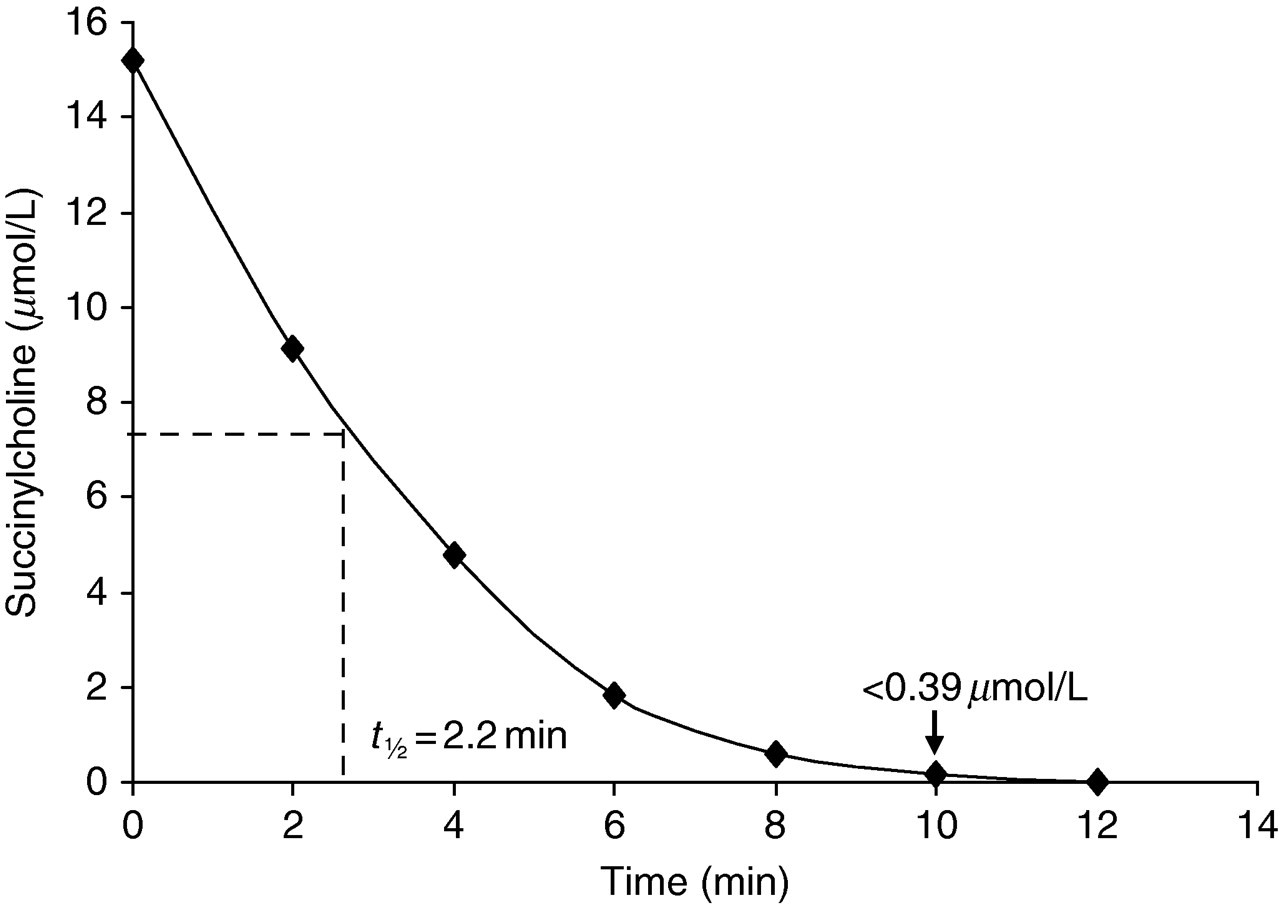
Kinetic studies demonstrating the time course of the metabolism of succinylcholine (15 μmol/L) measured using liquid chromatography linked to tandem mass spectrometry
Reaction kinetics over a range of succinylcholine concentrations (1–500 μmol/L) in the assay mixture were explored using the same serum sample but followed over the 0–2 min time period. Reaction tubes were set up for each succinylcholine concentration (1, 5, 10, 15, 30, 60, 80, 100, 500 μmol/L) containing 500 μL of assay buffer and 50 μL of serum. Reactions were started by the addition of 50 μL of the respective succinylcholine concentration and stopped at two minutes by taking 200 μL of the assay mix and adding it to 400 μL of cold acetonitrile. Zero time points were established for each substrate concentration as described above for the initial kinetic studies. Succinylcholine was measured in each sample as described later. This study demonstrated enzyme inhibition at a concentration of 80 μmol/L succinylcholine (Figure 2a). A Lineweaver–Burke plot was constructed from this assay data (Figure 2b) from which the K m was calculated as 54 μmol/L. This compared favourably with previous reported values of 53 and 37 μmol/L, respectively. 11,10
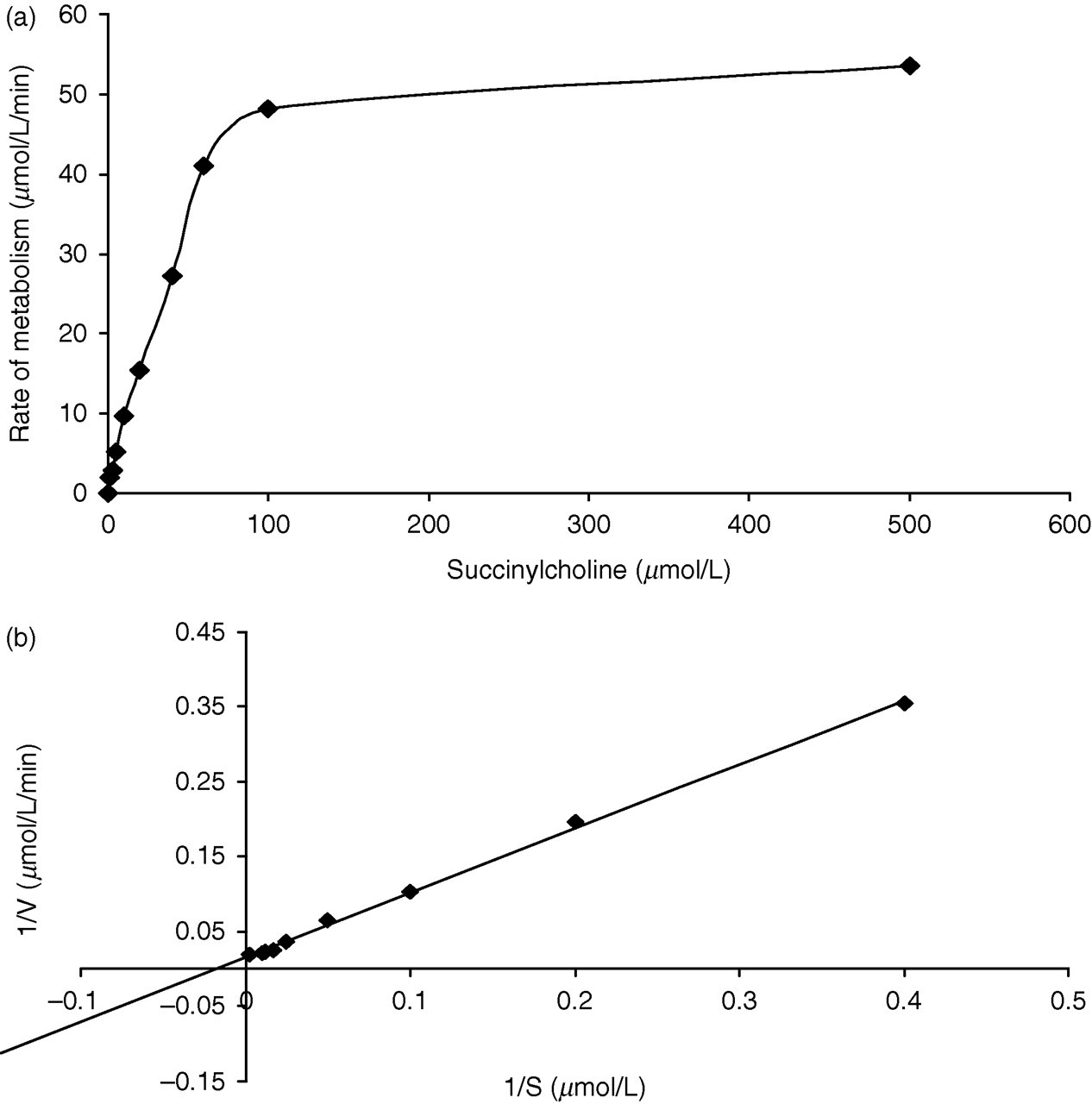
(a) Kinetic studies showing the rate of metabolism of succinylcholine over a range of substrate concentrations (0–500 μmol/L). (b) Lineweaver–Burke analysis of the rate of succinylcholine metabolism at concentrations of 0–500 μmol/L
Samples from different phenotypes, i.e. UA, AK and atypical, were examined using the above kinetic studies resulting in reduced K m values of 42, 16 and 7 μmol/L, respectively.
From the above studies it was decided that a substrate concentration of 15 μmol/L would be the most appropriate to investigate a wide range of samples with varying cholinesterase activity and phenotype. This substrate concentration would not produce any substrate inhibition and would provide adequate sensitivity for the measurement of residual succinylcholine following metabolism over two minutes.
All patients' samples were analysed for cholinesterase activity using the 15 μmol/L concentration of succinylcholine and the method as described above for the investigation of reaction kinetics.
Sample analysis
Liquid chromatography was performed using a Shimadzu system (Milton Keynes, Buckinghamshire, UK) comprising an SIL-HT autosampler, 2x LC10AC pumps, a DGU-14A degasser and a CTO-10AS column oven. The column effluent was directed to the tandem mass spectrometer via a Valco switching valve and the system operation, data acquisition and interpretation was controlled via Analyst 1.4 software (Applied Biosystems, Warrington, Cheshire, UK). The system was configured for direct infusion with the additional use of an ion exchange column to provide better peak area integration. Ten microlitres of sample was injected and a flow rate of 0.6 μL/min was maintained throughout at a temperature of 35°C.
The Applied Biosystems/MDS Sciex API 3000 Triple-Quad Mass Spectrometer equipped with a TurboIonSpray source (Applied Biosystems, Warrington, Cheshire, UK) was used in positive ion mode with an electrospray source and the instrument analysis time per sample was two minutes. The capillary voltage was 2 kV, source temperature 400°C and nitrogen was used for the nebulizer gas, curtain gas and collision gas at flow rates of 10, 10 and 8 L/min, respectively. With a collision energy of 17 eV, the multiple-reaction monitoring transitions used for quantification of succinylcholine and deuterated internal standard were 145.1m/z → 115.4 and 149m/z → 119.6, respectively (Figure 3a). Succinylcholine and internal standard eluted from the column at 0.35 min (Figure 3b and c).
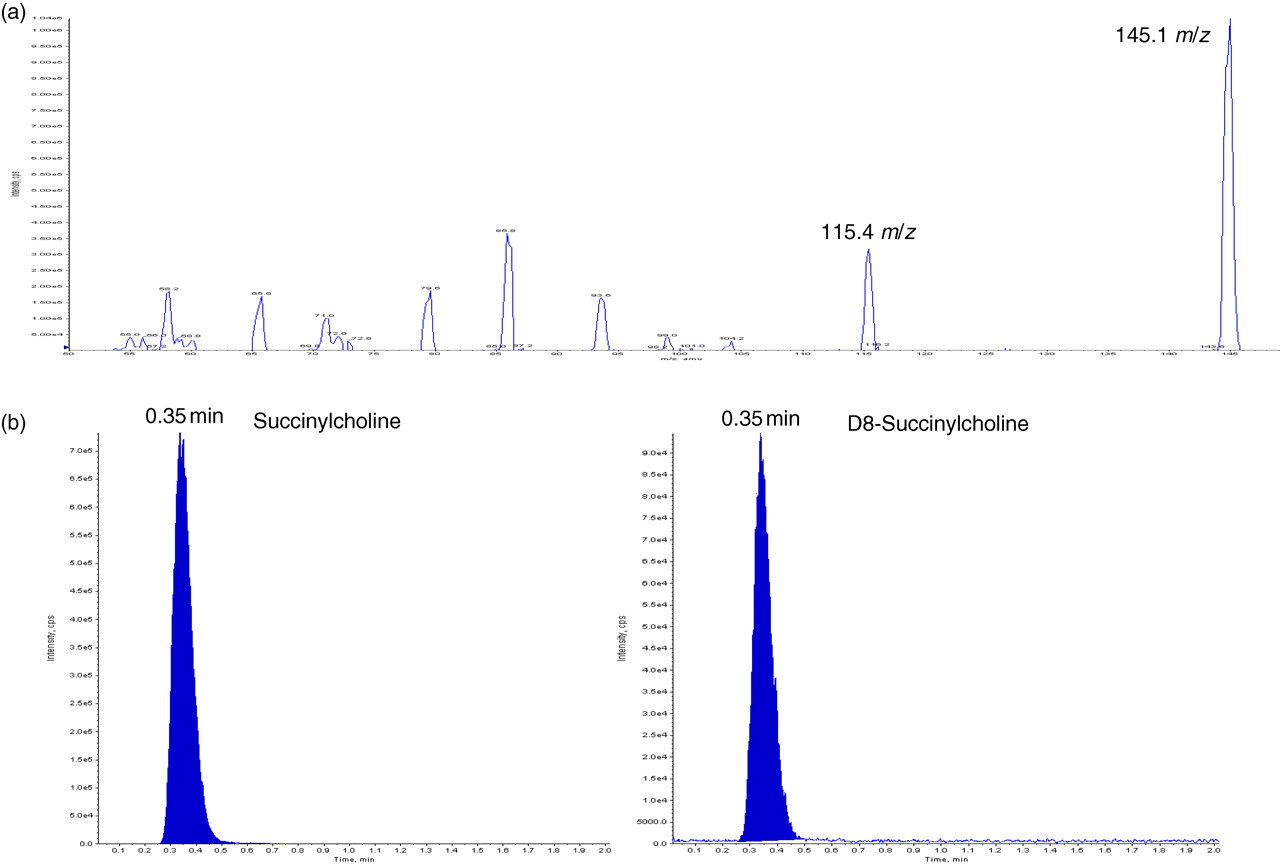
(a) Tuning profile for the multiple-reaction monitoring transitions used for quantification of succinylcholine (290 Da2+) shows the doubly charged parent ion appearing at m/z 145.1, while further fragmentation provides the product ion at m/z 115.4. (b) Example chromatogram showing succinylcholine and deuterated D8-succinylcholine internal standard
Statistical analysis
Data interpretation and statistical analysis used Analyse-IT software version 2.10 (Analyse-IT Software Ltd, Leeds, UK). Pearson's analysis was used to assess the correlation of cholinesterase activities determined by LC-MS/MS compared with propionylthiocholine. One-way analysis of variance was used to determine the significance between mean activities determined using LC-MS/MS compared with activities as determined by propionylthiocholine and phenotype.
Results
Linearity and sensitivity
Linearity for the measurement of succinylcholine was demonstrated up to 12.5 μmol/L (r = 0.998) and there was <10% coefficient of variation for all standards down to the lower standard of 0.39 μmol/L. The signal-to-noise ratio at the lowest standard was found to be >100 and no samples were measured below this value.
Intra- and interassay imprecision
Assay imprecision using two serum samples of Usual and UA phenotype and commercial SeronormTM control material is shown in Table 2.
Intra- and interday imprecision for succinylcholine hydrolysis by serum cholinesterase from two different serum samples of Usual and UA phenotype and SeronormTM control material (interassay) run in parallel with samples measured via LC-MS/MS
LC-MS/MS, liquid chromatography linked to tandem mass spectrometry; SD, standard deviation; CV, coefficient of variation
Method comparison succinylcholine versus propionylthiocholine as substrate
The rate of succinylcholine hydrolysis (μmol/L/min) as measured by LC-MS/MS corresponds to the SI unit of enzyme activity (IU/L) and results were compared with the propionylthiocholine substrate method. The enzyme activity of all samples regardless of phenotype demonstrated a significant correlation (r = 0.9, P < 0.0001) but with a wide degree of scatter (95% confidence interval [CI] 0.84–0.94) as shown in Figure 4. A greater degree of scatter and reduced correlation was observed when comparing results on samples with an activity below the current lower reference limit (<2300 IU/L) for the propionylthiocholine method (Figure 5, r = 0.6, P = 0.001; 95% CI 0.31–0.85).
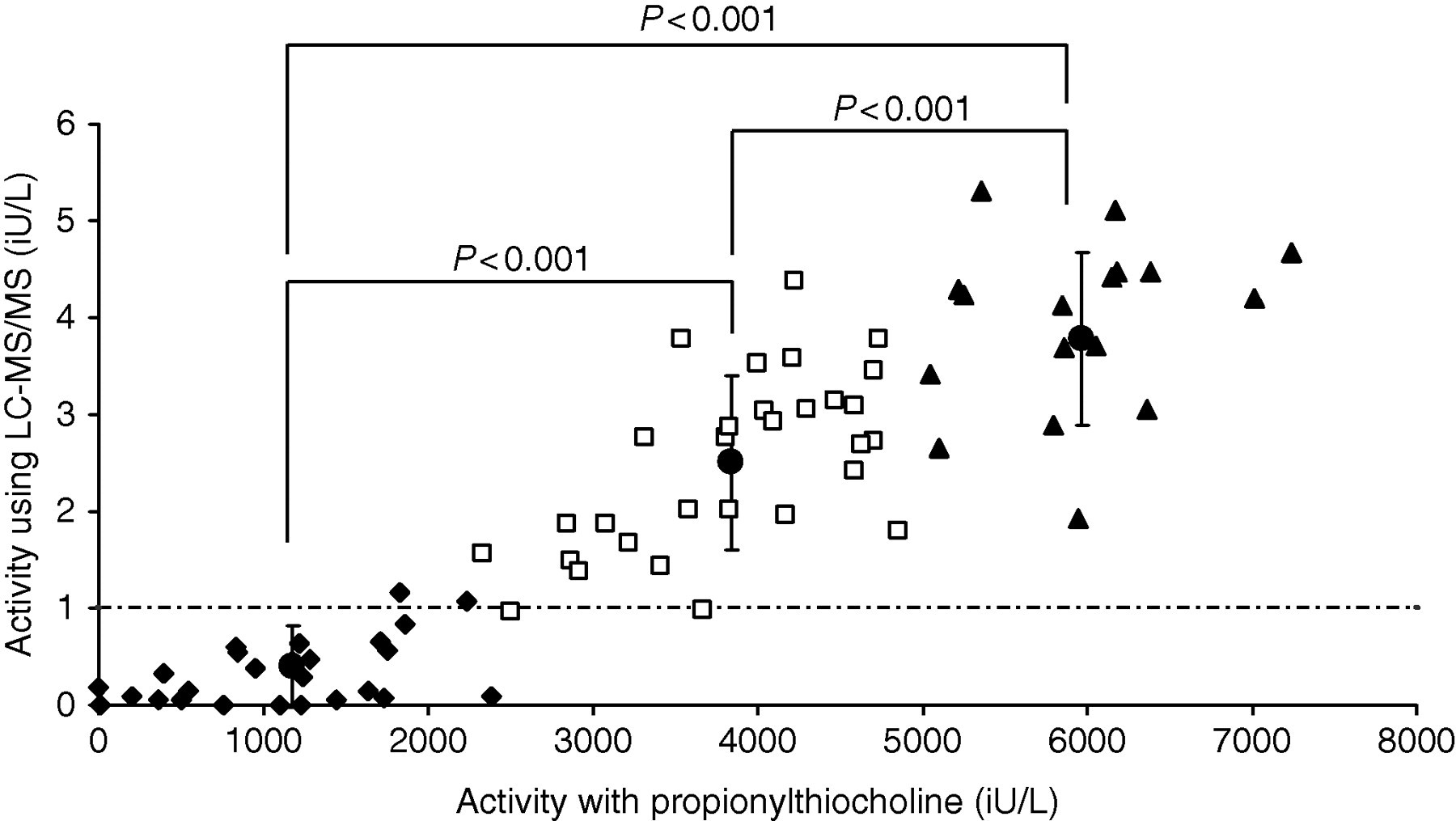
Correlation of cholinesterase activity measured by liquid chromatography linked to tandem mass spectrometry using succinylcholine as substrate compared with the activity measured by propionylthiocholine as substrate. Samples are grouped according to their activity determined using propionylthiocholine. ♦ = <2300 IU/L (n = 29), ▪ = 2300–5000 IU/L (n = 30), ▴ = >5000 IU/L (n = 20) and • = mean values with standard error
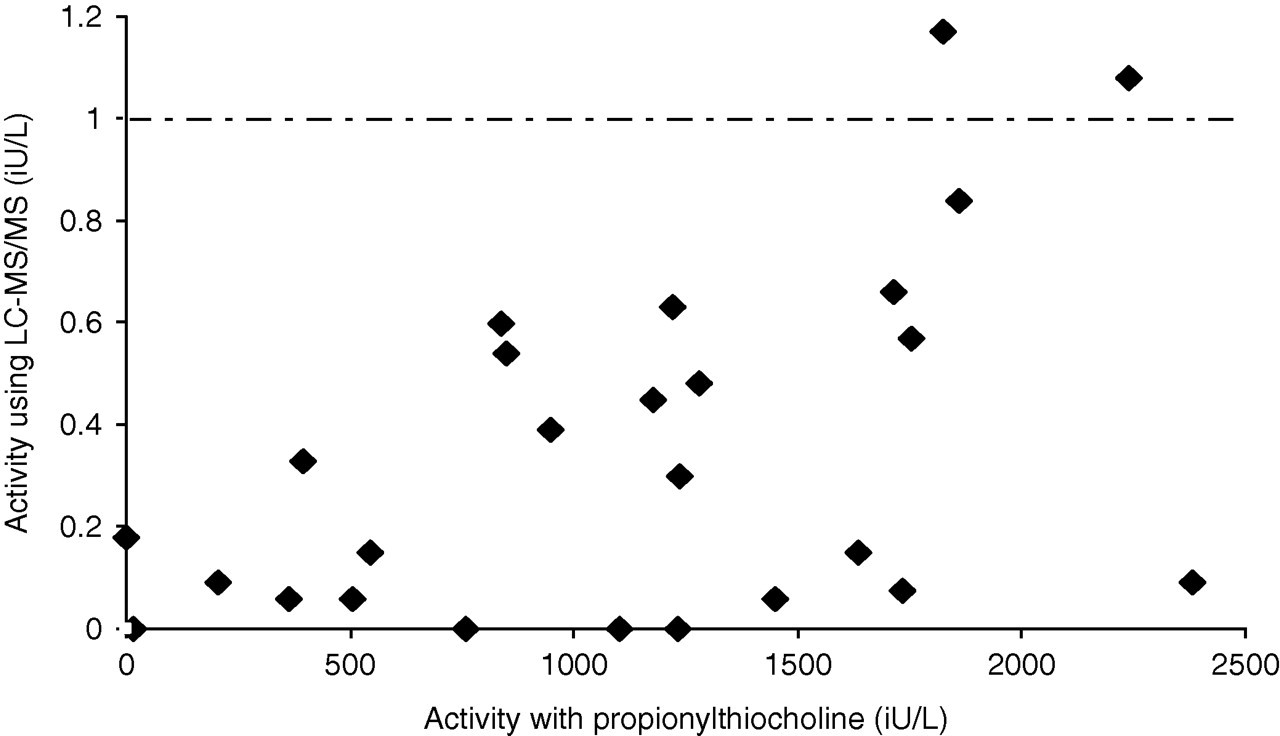
Correlation of cholinesterase activity in samples demonstrating a low cholinesterase activity of <2300 IU/L as measured by propionylthiocholine compared with activities determined by liquid chromatography linked to tandem mass spectrometry using succinylcholine as substrate
Patient comparison
Patient results were grouped according to their clinical significance with regard to their activity as measured by the propionylthiocholine method, whereby those with a cholinesterase activity of <2300 IU/L are deemed at high risk of significant succinylcholine sensitivity; those between 2300 and 5000 IU/L may be at some risk of sensitivity depending on their phenotype or if patients are pregnant (known to reduce cholinesterase activity during its course 14 ). A third group with results above 5000 IU/L currently would be considered at minimal risk of succinylcholine sensitivity. Comparison of the mean activities of these three patient groups as measured using succinylcholine as substrate demonstrated a clear significant difference (Figure 4). From these data a clinically significant cut-off for cholinesterase activity using succinylcholine as substrate could be set at 1 IU/L.
Further comparison of the cholinesterase activities obtained via LC-MS/MS, with respect to patient phenotypes, are presented in Figure 6. Mean activities for most phenotype groups show significant differences and all patients classified as having an atypical, AK or AJ phenotype exhibited a cholinesterase activity of below 1 IU/L using succinylcholine as substrate. This was significantly different from the Usual, UA and UF phenotypes. Significant differences were also observed between the mean activities for Usual and UA phenotypes (P < 0.01); however, it was not possible to distinguish between different heterozygotes including UA and UF as well as AJ and AK phenotypes.
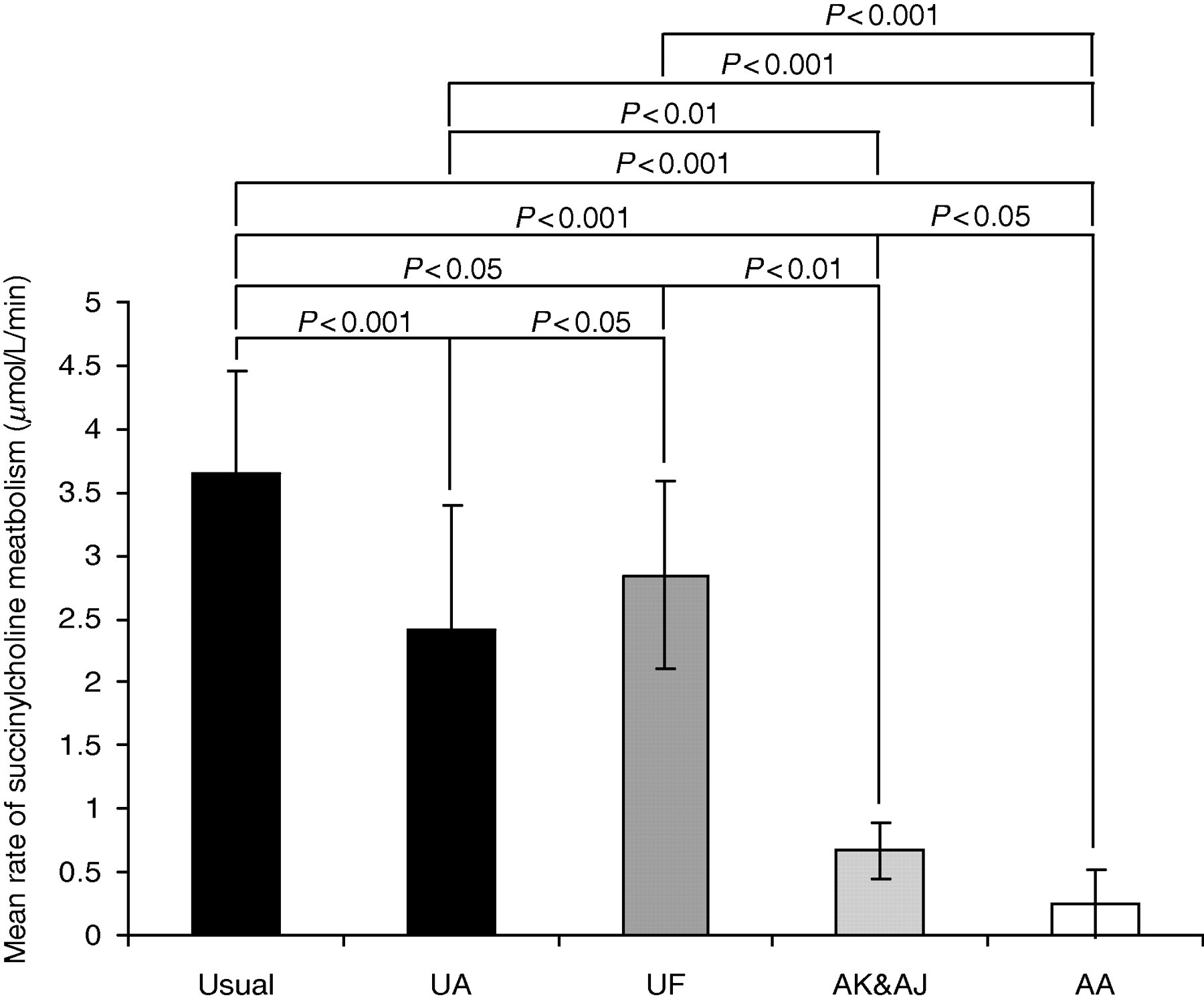
Statistical comparison of the mean activities of cholinesterase as determined by liquid chromatography linked to tandem mass spectrometry using succinylcholine as substrate between different phenotype groups, classified using benzoylcholine-based inhibitor studies
Discussion
Previous measurement of succinylcholine by LC-MS/MS has been challenging. Although succinylcholine ionizes well and provides suitable product ions for selective ion monitoring, its structure and polarity renders it a difficult molecule to chromatograph. Attempts with standard C18 columns have proved problematic and some authors have resorted to direct injection following extensive sample preparation. 16 Such direct injection methods have been suitable for forensic/toxicological analysis using a suitable stable isotopic internal standard. However, direct injection without any chromatography results in very sharp peaks which are difficult to integrate for quantitative analysis. Hence, we have employed an anion exchange resin that increases retention times, presumably due to ion pairing between the succinylcholine and the formic acid present in the mobile phase. This provides a broader peak for integration and enables the reliable quantitative measurement of succinylcholine.
To our knowledge, there are no published methods measuring succinylcholine in the investigation of cholinesterase activity. The use of succinylcholine as a substrate has previously been described but only through analysis of the product choline, using choline oxidase. 11,13,10 This system was deemed to be ‘at least as good’ as propionylthiocholine for the purposes of identifying succinylcholine sensitivity, but no additional benefits over the original method were found. 10 With regards to the benefits of measuring the disappearance of succinylcholine by LC-MS/MS over the choline oxidase method, this technique is not reliant on choline, which is known to increase in plasma independently of cholinesterase. 15 Moreover, as a result of the short kinetic times and the ease of analysis using LC-MS/MS, this method is extremely quick and easy to perform while turn around times are better than that currently offered. In addition, compared with methods utilizing artificial substrates this should represent a more pharmacologically relevant aspect of cholinesterase activity. Whether this technique identifies succinylcholine-sensitive patients where the use of artificial substrates does not remains to be seen. Further work using samples previously proven, by DNA analysis, to carry the ‘scoline variant’ would confirm the benefits of this LC-MS/MS method in identifying these potentially ‘at-risk’ patients deemed low risk by traditional activity methods. In addition investigation of this method with cholinesterase inhibitors such as dibucaine could be explored with regard to phenotype classification. Regardless, we feel the data presented here on enzyme activity alone would be clinically sufficient to identify patients susceptible to succinylcholine sensitivity.
Footnotes
Acknowledgements
The authors would like to acknowledge the guidance and advice of Ms Roberta Goodall with regards to the clinical biochemistry of succinylcholine sensitivity and phenotyping.
