Abstract
Background
Elevated Lipoprotein(a) concentrations are a risk factor for coronary heart disease; however, methodological problems have prevented its introduction to routine clinical practice.
Methods
Thirty-six laboratories each assayed 20 samples (the same 20 in each laboratory) using two different Lp(a) kits per laboratory, randomly assigned from the total of 12 used in the study.
Results
The duplicate error, i.e. the error between-duplicate analyses for each sample, for all kits was small, indicating all kits had a good precision for all the assays. However, there was a very large variation between the kits in the Lp(a) concentration assigned to a sample that could be over 100%. All methods showed a negative or positive bias as the concentration of Lp(a) increased. Most worryingly, as used in this study, several Lp(a) kits detected Lp(a) in a solution of 5% bovine serum albumin in phosphate-buffered saline. The between-laboratory variation in Lp(a) concentration measured using the same kit was very large, e.g. for a sample with a mean concentration of 78.8 mg/dL Lp(a) the between-laboratory variation was 29.7 mg/dL (37.7%). Even with samples with a relatively low Lp(a) concentration of 16.0 mg/dL had a between-laboratory variation of 12.3 mg/dL (76.8%).
Conclusion
There is wide variability in reported Lp(a) concentrations, assayed in the same sample, using different Lp(a) assays. At the present time these differences prevent the use of Lp(a) as a routine diagnostic tool.
Introduction
Lipoprotein (a) (Lp(a)) is a cholesterol-rich lipoprotein whose structure comprises a central low-density lipoprotein core and an apolipoprotein, apo (a), attached to apo B via a single disulphide bond. 1,2 Studies have suggested that Lp(a) is involved in the development of atherosclerosis and thrombosis leading to myocardial infarction and ischaemic heart disease. 3–7 However, data derived from previous epidemiological studies have prevented the use of Lp(a) in routine clinical practice due to limitations in the design/analysis of these studies. 8,9
Accurate measurement of Lp(a) is essential if it is to be used in the assessment of cardiovascular risk. There are more than 18 immunoassay diagnostic kits available in the UK from different manufacturers for the determination of Lp(a). They vary in antibody specificity, measurement principle, assay range and standardization – the latter being a complex issue since no universal standard exists.
In order to determine whether the kits available would yield consistent results we undertook a comparative study organized through the laboratory Sub-Committee of HEART, UK involving laboratories throughout the British Isles.
Methods
Thirty-six laboratories took part in the study (see the Appendix). Manufacturers or distributors provided Lp(a) assay kits. The following 12 kits were used in the study: ELISA kits were provided by Immuno (Vienna, Austria), Mercodia (Upsala, Sweden) and Biopool (Umea, Sweden). Immunoturbidimetric kits were from Immuno, Roche (Welwyn Garden City, UK), Incstar (Wokinghan, UK), Dako (Ely, UK), Wako (Osaka, Japan) and Denka Seiken (Coventry, UK). Immunonephelemetric kits were from Beckman (High Wycombe, UK) and Behring (Marburg, Germany) and an IRMA was provided by Mercodia.
Protocol
Nineteen samples were obtained from a variety of human sources and 36 aliquots produced from each sample. One sample of 5% bovine serum albumin (BSA) in phosphate buffered saline (PBS) acted as a control (Table 1). Each laboratory was sent one aliquot from each of the 20 samples. Only one person (MM) knew the identity of the samples; therefore, assays were performed blind by the laboratories. Of the 20 samples, five were from an identical human source (samples 1, 5, 10 15 and 20), two from another identical human source (samples 18 and 19), one was 5% BSA in PBS (sample 12) and 12 were different pools of human serum. The samples were sent to the participating laboratories the day after collection. Laboratories were requested to freeze the samples until analysis but to run all analyses on the same day. Each laboratory also received a minimum of two Lp(a) assay kits.
Source of study samples and Lp(a) phenotypes
FH, sample from a single subject with homozygous familial hypercholesterolaemia, i.e. samples identical; MG, sample from a single subject with macroglobulinaemia; BSA, bovine serum albumin; PBS, phosphate-buffered saline
Phenotypes were classified by migration rate – S4 = 29 and below, S3 = 30–34, S2 = 35–38 S1 = 39–45, B = 46–51
Phenotyping
Lp(a) phenotypes in the samples were kindly determined by Drs N Al-Khalaf and D Reaveley using immunoblotting following electrophoretic separation in 1% agarose gels (Table 1). Phenotypes were determined by their migration rate relative to apo B.
Results
The duplicate error, i.e. the error between-duplicate analyses for each samples, for all kits was small compared with between-kit variation, indicating all kits had a good precision for all the assays (Table 2). However, there was a very large variation between the kits in the Lp(a) concentration assigned to a sample which could be over 100%.
Results of Lp(a) analyses using the 12 different assay kits. Figures for the individual kits are variance from the grand mean in mg/dL of Lp(a)
The data are presented in three blocks
Block 1 (upper) represents the data from 14 different samples
Block 2 (middle) represents data from five identical samples
Block 3 (lower) represents data from two further identical samples
See Table 1 for details of sample sources
All methods showed a negative or positive bias as the concentration of Lp(a) increased (Table 2).
Most worryingly, as used in this study, several Lp(a) kits detected Lp(a) in a solution of 5% BSA in PBS (Figure 1).
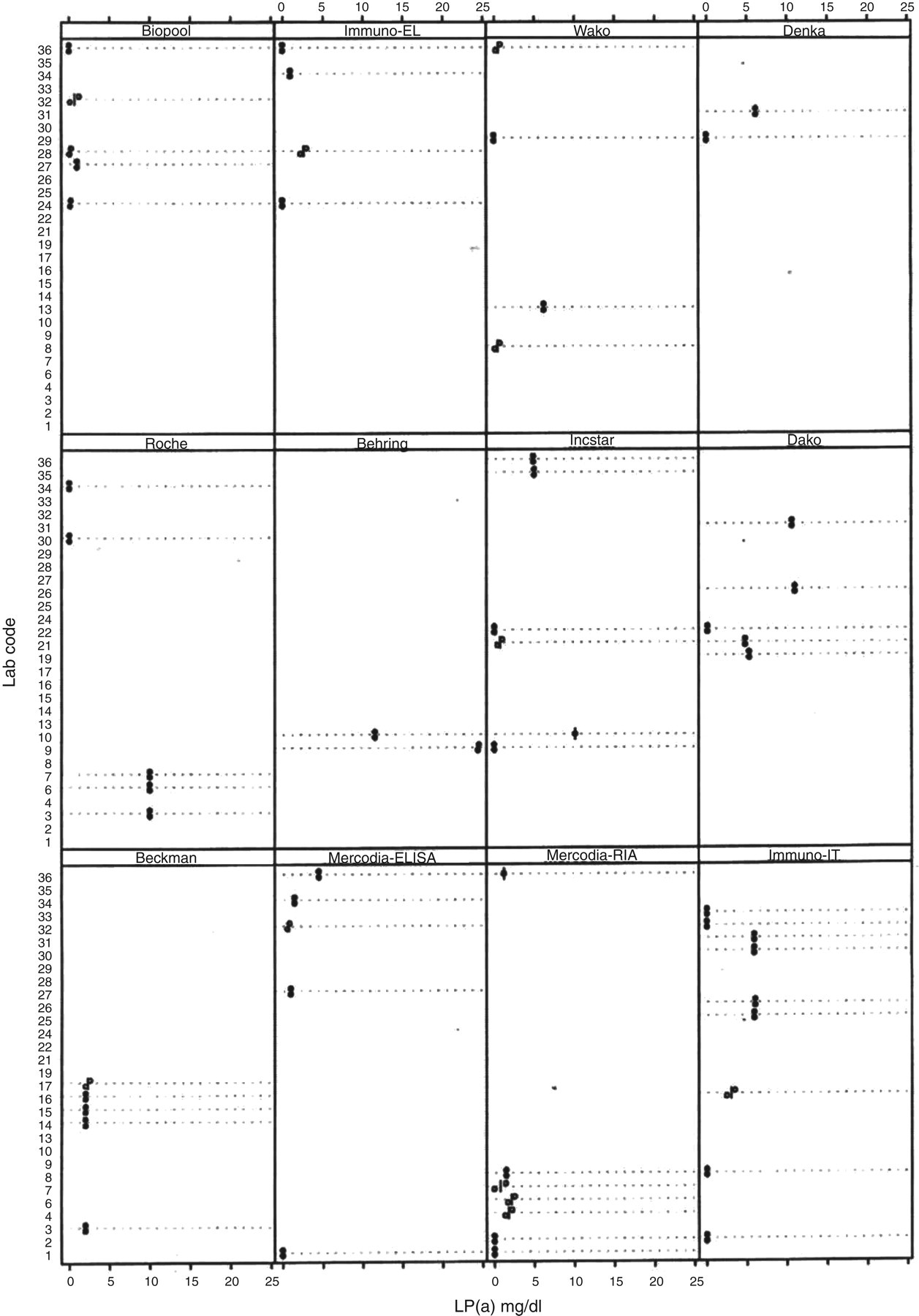
Determination of Lp(a) concentration in a solution of 5% bovine serum albumin in phosphate-buffered saline (containing no Lp(a))
The between-laboratory variation in Lp(a) concentration measured using the same kit was very large, e.g. for a sample with a mean concentration of 78.8 mg/dL Lp(a) the between-laboratory variation was 29.7 mg/dL (37.7%). Even samples with a relatively low Lp(a) concentration of 16.0 mg/dL had a variation of 12.3 mg/dL (76.8%) as calculated from the raw data provided by each laboratory (not shown).
This study also identified that in many cases the manufacturers' instructions for programming auto-analysers to run their assays were wrong, even when the manufacturer made both the Lp(a) assay and the auto-analyser.
Discussion
The results indicate that there are still a great deal of problems with the assay of Lp(a). Different assay kits gave widely different Lp(a) concentrations for the same sample, frequently with positive or negative bias. We have previously shown that the Lp(a) response to growth hormone treatment was entirely dependent on the assay kit chosen. 10 These problems would not be solved by an internationally recognized Lp(a) calibrant as the different assays, although precise, are not accurate and react differently to different Lp(a) concentrations. Manufacturers need urgently to address the problems inherent in the Lp(a) assays available.
In the British Isles, there is a tendency to request Lp(a) concentration as a clinical diagnostic tool. Our results indicated that the between-laboratory and between-assay variation in Lp(a) determination would often render these results as meaningless. There is a case for a moratorium on the use of Lp(a) concentration as a clinical tool until the problems inherent in the assays have been resolved.
This survey was carried out in 1998; however, the problems outlined still persist and the survey should be repeated to determine whether any improvements to the analysis of Lp(a) have been made. If not, those assays with the least problems should be identified and only these should be used until detailed studies can be undertaken to identify the source(s) of the problems and steps taken to eliminate them. In this survey, the assays with the least overall problems were the ELISA supplied by Immuno, the immunoturbidimetric assay from Denka Seiken and the IRMA from Mercodia. The others were associated with considerable problems and require their manufacturers attention.
Footnotes
Acknowledgements
HEART, UK thanks all those manufacturers/distributors of Lp(a) assays who provided their assays free of charge. Mrs Barbara Haynes is especially thanked for technical assistance. All the participating centres are thanked for their accuracy in the study and Ms Caroline Price is thanked for all her expert assistance in many aspects of the study, not least, in the preparation of this manuscript.
