Abstract
Background
Previous studies have shown that albumin in stored urine samples degrades over time, and that albumin losses are greatest in samples with low pH conditions (pH < 5). Furthermore, the high-performance liquid chromatography (HPLC) assay for urinary albumin has been shown to be particularly susceptible to the effects of prolonged storage.
Methods
Frozen urine samples, stored for 12 months at −70 and −20°C, were analysed for albumin fragmentation. Urinary protease activity was investigated in vitro in urine adjusted to pH 2.3–2.5. Albumin was measured by nephelometry, HPLC and sodium dodecyl sulphate-polyacrylamide gel electrophoresis.
Results
In the unadjusted samples, albumin was degraded in 11 out of 40 samples stored at −20°C. In the in vitro experiments, both endogenous albumin and exogenous albumin added to urine were rapidly degraded into large fragments within minutes after adjustment to low pH. The fragments produced were consistent with those produced during digestion with pepsin and urinary degradation was completely inhibited by pepstatin. Albumin concentration measured by HPLC was most dramatically affected, with near-complete loss of albumin-sized material within one hour of incubation at pH 2.3–2.5. Sample reactivity with antiserum in a nephelometry assay initially declined then increased, possibly due to exposure of internal epitopes during albumin digestion.
Conclusions
This study demonstrated that proteases are present and active in stored human urine samples. Urinary albumin digestion occurred in a manner consistent with activity of endogenous urinary proteases. Adjustment to neutral pH or addition of protease inhibitors may be useful techniques for sample preservation.
Introduction
Medical research often involves the use of biological samples that have been frozen or otherwise preserved. Indeed, there is a growing trend for laboratories to gather banks of stored samples that can be used for retrospective analysis, to make best use of limited time and resources available. Previous studies have measured the stability of albumin in frozen samples, with mixed results. Numerous studies have found negligible effect of freezing on urinary albumin concentration 1–3 while others have found considerable albumin losses. 4–9 The use of stored samples for albuminuria research has been criticized due to the potential for long-term degradation of albumin in stored urine.
The rate of apparent albumin loss depends on the assay used to measure urinary albumin. In both research and diagnostic settings, urinary albumin is traditionally measured using immunological methods such as nephelometry and turbidimetry. In 2004, Comper et al. 3 described an alternative assay for urinary albumin using high-performance liquid chromatography (HPLC). The HPLC method uses a size-exclusion separation to measure all albumin-sized material, and typically returns an albumin concentration higher than that measured using immunological assays. The discrepancy between these two assays has been attributed to the presence of immuno-non-reactive forms of albumin in urine, 10 although other authors have suggested that it may be the result of co-elution of other proteins in urine. 11 The exact nature of the material measured by the HPLC remains unknown, although it has been shown that the HPLC assay is more susceptible to changes over time in stored samples than nephelometry. 9
Previous work has suggested that degradation of albumin in urine may be catalysed by proteases. Brinkman et al. 9,12 observed that samples stored at −20°C and those with pH < 5 were most readily degraded during storage for 12 months (compared with samples stored at −70°C and with neutral pH). The same group has since shown that alkalinization of urine samples can preserve albumin concentration in urine. 13 The temperature- and pH-dependent nature of urinary albumin degradation suggests that urinary proteases may be responsible.
The present studies were designed to test the hypothesis that proteases are present in human urine, and that these proteases are able to degrade urine albumin under typical storage conditions.
Materials and methods
Ethics approval and urine samples
Collection of human urine samples was approved by the following committees: the Royal Perth Hospital Human Research Ethics Committee (approval EC 2007/087) for albuminuric patient urine; and the University of Western Australia Human Research Ethics Committee (approval RA/4/1/2263) for urine collected from healthy volunteers. Informed consent was obtained from healthy participants but waived by the committee for samples collected from patients.
Albuminuric samples were selected from a bank of samples collected from critically ill patients admitted to the Royal Perth Hospital Intensive Care Unit, stored in aliquots at either −20 or −70°C as specified.
Normoalbuminuric urine samples were collected from healthy volunteers and used fresh. All samples were screened for the presence of albumin using sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE), then 1 mg/mL of lyophilized human serum albumin (product A1653 from Sigma Aldrich, St Louis, USA) was added.
Albumin degradation in frozen clinical samples
Albumin degradation was examined in 40 clinical samples, stored for 12 months at physiological pH (no adjustments made). Samples were stored in aliquot pairs at −20 and −70°C and then compared using SDS-PAGE. Degraded albumin fragments were identified using mass spectrometry.
Urinary protease studies
Acute acidification in vitro was used as a tool to study the presence of urinary proteases. Urinary albumin degradation was accelerated by incubating samples at pH 2.3–2.5. Optimal pH was determined empirically and at pH 2.3–2.5 both endogenous and exogenous urinary albumin were degraded within minutes of pH adjustment (analysed using SDS-PAGE).
Urinary pH was adjusted to 2.3–2.5 with HCl and maintained with regular monitoring for a period of 48 h. The samples were stirred on a rocker either at room temperature or at 37°C. Aliquots were removed at timed intervals, immediately adjusted to pH 7–10 and stored frozen at −20°C until analysis for albumin and protein (performed within 1 month after the experiment). The t = 0 aliquot was collected immediately after the pH reduction to 2.3–2.5, then returned to neutral pH and frozen as soon as possible.
Urinary proteolytic activity was compared with a pepsin digest, prepared in a separate tube. Pepsin (EC 3.4.23.1) was added to a 1 mg/mL solution of lyophilized albumin at a ratio of 1:1000 w/w (0.8–2.5 units per mg albumin) and stirred at medium speed with a magnetic stirrer at room temperature at pH 2.3–2.5.
Protease inhibition
Urinary degradation of albumin was accelerated as described above in the presence of protease inhibitors. Solutions of pepstatin and leupeptin (Sigma Aldrich products P5318 and L9783, respectively) were prepared according to the manufacturer's instructions. Pepstatin (5 mg) was dissolved by inversion in 90% methanol and 10% acetic acid to a concentration of 1 mg/mL then further diluted in distilled water to make a working solution of 0.01 mg/mL. Pepstatin was added to albumin (1 mg/mL) in urine at a final concentration of 1 μmol/L. Leupeptin was dissolved in water and added to albumin in urine at a final concentration of 100 μmol/L. Inhibition was determined using SDS-PAGE.
Protein assays
Sodium dodecyl sulphate-polyacrylamide gel electrophoresis
Polyacrylamide gels were prepared using the Laemmli method 14 with 12.5% acrylamide (3% crosslinker), and a 4% stacking gel. Ten microlitres of each sample were combined with 10 μL of sample buffer containing 10% SDS, 20% glycerol, 0.2 mol/L dithiothreitol and bromophenol blue and treated in a 95°C water bath for 90 s. A total of 10 μL mixed sample and sample buffer was added to the gel and run at 200 V until the dye front had completely run through the gel. Gels were stained using a solution of 0.2% Coomassie Blue w/v in 50% ethanol and 10% acetic acid.
Gels were allowed to dry between sheets of plastic and selected bands were excised for identification by mass spectrometry.
Mass spectrometry
Protein identification was carried out at the LotteryWest State Biomedical Facility, Proteomics Node located at the Western Australian Institute for Medical Research.
Protein samples were trypsin digested and peptides were extracted according to standard techniques. 15 Peptides were analysed by MALDI-TOF-TOF mass spectrometry using a 4800 Proteomics Analyzer (Applied Biosystems, Life Technologies Corporation, Carlsbad, USA). Spectra were analysed to identify protein of interest using Mascot sequence matching software (Matrix Science, London, UK) with the Ludwig NR Database and the taxonomy set: human. Mascot scores were used to determine confidence in identification and to rank potential protein matches.
Biuret
Total protein was measured using the biuret assay. 16 Four parts biuret reagent (0.15% cupric sulphate, 0.6% sodium potassium tartrate and 3% NaOH) were mixed with one part of sample and the absorbance at 550 nm was measured. Concentrations were calculated using a standard curve of agarose gel electrophoresis purified fraction V bovine serum albumin in distilled water, calibrated at 0, 2, 4, 6, 8 and 10 mg/mL.
Nephelometry
Nephelometry was performed on a Dade Behring BNII Nephelometer using reagents from Dade Behring (Eschborn, Germany). Frozen samples were thawed and centrifuged prior to analysis. This assay had a coefficient of variation (CV) of <3% throughout the study period.
High-performance liquid chromatography
HPLC analysis was performed according to the method of Comper et al. 3 Frozen urine and albumin digest samples were thawed, centrifuged to remove debris and analysed by size separation on a Zorbax Bio-Series GF-450 chromatography column (Agilent Technologies, Santa Clara, USA) in a mobile phase of phosphate-buffered saline. Eluted protein was measured using absorbance at 214 nm relative to 30 and 300 mg/L albumin standards diluted in deionized water from a commercial solution of human serum albumin (Sigma Aldrich product A6909). The interassay CV of this assay was 4.4% at a concentration of 170–198 mg/L.
Results
Albumin fragments produced over time in stored urine samples
Three urine samples (all proteinuric) from the 40 samples analysed by SDS-PAGE after storage are shown in Figure 1. The albumin band present in the Sample 1 aliquot stored at −70°C has almost completely been degraded in the aliquot stored at −20°C. Furthermore, two new bands have appeared that were confirmed by mass spectrometry to be fragments of albumin with Mascot scores of 276 and 199 (indicates identity or extensive homology with confidence level P < 0.0001). These bands were not present in the sample stored at −70°C. Samples 2 and 3 had similar protein patterns in the two aliquots and did not appear to be susceptible to degradation at −20°C. Of 40 samples analysed by SDS-PAGE, 11 appeared to show degradation of the albumin band when stored at −20°C compared with their matched aliquots stored at −70°C.
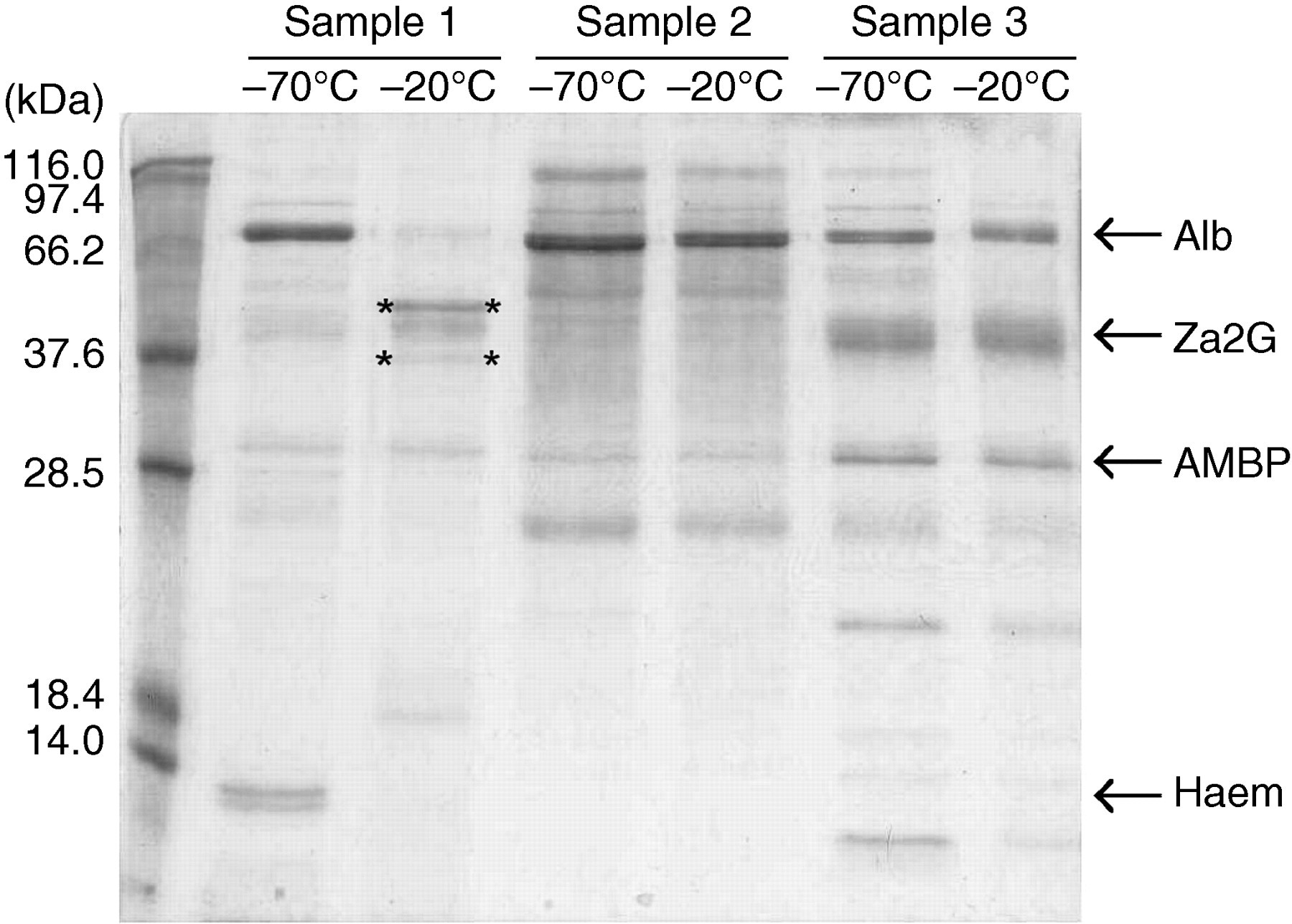
Urinary proteolytic activity under conventional storage conditions. Proteinuric urine samples were stored frozen in aliquots for one year at −20 and −70°C. The albumin band at 66 kDa in the −70°C aliquot of Sample 1 has been degraded during storage at −20°C. Bands indicated with ** were confirmed by mass spectrometry as fragments of human serum albumin. Samples 2 and 3 were not affected by storage temperature. Out of 40 samples analysed, 11 exhibited albumin fragmentation when stored at −20°C. Alb, human serum albumin; Za2G, zinc-alpha-2-glycoprotein; AMBP, alpha-1-microglobulin; Haem, haemoglobin subunits alpha and beta
Degradation of albumin by urinary proteases activated at low pH
Addition of HCl to pH 2.3–2.5 induced rapid degradation of both endogenous and exogenous urinary albumin at room temperature. As degradation progressed over time, fragments were produced in a distinct pattern including prominent bands at sizes 67, 48, 31, 28 and 18 kDa (Figure 2). The pattern of urinary albumin fragments was similar in appearance to that produced by digestion of albumin with pepsin. By t = 1 h the albumin band at 66 kDa was no longer visible and the only fragments remaining after 48 h of incubation were <25 kDa in size. Degradation was not observed in the following controls: albumin in water incubated at pH 2.3–2.5 (no urine control), albumin in unadjusted urine (pH not known, 48 h) or albumin in urine adjusted to pH 2.3–2.5 and immediately returned to pH 7–10 (t = 0 control) (Figure 3).
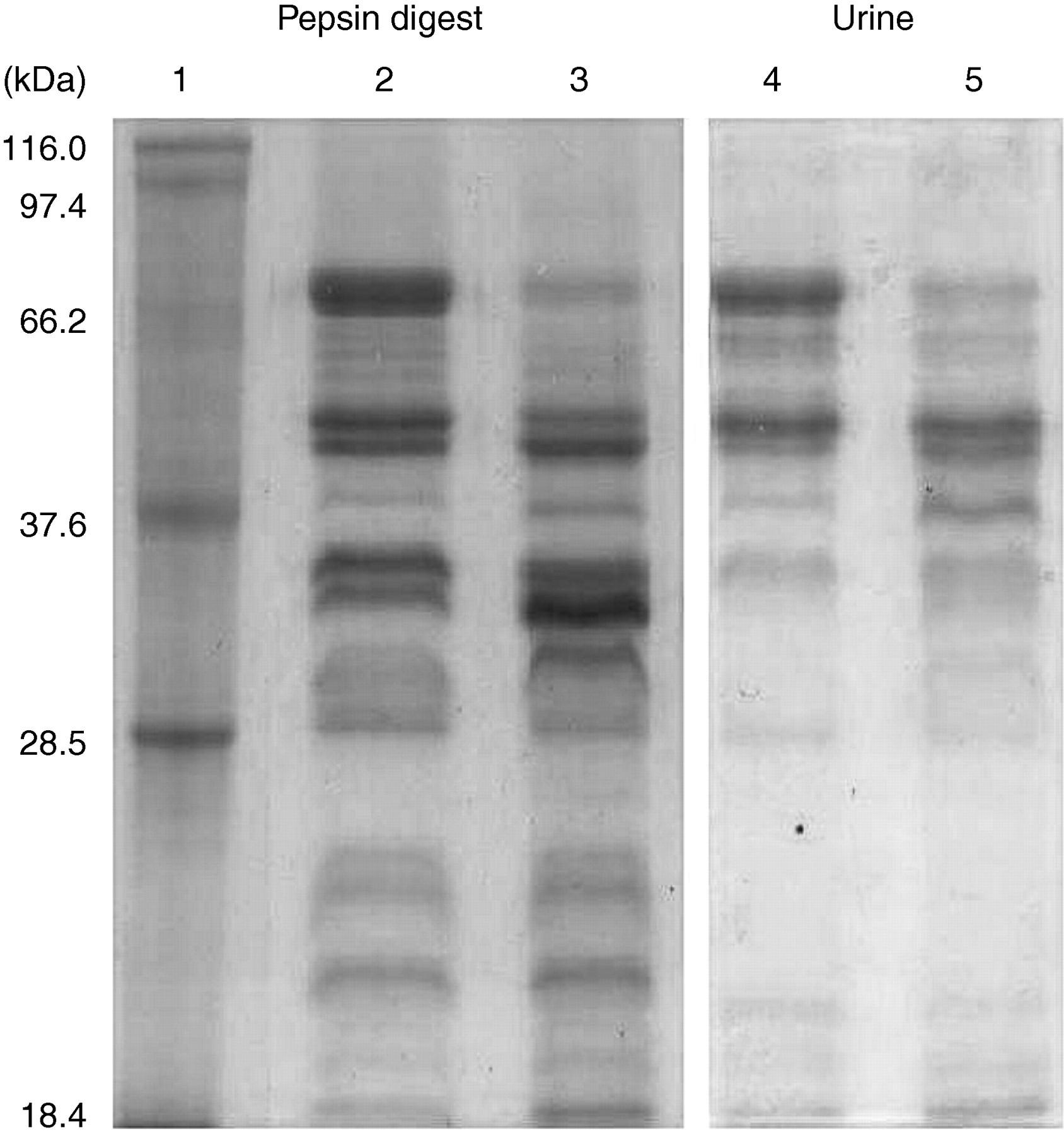
Activation of urinary proteases leads to rapid degradation of albumin into fragments (lanes 4 and 5) similar to those observed during in vitro digestion with pepsin (lanes 2 and 3). Lane 1: Mw standards; lanes 2 and 3: a solution of 1 mg/mL HSA with pepsin added at a ratio of 1:1000, incubated at pH 2.3–2.5 for 30 and 60 min, respectively; lanes 4 and 5: Healthy urine with 1 mg/mL HSA added, incubated at pH 2.3–2.5 for 30 and 60 min, respectively
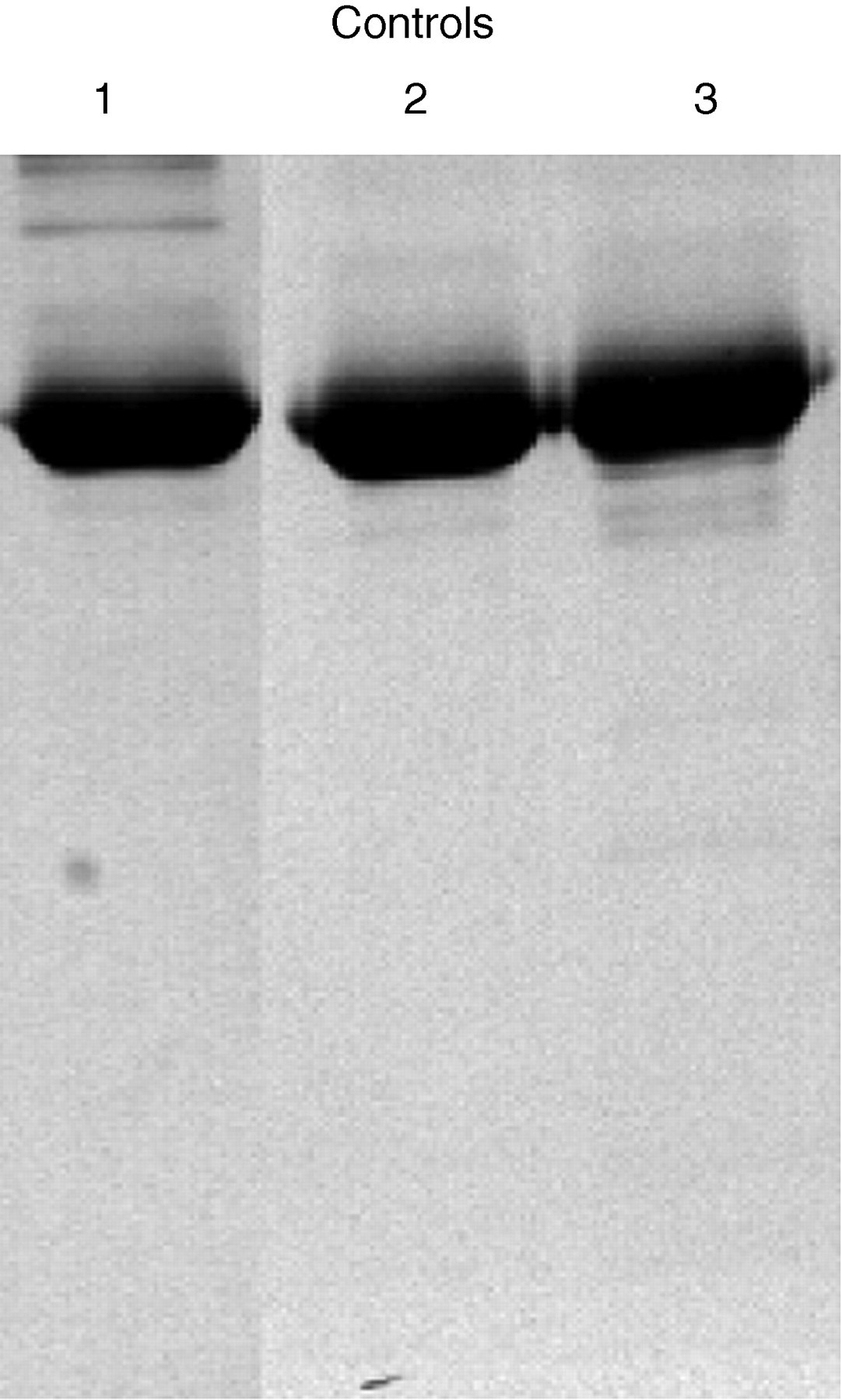
Control samples showing no degradation. Lane 1: albumin in water incubated at pH 2.3–2.5 (no urine control); lane 2: albumin in unadjusted urine (mildly acidic control, 48 h); lane 3: albumin in urine adjusted to pH 2.3–2.5 and immediately returned to pH 7–10 (t = 0 control)
Urinary degradation of albumin was time dependent, pH dependent and accelerated at 37°C. Degradation was completely inhibited by the presence of pepstatin, but leupeptin had no effect on urinary proteolytic activity (Figure 4), which suggests that the urinary proteases may belong to the pepstatin-sensitive aspartyl protease family.
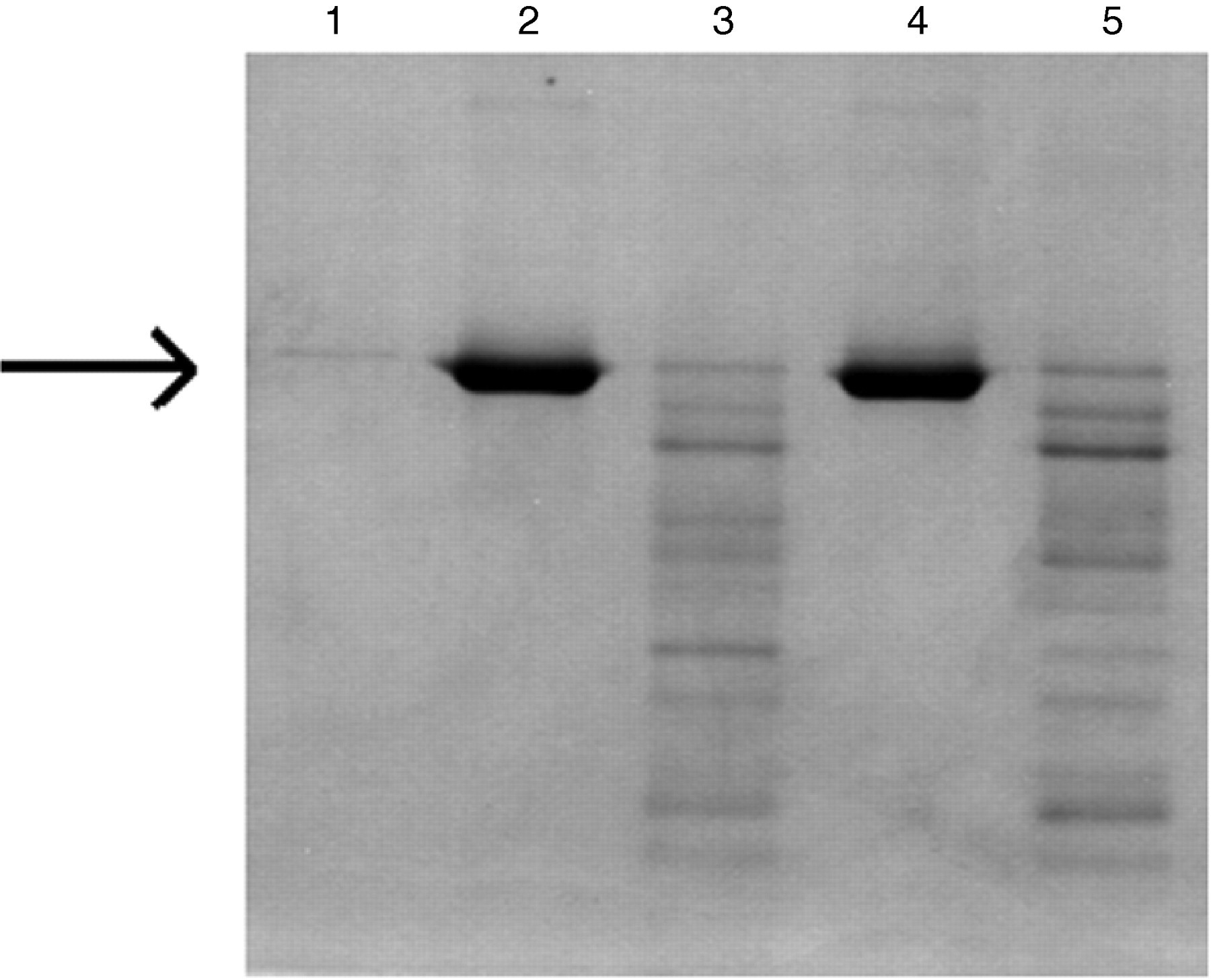
Effect of protease inhibitors on urinary digestion of albumin (arrow). Lane 1: non-albuminuric urine; lane 2: urine with HSA added; lane 3: urine with HSA, incubated at pH 2.3–2.5 for one hour; lane 4: pepstatin added to urine with HSA, pH 2.3–2.5 for one hour; lane 5: leupeptin added to urine with HSA, pH 2.3–2.5 for one hour
Effects of urinary proteolytic activity on measured urinary albumin concentration
Total urinary protein (determined by the biuret assay) was not affected by incubation at low pH (Figure 5). The HPLC assay was most dramatically affected by low pH degradation by urinary proteases (Figure 5a) and pepsin (Figure 5b), with 90% of albumin reactivity lost in the first 30 min after pH adjustment. Nephelometry was also affected by degradation, although the loss of reactivity was slower than for HPLC. Albumin digested with pepsin gradually lost reactivity with the nephelometry assay until approximately t = 30 min, then gained reactivity at approximately t = 40 min to 1 h. The increase was confirmed in independent reactions (Figure 6).
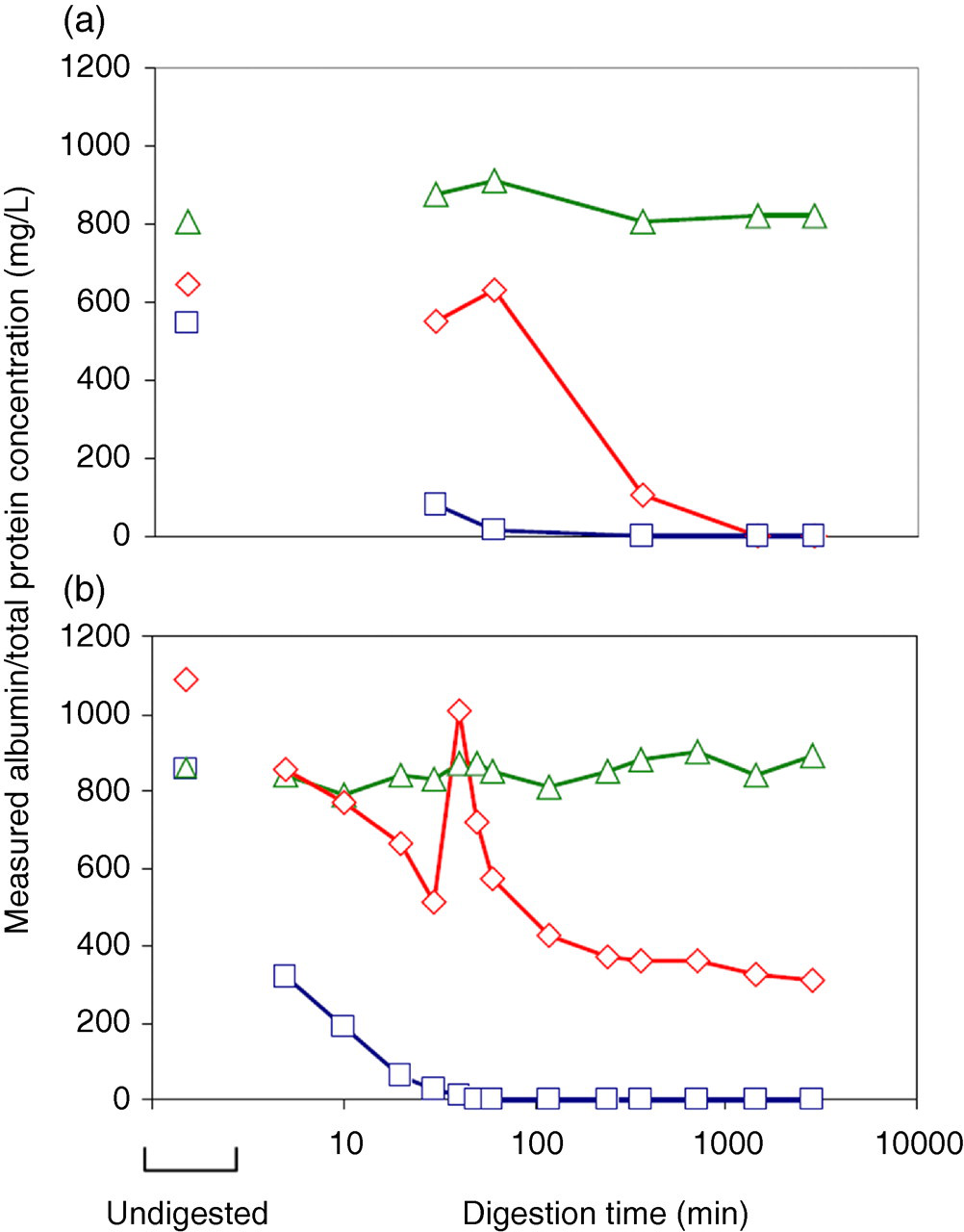
Apparent concentration of albumin during proteolytic digestion as measured by the biuret assay for total protein (triangle), nephelometry using albumin antiserum (diamond) and the HPLC assay for urinary albumin (square). (a) Albumin degradation by proteases in human urine, incubated at low pH; (b) in vitro degradation of albumin with pepsin. Representative profiles are shown from six pepsin digests of human serum albumin and nine human urine samples. Note the logarithmic scale on the x-axis
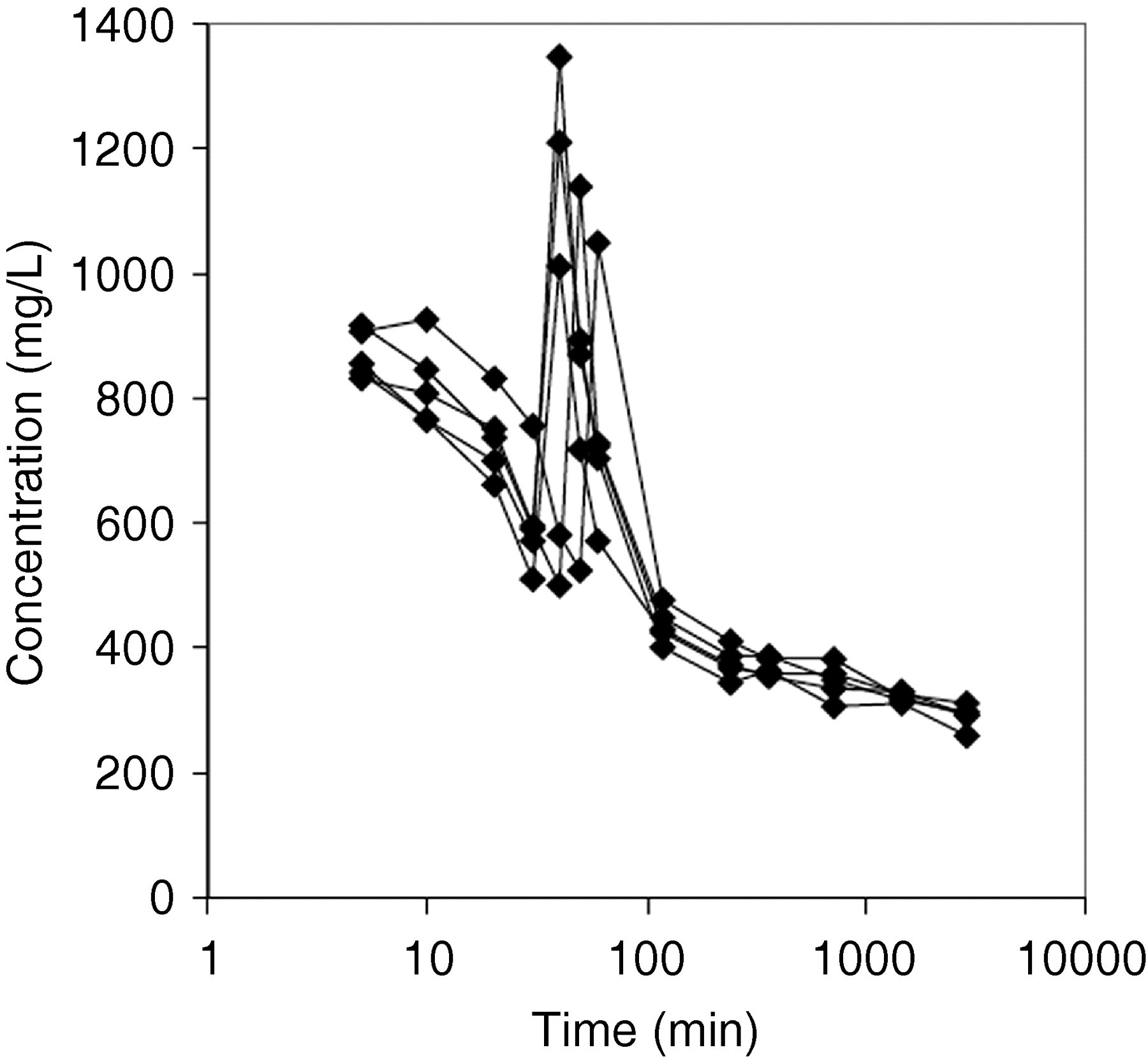
Effect of pepsin digestion at pH 2.3–2.5 on the concentration of albumin measured using nephelometry. Replicate data from five independent reactions are shown. Note the logarithmic scale on the x-axis
Discussion
Previous observations 9,12 of albumin degradation in stored urine suggest that urinary proteases may be involved. This study confirms that urinary proteases are indeed present in urine and that they are capable of degrading albumin rapidly (within minutes) when activated at pH 12.3–2.5. Addition of pepstatin, an aspartyl protease inhibitor, completely inhibited urinary proteolytic activity. Urinary proteases were also shown to degrade albumin under conventional storage conditions, in samples frozen at −20°C and at physiological pH (>5) over a 12-month period.
Proteolytic activity at low pH values has previously been observed in urine samples 17 and cathepsin D has been implicated as a source of urinary proteolysis. 18 Although this study compared urinary proteolytic activity with that of pepsin, cathepsin D is a good candidate as the enzyme responsible for urinary proteolysis; it has a similar cleavage preference to pepsin and is also inhibited by pepstatin. Previous attempts have shown no proteolytic activity in urine over a period of 48 h at physiological pH. 19 Short-term results at physiological pH do not demonstrate the absence of urinary proteases as proteases are only moderately active at pH >5. Furthermore, small studies may show bias as there appears to be some intersample variation in urinary proteolytic activity.
The degradation induced in this study produced a characteristic series of large albumin fragments. Similar fragments have been observed previously in vitro 20,21 and in human urine samples. 22–24 Although these fragments may have diagnostic utility, it has been speculated that urinary albumin fragments are products of albumin degradation during storage. 25 During this study, we confirmed that fragmentation patterns can change as a result of storage (Figure 1). Degradation was not seen uniformly across all samples, suggesting intersample variability that may be due to several factors, including differences in pH, concentration and composition of urinary proteases, and the presence of co-factors that may contribute to or inhibit endogenous protease activity.
Low pH degradation of albumin with pepsin caused both a loss and gain of reactivity with nephelometry antiserum. Approximately 40 min after adjustment to low pH, the apparent concentration of albumin measured by nephelometry increased rapidly, in some cases to be higher than the starting concentration of albumin in the sample. This may be postulated to reflect exposure of new epitopes on the surface of the albumin molecule. To our knowledge, this phenomenon has not previously been described. Some studies have reported an increase in albumin concentration in urine samples after prolonged storage, which could occur due to endogenous proteolytic activity. 12,26 These findings have important implications for measurement of albumin using immunological methods. A single urinary albumin concentration may be erroneously high or low depending on the extent of degradation and the availability of immunoreactive epitopes on albumin molecules. This study employed only one immunogenic assay, using a Dade Behring nephelometer. It is possible that different assay conditions or antisera may yield different results, and further studies would add to our characterization of changes to the albumin molecule during proteolytic degradation. Reference protocols and standards for measuring urinary albumin are needed.
The HPLC assay was the most dramatically affected by low pH incubation. The apparent concentration of albumin was reduced to <10% within the first hour of incubation. Previous studies have shown that this assay is also highly susceptible to changes during freezer storage. 9 When fresh urine samples are analysed, the HPLC assay invariably measures an albumin concentration higher than that measured using nephelometry. The difference between the assays has been attributed to the presence of immuno-non-reactive albumin in urine 10 and it may be partially due to co-elution of albumin with other urinary proteins during size exclusion. 11 The HPLC assay has been of interest since Comper et al. 3 demonstrated that this assay is a superior method for detection of progression towards persistent albuminuria, with lead times of 3.9 and 2.4 y over immunoassays in type I and type II diabetic patients, respectively. Urinary degradation of protein has a disproportionate effect on the HPLC assay, so concentrations measured using the HPLC assay in stored samples as was the case in the Comper study are likely to be underestimated.
While most studies conclude that albumin is stable in urine stored at −70°C, the stability of albumin in samples stored at −20°C is controversial. Some studies have found substantial degradation of albumin in frozen samples, 4–9 while others have reported stable urinary albumin concentrations in samples stored at −20°C for several months 1,2 or even years. 3 These studies invariably measure albumin using immunological methods, which may not be able to detect degradation of albumin. The present study confirmed earlier observations 9 that the degree of albumin degradation observed is highly dependent on the assay used. Furthermore, intersample variability may give rise to variable results between studies. Brinkman et al. 12 demonstrated greater losses in measured albumin concentration in those samples with low pH,and that adjustment of samples to pH 8.0 prior to storage can preserve urinary albumin concentration. 13 Their results are consistent with the proposal that urinary proteolytic activity is responsible for changes to urinary albumin concentration in stored samples. Other models have been proposed to explain albumin degradation during freezing, including albumin loss through the formation of a precipitate. 4 The precipitation model is supported by the finding that the addition of Tween-20 can restore albumin concentrations in samples that have previously been frozen. 27 Our study confirmed the presence of endogenous urinary proteases with a capacity to degrade albumin; however, it is possible that changes to albumin concentration observed in stored urine samples may arise from more than one mechanism. When comparing results from different assays, it is important to consider the standards used. In the current study, different standards were used to calibrate the HPLC, nephelometry and biuret assays, which may explain subtle differences in the concentration of control (undigested) samples (Figure 5).
Although this study was limited to urinary albumin, it is important to note that urinary protease activity is likely to have a detrimental effect on storage of all proteins. Banks of stored samples are increasingly being used to search for novel biomarkers of disease. It is not yet known to what extent urinary proteases will affect the integrity of novel biomarkers in urine; however, storage conditions must be considered at the time of collection if the samples are to be used for retrospective analysis. Pepstatin was able to completely inhibit urinary digestion of albumin, so for some studies it may be possible to add this protease inhibitor to samples prior to storage as a means of preservation. Alternatively, samples may be adjusted to neutral pH prior to freezing 13 and frozen at low temperatures (−70°C) to prevent degradation. Ideally, analysis for novel biomarkers should be conducted as soon as possible after sample collection.
Conclusions
Albumin in stored urine samples is degraded over time, most likely due to the presence of endogenous urinary proteases. Degradation occurs rapidly at low pH and has a profound effect on assays for urinary albumin. Albumin measured by the HPLC assay is particularly susceptible to proteolytic degradation. Studies that measure albumin in stored urine samples must be interpreted with caution. Storage at neutral pH, at low temperatures (−70°C) or in the presence of protease inhibitors may offer protection against urinary albumin degradation.
DECLARATIONS
