Abstract
We present the first national audit of the Short Synacthen Test (SST), identifying the clinical, analytical and interpretative procedures adopted by 89 laboratories.
Background
The SST has replaced the insulin stress test as the first-line test to assess adrenal insufficiency and has received considerable attention regarding its sensitivity and specificity. Concerns regarding this test include the bias of cortisol methods, cut-off values used, contraindications and the limitations of the test in diagnosing recent, mild secondary adrenal insufficiency. The audit took into consideration the protocols used by laboratories, the advice provided prior and after the SST and the analytical bias of the methods used.
Methods
A web-based questionnaire using Microsoft FrontPageTM was prepared to collect data from laboratories and provided drop-down lists and other form-field elements to capture additional comments. The resultant data were exported to Microsoft ExcelTM for data clean-up and analysis.
Results
The workloads were highly variable; however, most laboratories were in general agreement to the indications, contraindications, timing and reference ranges. In contrast, there was variability in the bias of the cortisol methods, which had not been translated to the cut-off values used by the majority of laboratories.
Conclusions
The audit has shown that though the preanalytical procedures were similar in most laboratories, there is a requirement to recognize the effect that method bias may have on the reference ranges and consequently on the diagnosis of adrenal insufficiency. There is a need to develop consensus guidelines, which can aid both clinicians and laboratories.
Introduction
The hypothalamic–pituitary–adrenal (HPA) axis is a well-documented endocrine pathway, which utilizes negative feedback to regulate various essential hormones. Corticotrophin-releasing hormone, produced by the hypothalamus, stimulates the production of adrenocorticotrophic hormone (ACTH) in the pituitary, which in turn stimulates the secretion of cortisol by the adrenal cortex. The HPA axis has an important role to enable the body to cope with stresses such as infection, hypotension and surgery. 1 Suppressed cortisol production by the adrenal gland (hypoadrenalism), even to a mild degree, has been associated with increased mortality in an acute setting. 2 The causes of primary and secondary adrenal insufficiency are shown in Table 1. It is important in patients where adrenal insufficiency is suspected that a quick diagnosis be made to avoid adrenal crisis. 3
Causes of adrenal insufficiency, taken from Oelkers 1 . Copyright © [1996]. Massachusetts Medical Society. All rights reserved.
Permission reference number RY-2010-2442
The ‘gold standard’ test for the integrity of the HPA axis is the cortisol response to insulin-induced hypoglycaemia – the insulin stress test – IST. However, this is now considered too hazardous and resource intensive, and has been largely replaced by the Short Synacthen Test (SST). 4 Synacthen (tetracosactide, tetracosactrin) is a synthetic analogue of ACTH, consisting of the N-terminal 24 amino acids of the 39 amino acid native molecule and possesses full biological activity. The test is usually performed in an outpatient setting, and involves blood sampling for cortisol before and 30 min (and sometimes 60 min) after administration of an intramuscular or intravenous bolus of Synacthen.
Previous audits of the procedures and scientific measurement of the SST have been carried out by UK local regions, with the most comprehensive being the Wales regional audit, 5 published in November 2005. This audit surveyed 13 Welsh laboratories and showed wide differences in protocols for performing and interpreting SSTs in general medical patients. Its design and recommendations were used as a starting point for this national audit. Whereas some of the most useful clinical audits are those conceived locally and which have direct relevance to the delivery of a particular local service, national audits can avoid duplication of effort and allow a standardized means of assessing practise to ensure UK-wide consistency with national standards.
The audit aimed to ascertain the protocols recommended by laboratories, which included the details of the procedures adopted and the analytical aspects that may affect the interpretation of results.
Methods
Survey design
The Association for Clinical Biochemistry (ACB) national audits have normally been conducted using paper forms. These, however, have a number of disadvantages, principally that a hand-written text is difficult to read and time consuming to enter into databases. Furthermore, free-text responses, even when legible, are hard to parse and analyse. It was therefore decided to take a web-based approach using forms, and exploit the use of drop-down lists and other form-field elements that enable responses to be structured in a way that facilitates analysis. For responders not able to find their selection in a drop-down list, a free-text field was provided, thus enabling laboratory-specific information to be captured.
Data gathering and analysis
A further advantage of the web-based approach was the facility offered by the form handler inside Microsoft FrontPageTM, for responses to be cumulatively assembled into a tab-delimited file, which could be exported into a spreadsheet or relational database for analysis, without the need for manual data entry. The final webserver database was exported to Microsoft ExcelTM, where it was edited to remove duplicates and formatting inconsistencies. Separate columns were added for separating free-text comments. This resulted in 89 responses. This spreadsheet was then imported into Microsoft AccessTM to enable specific questions to be asked about the data-set.
Review of laboratory protocols
As part of their responses, laboratories were asked to supply copies of their SST protocols. It was decided to analyse these (a) against Certified Public Accountant (CPA) (UK) Ltd accreditation standards for document control and (b) matching of the protocol with the audit survey response.
Results
Eighty-nine responses were received using the web-based questionnaire and we present each response or group of responses, by stating the question first in italics, and then a summary of the responses given below.
[Q2.1] How many SSTs were assayed in your lab in 2006?
The number of SST's assayed in the laboratories varied greatly, with some not doing any to some doing over a 1000 a year.
[Q2.2] How many of these tests were performed by your laboratory's medical staff?
Numbers were highly variable, the median being 200 per annum. Numbers performed by the laboratory medical staff within their departments were much lower.
[Q3.1] Do you use random or 09:00 hr cortisol to screen for adrenal insufficiency?
Fifty-one percent of respondents used a 09:00 timed test (Figure 1), 39% used other times and 10% performed the test randomly. By providing a section for participants to comment on the screening test, we found that nine laboratories who selected ‘other’ as a response to this question would prefer 09:00 sample but would accept a random, whereas seven laboratories would proceed immediately to an SST.
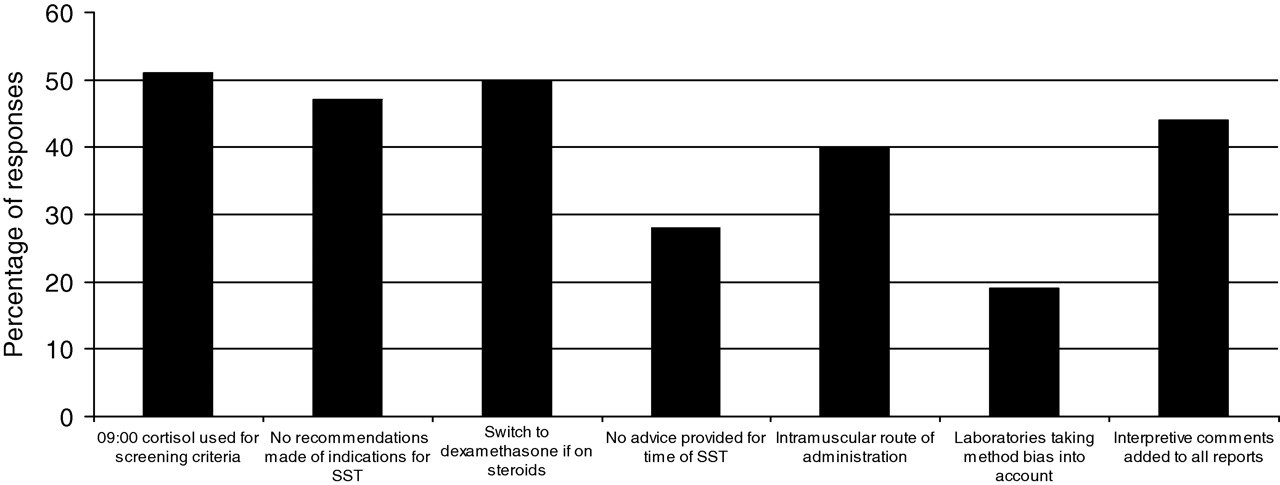
Summary of responses to audit questions regarding preanalytical, analytical and postanalytical aspects of SST tests. SST, Short Synacthen Test
[Q3.2] Do you make recommendations about the indications for testing?
Forty-seven percent of respondents do not make recommendations for testing (Figure 1), while those that did stated the most common indications for an SST were primary and secondary adrenal failure, clinical picture, hyponatraemia and to assess adrenal glucocorticoid reserve.
[Q3.3] What contraindications for testing do you suggest?
Laboratories generally agreed that the contraindications for testing are steroid therapy, allergy, asthma and pituitary surgery. A few laboratories also identified others such as oral contraceptive pill, hormone replacement therapy, pregnancy and patients already in crisis.
[Q4.1] Does your laboratory have a written protocol for the SST?
[Q4.2] Has this protocol been agreed with your local endocrinologists?
Ninety percent of respondents had written protocols, but 88% of these written protocols had been agreed with clinicians.
[Q4.3] For patients already on steroid therapy what do you recommend before doing the SST?
Fifty percent of respondents suggested switching to dexamethasone (Figure 1) and 28% would omit the evening dose of steroid.
[Q4.4] Do you advise that the patient is fasting before the test is done?
Only two respondents (∼2%) recommended fasting.
[Q4.5] Do you advise on any particular time of day for performing the test?
Twenty-eight percent of laboratories would not provide advice on the time the test was performed (Figure 1), while the other 72% of laboratories would advise a 09:00 or morning test.
[Q4.6] What dose of Synacthen do you suggest for a standard test in an adult?
One hundred percent of respondents recommended 250 μg.
[Q4.7] What dose of Synacthen do you suggest for use in children?
The majority of the replies to this question used the general rule of using a calculation (250 μg/1.73 m2) to determine the body surface area to adjust the dose accordingly.
[Q4.8] Do you use ‘physiological dose’ tests (eg 1 μg or 5 μg)?
Fifteen laboratories used 1 μg. One laboratory used 5 μg and one used 0.5 μg.
[Q4.9] What routes of administration do you use/recommend?
Forty-four percent of laboratories used or recommended intramuscular routes of administration (Figure 1) and 12% would use intravascular routes of administration, while 40% would recommend both.
[Q4.10] What sampling times do you recommend?
Fifty-two laboratories recommended sampling times of 0 and 30 min, while 35 laboratories recommended 0, 30 and 60 min. One laboratory used sampling times of 0, 30 and 40 min, while another used 0, 30 and 45 min.
[Q5.1] Which analytical method do you use for serum cortisol?
The most widely used analytical cortisol methods were the Roche E170 Modular and the Siemens ADVIA Centaur followed by the Immulite 2000/2500 and the Beckman Access/DXL.
[Q5.2] What are the stated percentage cross-reactivities for your method for these compounds?
Of 89 laboratories, 84 (94.4%) responding to this question gave correct cross-reactivity figures for cortisone, prednisone and prednisolone for their method.
[Q5.3] What is the imprecision (%CV) of your method at a cortisol level of 500–600 nmol/L?
Figure 2 shows the imprecision of each method as reported by laboratories.
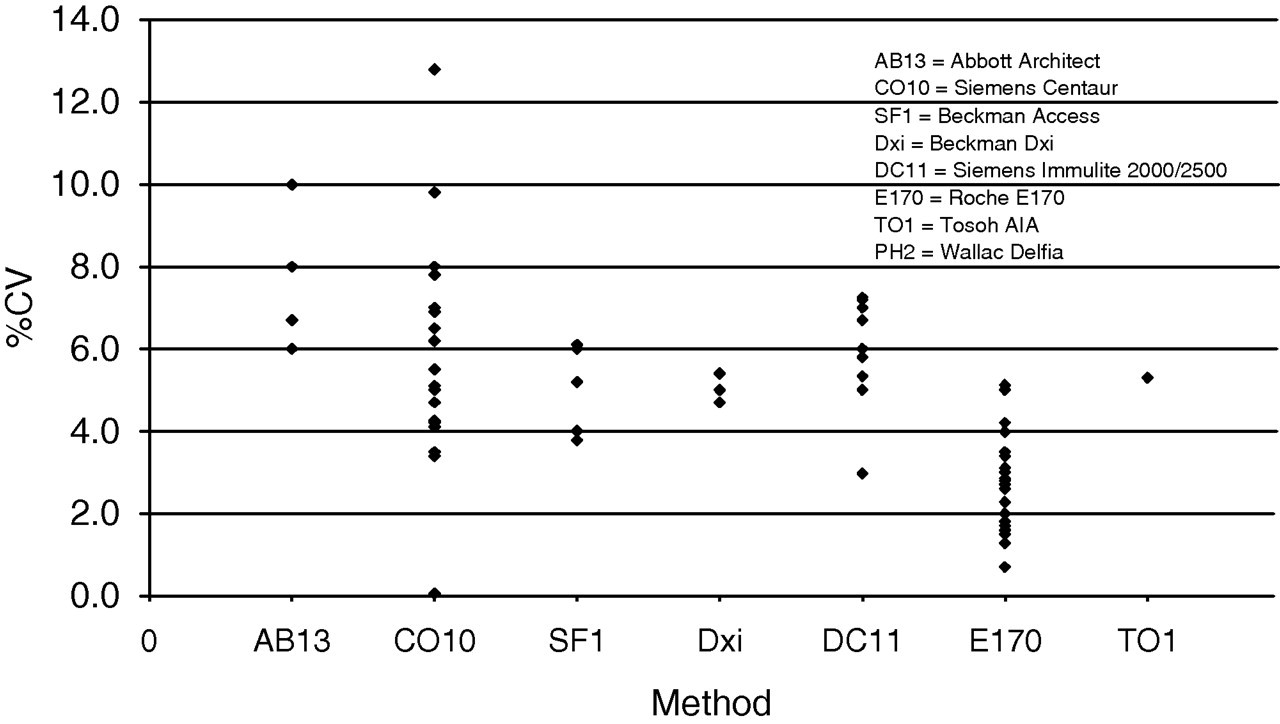
Reported %CV at a cortisol concentration of 500–600 nmol/L for each of the cortisol methods used by laboratories. %CV, percent coefficient of variation
[Q5.4] In which EQA schemes do you participate for serum cortisol?
One hundred percent of respondents were participating in the UK NEQAS for steroid hormones. Two laboratories used BioRad as a secondary EQA scheme. One laboratory used WEQAS as a secondary EQA scheme.
[Q5.5] What is/are the current mean method bias(es) in your EQA scheme(s)?
It was found that 44% of laboratories did not report the correct method bias. Figure 3 shows the correct method bias for distribution 328 at the cut-off values used by the laboratories for the baseline, increment and 30 min cortisol.
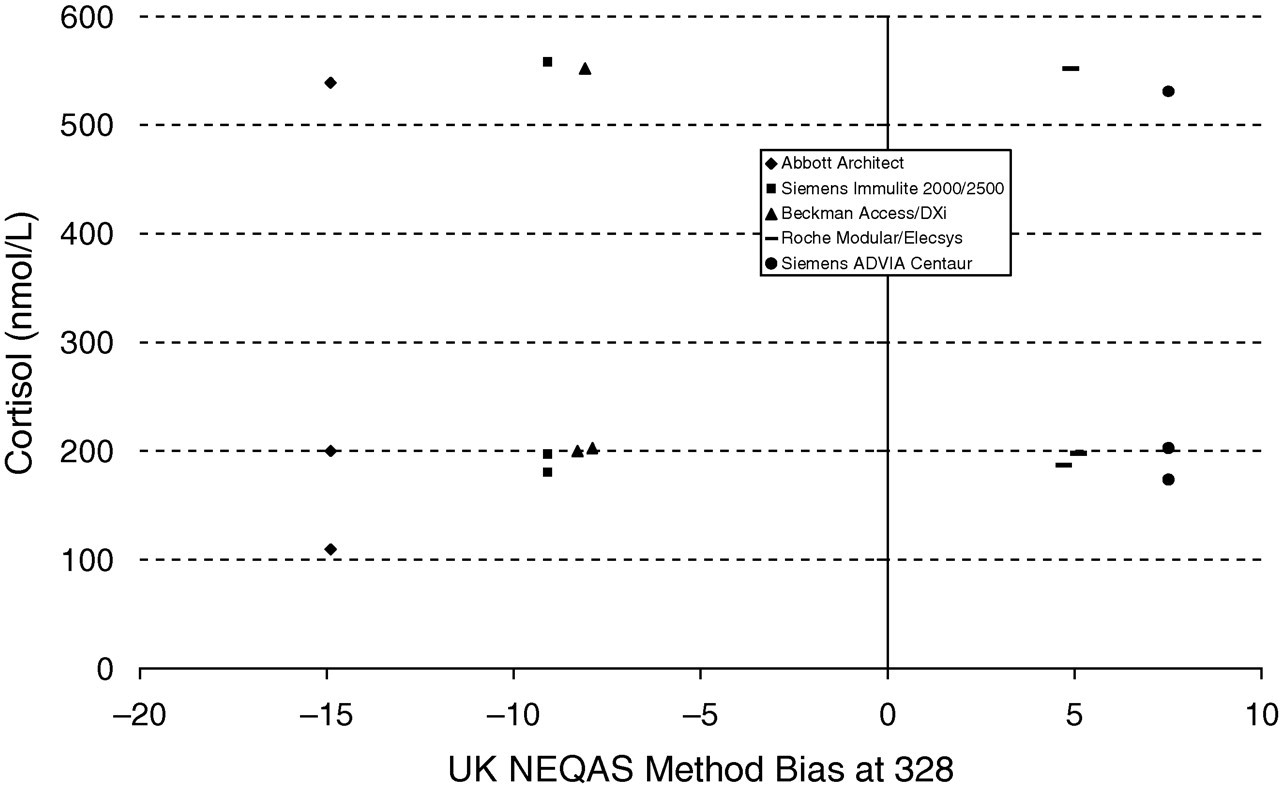
Cut-off values for baseline, increment and 30 min cortisol reported by laboratories in relation to the mean UK NEQAS method bias at distribution 328
[Q6.1] Do you take account of your method's bias when interpreting SST results?
Only 19% of laboratories take method bias into account when interpreting their SST results (Figure 1).
[Q6.2] Do you add interpretative comments to SST reports?
Forty-four percent of laboratories would add an interpretative comment to all reports (Figure 1), while 39% would add comments to some reports, and 17% of laboratories would not add any comments to the reports. Of the 39% of laboratories that add comments to some of the reports, 20 laboratories would only add comments if the request was from a non-endocrinologist and two laboratories would make comments depending on the user. One laboratory would only comment if the result was abnormal or equivocal, while another one would only do so if the request came from a General Practitioner.
[Q6.3] What criteria do you use to define a normal test result (cortisol concentrations in nmol/L please, times in minutes)?
Sixty-nine percent of laboratories stated that a baseline cortisol between 100 and 200 nmol/L would be considered a ‘normal’ response. Seventy-three percent of laboratories consider a peak cortisol between 450 and 600 nmol/L a ‘normal’ response and 89% of laboratories consider an incremental increase of 200 nmol/L a ‘normal’ response to an SST. In contrast, two laboratories use an incremental rise of 170 nmol/L of cortisol.
[Q6.4] What are the sources of your interpretative criteria?
The sources of the interpretative criteria varied greatly, with most laboratories stating that they had agreed the criteria with the endocrinologist, used Barth, Butler and Hammond, 2001, 6 or used in-house-derived data.
[Q6.5] What actions or recommendations, if any, do you make if a test is abnormal?
The majority of laboratories would phone the result to the relevant clinician and refer the patient to an endocrinologist.
[Q7.1] Please add any comments you feel may be relevant or helpful to this survey.
One of the issues raised was that there are no separate reference ranges for men and women. Additionally, there were comments advocating standardization of SST procedures.
Analysis of SST protocols (standard operating procedures)
As part of the audit, we asked participating laboratories to provide a copy of their standard operating procedures (SOPs) for the SST. We then examined these in two ways: (1) did they comply with current CPA (UK) Ltd accreditation standards (version 2.00 September 2007) for document control and (2) did the SOP correspond with the response to our survey.
Ninety percent of the SOPs were legible; however, less than 30% were approved and less than 20% had risk assessments included.
Over 80% of the SOPs had reference ranges, clinical indications, procedures and interpretations stated.
One SOP fulfilled all of the criteria, with eight SOPs matching 40–50% of the criteria.
Fifty percent of the SOPs matched the reference ranges provided on the questionnaire, while over 90% had stated the correct cut-off values.
Discussion
This ACB national audit showed that large and complex data could be collected using a web-based questionnaire and as such could be used as a template for further ACB national audits, negating the production of paper questionnaire and reducing difficulties in collating and analysing data. The design of this audit allowed us to capture data that could be separated into different parts: (1) the recommendations and procedures given by laboratories; (2) the analytical procedures and (3) the advice provided with the results.
The need for a national SST audit has been driven by the knowledge that the use of SSTs, since they were first introduced in the 1960s, 7 have largely replaced IST as a first-line test to assess adrenal insufficiency and the HPA axis. 8 However, though excellent correlation has been shown between the 30 min cortisol response and IST in aiding diagnosis of primary adrenal insufficiency, chronic secondary adrenal insufficiency 9,10 and patients on long-term corticosteroid use, 11 there have been concerns that the supraphysiological dosage of 250 μg ACTH can in patients with mild, recent or acute secondary adrenal insufficiency give a normal SST response but an abnormal IST. 12,13 As such, both laboratories and clinician should be aware of these limitations. It has been suggested that a ‘physiological’ dose, 1 μg ACTH, SST may be more suitable for detecting mild secondary insufficiency, 14 yet it has seen stated to be less practical and more prone to dosing errors, as it requires dilution of the 250 μg vials. 4
Another concern raised relates to the use of cut-off values that have been derived from fluorimetric methods for the measurement of free 11-hydroxycorticoids in human plasma, 7,15 which has been concluded to give significantly higher results than immunoassays, because of the measurement of both cortisol and corticosterone in these fluorimetric assays, particularly at high concentrations. 8 The data collected from this audit has shown that the majority of laboratories would consider a basal cortisol of 200 nmol/L, a 30 min cortisol of >550 nmol/L and an incremental cortisol increase of 200 nmol/L from the baseline, as a normal response to synthetic ACTH stimulation. However, despite knowing the bias of their own methods from NEQAS returns, only 19% of laboratories take this into consideration when interpreting the results, which is recommended by the All Wales Clinical Biochemistry Audit Group, recommendation 6b. 5 Additionally, recommendation 6a states that the incremental increase in cortisol after Synacthen is an unreliable index of adrenal function, though it may have merit in some instance. 5
The requirement to take method bias into account is highlighted in Figure 3, which shows the cut-off values reported by laboratories in relation to the mean UK NEQAS method bias at distribution 328. It can be seen that Siemens Immulite 2000/2500 has a method bias of −9% and the Roche Modular/Elecsys method has a bias of 5%, but the cut-off levels are the same, which clearly could affect interpretation of SST results.
Despite improvements in calibration and specificity of immunoassays, there is still significant method variation. 16 At nominal cortisol concentrations of 200 and 550 nmol/L (derived by the ID-GCMS reference method), routine immunoassay analysers would give results that vary from 175 to 215 nmol/L and 435 to 609 nmol/L, respectively (Table 2). Clearly, the same patient would ‘pass’ the SST if assessed at one hospital but would ‘fail’ the test at another, leading to misdiagnosis and mismanagement. Additional analytical variation can be demonstrated by Figure 2, which shows the reported percent coefficient of variation (%CV) for each method, with Siemens Centaur (CO10) having a reported %CV of 0–12.8% by different users. If the data are correctly stated on the questionnaire, then cortisol analysis on the same method can provide significantly different results depending on the location of the laboratory. Cortisol measurement can also be further confounded by the release of steroids other than cortisol in response to synthetic ACTH, 17,18 which may cross-react in the assay.
Cortisol values (nmol/L) for the different cortisol methods calculated using the ID-GCMS regression parameters (method mean versus ID-GCMS assigned value) in the UK NEQAS for steroid hormones reference value exercise
AB13, Abbott Architect; CO10, Siemens Centaur; SF1, Beckman Access; DC11, Siemens Immulite 2000/2500; E170, Roche E170 modular; BO5, Roche Elecsys; DC7, Siemens Immulite 1
The audit showed that although laboratories analysed SSTs routinely, the screening criteria for an SST varied with over 51% recommending a 09:00 cortisol and 39% selecting ‘other’, which included the acceptance of a random cortisol and the need for strong clinical suspicion. Of that 39%, seven of the laboratories would proceed to SST without screening patients first. This can be supported by the recommendations from the Wales Audit Group, 5 who do not recommend a random cortisol, except in certain clinical settings (e.g. hypoglycaemia). However, they do not make any recommendations regarding screening using a 09:00 cortisol, which has been reported to have a sensitivity of 91% and a predictive value to diagnose ACTH deficiency of 67% at cortisol concentrations of >400 nmol/L in postoperative patients. 19 It has been stated that a cut-off value of >525 nmol/L for a 08:00–09:00 cortisol can be accepted to rule out adrenal insufficiency, 20 which three of the laboratories that responded to this questionnaire have adopted for screening. Whether other laboratories use the same cut-off value could not be shown from the data gathered and may be a point to consider in a re-audit and additionally, identify whether method bias is translated to the values used in this instance.
A number of considerations must also be taken into account when using a basal morning cortisol value, as there is a broad range (139–695 nmol/L), which may be affected by stress, exercise and food intake. 18 The effect of oestrogen on the production of cortisol binding globulin and surgery must also be considered with evidence showing that SST is of limited use in the early postoperative phase after pituitary surgery. 21 Some of the laboratories were aware of these contraindications, but the majority acknowledged the importance of identifying patients with asthma and hypersensitivity to ACTH.
The provision of advice by laboratories to clinician's prior and after an STT highlights the caveats associated with the test, particularly with patients taking steroid therapy as they cross-react with the methods, and suppress the adrenals. 22 It is recommended that patients be switched to dexamethasone three days prior to the test; 5 however, measurement 24 h after switching has also been suggested. 22 Interestingly, though 50% of laboratories would suggest switching to dexamethasone, we did not gather data regarding the accepted time after switching before performing an SST. Also, 28% would omit the evening/morning dose, which if the test were performed in the morning would fail to meet the above recommendations, possibly affecting cortisol measurement.
All laboratories stated that a dosage of 250 μg Synacthen is administered during the Synacthen test and most generally agreed that a basal and 30 min blood sample should be taken, while a significant number analysed a 60 min sample. The dosage for paediatric patients are determined using a calculation; 250 μg/1.73 m2 body surface area.
The need for national consensus guidelines was highlighted by the varied response to the literature source used to interpret SSTs. Though there are standards produced by the Wales Audit Group, only four laboratories in this audit referred to them. However, there are US guidelines available for SSTs in critically ill patients, 23 which only states three recommendations regarding diagnosis for adrenal insufficiency.
Based on the data produced from this audit and standards from Wales Audit Group, we can make a number of recommendations, the first being that laboratories should have clear protocols for SSTs, which conform to national document control standards and must be agreed with clinicians. Laboratories should be clear on recommendations and contraindications and should have better understanding of the technical limitations of their methods in terms of cross-reactivity and bias. Importantly, laboratories should take bias into account when setting reference ranges and decision limits. In addition, there is evidence that there may be gender differences, 8 which may not have been identified in earlier methods, 7 due to imprecision and non-specificity. 8 This particular point was mentioned in the section for final comments, which may need further investigation. The production of a national guideline is recommended and clearly required, which with clear standards can help reduce variability in the procedures and interpretation of SSTs.
Additionally, there are a number of issues that must be resolved including understanding method bias and interpreting this from UK NEQAS reports, as we have found in this audit that 44% of laboratories gave the incorrect methods bias on the questionnaire. We recommend adjusting cut-off points for bias and standardizing the timing of the test along with a definition of baseline cortisol concentrations. A significant number of laboratories (56%) are not commenting on all the results, which is recommended by the Wales Audit Group, recommendation 6c. 5 Laboratories should also research their sources of information for the interpretation of SSTs again, as our data have identified the use of varied references, some of which may not be up to date. This may be aided by the production of national guidelines. Standard operating procedures should be updated to meet CPA document control standards as we showed that of the 22 SOPs we received, only seven laboratories matched over 50% of the standards.
In conclusion, though this audit demonstrated limitations of the current SST procedures, methodology and interpretation, they still play an important role as a first-line test to exclude adrenal insufficiency; 4 however, strong clinical suspicion to identify false-negative results must be maintained. 19 Although the analytical performance of cortisol methods has improved, 16 the method bias of cortisol analysis must be considered. The development of clinical guidelines for SSTs would present an opportunity to re-audit the processes against the standards and aid in the improvement in the quality of the service that laboratories provide.
DECLARATIONS
