Abstract
Background
Liquid chromatography linked to tandem mass spectrometry (LC/MS/MS) provides the ability to identify a range of benzodiazepines in accordance with European Union criteria and is an attractive method for the confirmation of benzodiazepines following immunoassay screening.
Methods
An LC/MS/MS method to detect and quantitate the six most common benzodiazepines/metabolites (diazepam, nitrazepam, nordiazepam, oxazepam, temazepam and 7-aminonitrazepam) was developed together with a qualitative screening method for a further 11 benzodiazepines/metabolites. These methods were used for confirmation of 250 urine samples submitted for routine drug screening by immunoassay for benzodiazepines (100 samples positive for a benzodiazepine, assay cut-off >200 μsg/L).
Results
The lower limits of detection and quantitation were less than 2.5 and 5 μg/L for the six most common benzodiazepines. Recoveries ranged between 97% and 102% and calibration curves were linear to at least 4000 μg/L (r = 0.99). Intra and inter-assay imprecision were <10% (n = 10) and <20% (n = 15), respectively. Confirmation of benzodiazepines using LC/MS/MS was achieved for 89% of the immunoassay-positive urine samples. Of the immunoassay-negative urine samples, 31% of these demonstrated a benzodiazepine using LC/MS/MS.
Conclusion
The validated LC/MS/MS method developed is effective for the confirmation of immunoassay screening results for benzodiazepines. The lower limit of detection and assay specificity offers a longer window of detection and more detailed clinical information compared with immunoassay screening.
Introduction
The identification and quantification of benzodiazepines in urine is clinically important for the detection of abuse and for compliance monitoring, where a drug treatment programme is in place. The detection of benzodiazepines in urine must combine specificity, sensitivity and repeatability.
The development of a comprehensive benzodiazepine screening method is challenging as a result of the large number of benzodiazepines available and the wide variation in urine concentrations of benzodiazepines and their metabolites. Due to the extensive metabolism of benzodiazepines their detection has focused around the reliable identification of the excreted metabolites. A number of methods have been proposed for the detection of benzodiazepines and their metabolites including radioimmunoassays, 1 radioreceptor assays, 2 fluorescence polarization immunoassays 3 and enzyme multiplied immunoassay technique (EMIT). 4 Immunoassays are the most common method used for the screening of urine samples for benzodiazepines; but their use is not without considerable limitations. Immunoassays are unable to distinguish between individual benzodiazepines present and no individual immunoassay kit can recognize all benzodiazepines and their metabolites at clinically relevant concentrations. The goal of any drug screening programme must be 100% accuracy, with no false-positive or false-negative results reported. However Fitzgerald et al. 5 and Borrey et al. 6 demonstrate that immunoassays commonly underestimate the presence of benzodiazepines. Immunoassay kits for the detection of benzodiazepines, therefore, remain an initial qualitative screening tool and the results should be considered presumptive until confirmed, as highlighted by the Society of Forensic Toxicology (SOFT) guidelines. 7
Gas chromatography linked to tandem mass spectrometry (GC/MS) remains the gold standard in drug confirmation; however, liquid chromatography linked to tandem mass spectrometry (LC/MS/MS) continues to gain acceptance. LC/MS/MS in multiple reaction monitoring (MRM) mode allows many drugs to be detected in one chromatographic run, offering the ability to identify and quantify a panel of benzodiazepines/metabolites, larger than with current GC/MS methods, with simpler sample preparation and shorter run times. 8,9
Several LC/MS/MS methods for the identification of benzodiazepines in urine have been described but without complete reference to international identification criteria. 8–12 Methods that do not use time-of-flight mass spectrometry are subject to co-elution of isobaric compounds that may result in the reporting of false-positive results. To date, there is very little work in assessing the use of LC/MS/MS for the confirmation of benzodiazepines following immunoassay screening in routine practice. In order to improve the confirmation rate for benzodiazepines, while minimizing false-positive results, a sensitive LC/MS/MS method, with well-defined internationally accepted identification criteria, is required. We have developed an LC/MS/MS method for the detection and quantitation of six common benzodiazepines/metabolites, and a screening method for a further 11 benzodiazepines/metabolites. This method was assessed in the confirmation of results on urine samples screened for benzodiazepines following analysis by EMIT.
Materials and methods
Solvents and reagents
Standards of diazepam, nitrazepam, nordiazepam, oxazepam, temazepam, 7-aminonitrazepam, flunitrazepam, 7-aminoflunitrazepam, clonazepam, 7-aminoclonazepam, flurazepam, 2-hydroxyethylflurazepam, alprazolam, α-hydroxyalprazolam, chlordiazepoxide, lorazepam and lormetazepam were purchased from LGC Standards (LGC, Middlesex, UK). Internal standards, diazepam-d5, nitrazepam-d5, nordiazepam-d5, oxazepam-d5, temazepam-d5, 7-aminoflunitrazepam-d7, 7-aminoclonazepam-d4, 2-hydroxyethylflurazepam-d5, α-hydroxyalprazolam-d5, alprazolam-d5, chlordiazepoxide-d5 and lorazepam-d4 were also purchased from LGC standards. β-Glucuronidase Type L-II from Patella vulgata (keyhole limpet), 1,000,000–3,000,000 units per grams solid was purchased from Sigma-Aldrich (Dorset, UK). All solvents were high-performance liquid chromatography (HPLC) grade, purchased from Sigma-Aldrich. Deionized water was generated in-house with an ELGA Purelab system (Marlow International, Marlow, UK).
Preparation of standards, internal standards and buffer solutions
A working stock solution of the six common benzodiazepines/metabolites was prepared in methanol containing 10 μg/mL diazepam, nitrazepam, nordiazepam, oxazepam, temazepam and 7-aminonitrazepam. The working stock was used to spike drug-free urine to 4000 μg/L, which was serially diluted to give a set of standards over a range of drug concentrations, 4000, 2000, 1000, 500, 250, 125 and 62.5 μg/L.
A mixed internal standard for the six most common benzodiazepines/metabolites was prepared in methanol containing 1 mg/L of diazepam-d5, nitrazepam-d5, nordiazepam-d5, oxazepam-d5 and temazepam-d5. A deuterated form of 7-aminonitrazepam was not available; therefore, diazepam-d5 was used as an internal standard for this metabolite.
A mix of the less common benzodiazepines/metabolites was also prepared in methanol containing 10 mg/L flunitrazepam, 7-aminoflunitrazepam, clonazepam, 7-aminoclonazepam, flurazepam, 2-hydroxyethylflurazepam, alprazolam, α-hydroxyalprazolam, chlordiazepoxide, lorazepam, lormetazepam. This was further diluted to 500 μg/L to provide a standard marker for screening.
Corresponding deuterated internal standards for the above benzodiazepine/metabolites were also prepared at 1 mg/L in methanol, for use where confirmation of the corresponding benzodiazepine was required: 7-aminoflunitrazepam-d7, 7-aminoclonazepam-d4, 2-hydroxyethylflurazepam-d5, α-hydroxyalprazolam-d5, alprazolam-d5, chlordiazepoxide-d5 and lorazepam-d4.
Alkaline extraction buffer was made by dissolving 318 g ammonium sulphate, 75 g sodium carbonate and 51 g sodium hydrogen carbonate (all from BDH, Lutterworth, Leicestershire, UK) in 1000 mL deionized water. Extraction solvent was made by mixing 520 mL dichloromethane (VWR, Lutterworth), 520 mL dichloroethane (York Glassware, York, UK), 600 mL heptane (York Glassware) and 380 mL propan-2-ol (York-Glassware).
A 2 mol/L sodium acetate buffer pH 4.4 was made by mixing 370 mL of 2 mol/L sodium acetate solution (272.16 g sodium acetate trihydrate [BDH] dissolved in 1 L deionized water) and 630 mL 2 mol/L acetic acid (57.2 mL glacial acetic acid [BDH] added to 442.8 mL deionized water) and was adjusted to pH 4.4.
β-Glucuronidase enzyme solution was prepared at 50,000 units/mL from the solid by dissolving powder containing 500,000 units in 10 mL of deionized water and stored at −20°C in aliquots.
Urine specimens
All the urine specimens used were anonymized samples, submitted for routine clinical drugs of abuse testing and surplus to clinical analytical requirements.
A total of 250 urine samples were analysed, of which 100 were immunoassay positive and 150 immunoassay negative. Drug-free (blank) urine was obtained from one individual member of laboratory staff over a two-day period.
Immunoassay screen
Urine specimens transferred to 5 mL polystyrene test tubes and centrifuged at 3000
Extraction and hydrolysis method
A volume of 500 μL urine, standard or quality control (QC) and 25 μL internal standard were added to 2 mL microtubes (Sarsdedt, Leicester, UK) containing 50 μL sodium acetate buffer. β-Glucuronidase (50 μL) (50,000 units/mL) was added to each tube, prior to mixing and incubated for 30 min at 56°C in a water bath. Hydrolysed samples were added to 10 mL glass tubes containing 2 mL of alkaline extraction buffer. Two mililitres of extraction solvent were then added to each tube. Tubes were then placed on a rotating mixer for 10 min and centrifuged at 3500 rpm for 10 min. The upper solvent layer was removed by pipette and placed into clean 10 mL wide neck glass tubes (Lab sales, Cambridge, UK) and evaporated to dryness under a stream of nitrogen with the tubes immersed in a Turbovap water bath at 60°C. Samples were reconstituted in 300 μL of mobile phase A (4 mmol/L ammonium acetate, pH 6.8 containing 5% [v/v] methanol in HPLC grade water) and mixed by vortex for 10 s before being transferred to a clean 96-well microtitre plates (Sarsdedt), sealed with cling film and placed into the mass spectrometer for immediate analysis.
High-performance liquid chromatography
Liquid chromatography was performed using a Shimadzu system (Shimadzu UK, Milton Keynes, UK) comprising a SIL-HT autosampler, 2× LC10AS pumps, a DGU-14A degasser and a CTO-10AS column over. The column effluent was directed either to the tandem mass spectrometer or to waste via a Valco switching valve. The set-up was controlled by Analyst 1.4 software (Applied Biosystems, Warrington, Cheshire, UK), which was also used for data collection and interpretation. Chromatographic separation was achieved using a 150 mm × 3 mm HyPURITY C8 column (Thermo Electron Corporation, Runcorn, Cheshire, UK) fitted with a security guard column of the same packing material (Phenomenex, Cheshire, UK). The column was maintained at 35°C and equilibrated at a flow rate of 0.8 mL/min using Mobile phase A. A gradient elution was initiated at this flow rate following injection of 50 μL of sample. Mobile phase A was maintained at 100% for one minute followed by a linear increase in Mobile phase B (1% [v/v] propan-2-ol, 0.05% [v/v] formic acid in 100% HPLC grade methanol) over three minutes. Mobile phase B was maintained at 100% for 1.5 min and then reduced to 0% over 0.1 min and maintained at this level for 1.4 min. The column eluate was directed to waste for the first 1.5 min, to the mass spectrometer for four minutes and then to waste for the remainder of the time programme during column re-equilibration.
Tandem mass spectrometry
The drugs were detected using an Applied Biosystems/MDS Sciex API 3000 Triple-Quad Mass Spectrometer equipped with a TurboIonSpray source (Applied Biosystems). Analytes were detected in positive ion mode using MRM of the molecular ion transitions listed in Table 1. A primary and secondary transition was monitored for each analyte. Nitrogen was used for the nebulizer, curtain and collision gases with flow rates of 10, 10 and 9 L/min, respectively. The ion-spray voltage was 5 kV and temperature was 475°C. Identification and quantification of the drugs were carried using Analyst Software Version 1.4 (Applied Biosytems). Peak area ratios for each analyte in relation to the internal standards were computed for the common benzodiazepines/metabolites and fitted to calibration curves that were calculated using a linear fit model.
Multiple reaction monitoring (MRM) transitions and retention time used for each analyte
*Denotes primary MRM transition
Drug identification criteria
Following LC/MS/MS analysis, drug confirmation was performed in accordance with the European Union (EU) criteria. 13 A signal-to-noise ratio of >1:10 of the primary MRM transition and >1:3 of the secondary MRM transition indicated the presence of the analyte. Retention time criteria: the relative retention time (RRT), i.e. ratio of the chromatographic retention time of the analyte to that of the internal standard must correspond to that of an external standard, at a tolerance of ±2.5%. Relative MRM ion intensity criteria: the relative MRM ion intensity of the test specimen was compared with the relative ion intensity obtained from the respective 125 μg/L standard for each analyte within each analytical batch. The 125 μg/L standard represented an average median benzodiazepine/metabolite concentration found in patient samples. Drugs were classified as confirmed when the RRT was correct and the relative ion intensity was within the correct range. 13
Mass spectrometer tuning
Voltages and gas flows for optimal detection of the benzodiazepines/metabolites and internal standards were determined after infusion of pure solutions (500 μg/L) in 100% methanol. MRMs of ion pairs specific to each analyte are shown in Table 1.
A method to detect and quantify the six most common benzodiazepines/metabolites (diazepam, nitrazepam, nordiazepam, oxazepam, temazepam and 7-aminonitrazepam) was fully validated. An additional screening method was set up to identify 11 less common benzodiazepines (flunitrazepam, 7-aminoflunitrazepam, clonazepam, 7-aminoclonazepam, flurazepam, 2-hydroxyethylflurazepam, alprazolam, α-hydroxyalprazolam, chlordiazepoxide, lorazepam and lormetazepam).
Results
Investigation of matrix effects
Interference and matrix effects were investigated by analysing water, neat blank urine, and hydrolysed and extracted blank urine (n = 5) by LC/MS/MS. No benzodiazepines were detectable in any of these samples. Ion suppression was investigated by direct infusion of each of the six common benzodiazepines/metabolites at 1000 μg/L in methanol directly into the mass spectrometer to provide a constant signal. An extracted urine sample containing a low dose of each benzodiazepine was injected via the autosampler. Ion suppression would be indicated by a reduction of greater than 10% in the signal for each compound provided by the constant infusion. 14 No significant ion suppression was observed for each of the benzodiazepines/metabolites.
Recovery
Recovery (extraction efficiency) was calculated by spiking 500 μL of drug-free urine and mobile phase A with the six common benzodiazepines to concentrations of 0.125, 250, 500, 1000, 2000, 4000 μg/L, in duplicate. Internal standards were added at a concentration of 100 μg/L. Urine samples were subjected to hydrolysis and extraction and were reconstituted with 500 μL mobile phase A. Average percentage recoveries were calculated by comparing peak area ratios from the analytes to the internal standards in spiked urine samples, to those of the unextracted spiked mobile phase A. Mean percentage recoveries ranged between 97.3% (temazepam) and 101.7% (oxazepam).
Sensitivity and specificity
A serial dilution was performed to produce a series of urine samples spiked with the six common benzodiazepines/metabolites at concentrations of 10, 5, 2.5, 1.25, 0.625, 0.3125 and 0.15625 μg/L. A volume of 500 μL of each spiked urine sample was hydrolysed and extracted, in duplicate.
Lower limit of detection (LLOD) was the lowest concentration for which the peaks of interest (both MRM transitions) had a signal-to-noise ratio of >3. The lower limit of quantitation (LLOQ) was defined as the lowest concentration that could be both identified and quantitated (signal-to-noise ratio >10). The LLOD and LLOQ were less than 2.5 and 5 μg/L, respectively, for all six benzodiazepines (Table 2). For continuity and ease of reporting a blanket LLOQ, cut off of 5 μg/L was set for all analytes. Samples with a concentration <5 μg/L were reported as such. Specimens with a concentration exceeding 4000 μg/L were reported as >4000 μg/L.
LLOD, LLOQ, r 2 and linear regression equation for each of the six common benzodiazepines/metabolites
LLOD, Lower limit of detection; LLOQ, lower limit of quantitation
Linearity
Linearity was evaluated by constructing a standard curve using the six common benzodiazepines. Blank urine was spiked at 4000 μg/L and was serially diluted to given standards of 2000, 1000, 500, 250, 125 and 62.5 μg/L. Calibration curves were linear to at least 4000 μg/L for each of the six common benzodiazepines (Table 2).
Inter- and intra-assay imprecision
In-house QC was generated by pooling 25 immunoassay-positive urine samples. Analysis of this pooled urine revealed diazepam, nitrazepam, nordiazepam, oxazepam, temazepam and 7-aminonitrazepam at concentrations ranging from 250 to 2500 μg/L. This sample was used as a high QC, which was diluted 1:1 with blank urine to provide a low QC.
Intra-assay coefficient of variation (CV) was found to be <10% (n = 10) for all benzodiazepines/metabolites in the quantitative assay for both QC materials. Inter-assay CVs (n = 15) were found to be <15% for all benzodiazepines/metabolites in the quantitative assay.
Patient samples
A total of 250 routine urine samples measured by the Synchron LX20 immunoassay (100 immunoassay-positive, 150 immunoassay-negative) were subjected to confirmation by LC/MS/MS. Peak resolution for each of the benzodiazepines was excellent overall, with very few samples showing isobaric interference or poor chromatography.
The most prevalent benzodiazepines identified were oxazepam (5–3060 μg/L, n = 94), temazepam (5–3020 μg/L, n = 106) and nordiazepam (5–3210 μg/L, n = 64), with these three benzodiazepines commonly confirmed in the same urine sample (n = 50); being consistent with diazepam metabolism. A small number of samples contained diazepam (5–56 μg/L, n = 11), nitrazepam (7–57 μg/L, n = 8) and/or 7-aminonitrazepam (9–2610 μg/L, n = 10). A very small number of samples had oxazepam (n = 1) or temazepam (n = 4) present at less than the LLOQ (5 μg/L), similarly the numbers of samples with concentrations greater than 4000 μg/L were very small, nordiazepam (n = 1), oxazepam (n = 6) and temazepam (n = 1). Of the 11 benzodiazepines analysed in the screening method, only lorazepam was confirmed (n = 6).
Confirmation of immunoassay results
Of the urine samples shown to be positive by immunoassay for benzodiazepines (n = 100), 89 could be confirmed by LC/MS/MS, with 73 samples being positive for at least two benzodiazepines/metabolites (Figure 1). The remaining urine samples unconfirmed by LC/MS/MS (false-positives) may be a result of drug interference in the immunoassay. Six of these urine samples were from patients prescribed quetiapine (n = 2), carbamazepine, clozapine, sertraline and mitazapine.
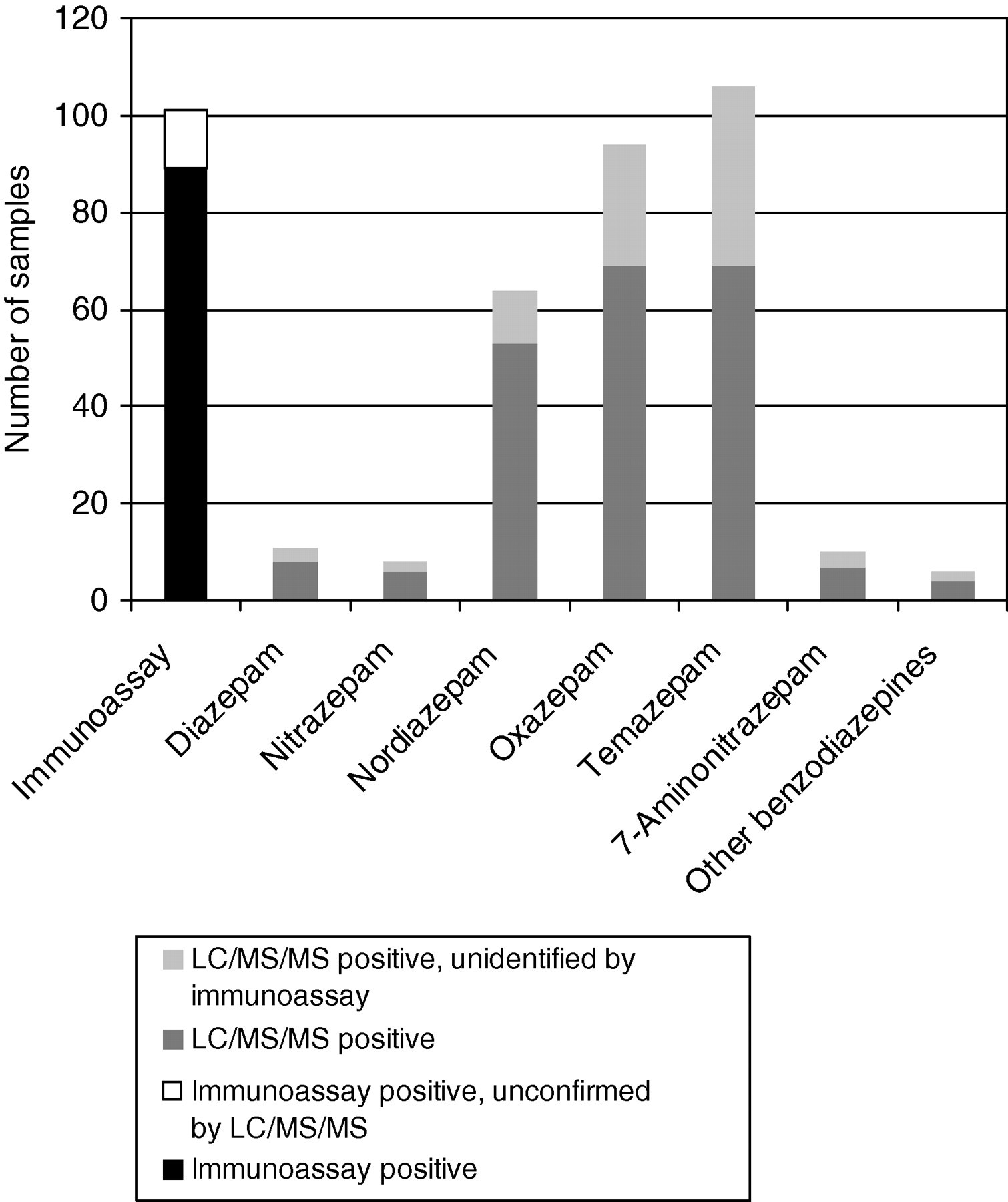
Confirmation of immunoassay (enzyme multiplied immunoassay technique) by LC/MS/MS for the detection of benzodiazepines in urine samples (n = 250). LC/MS/MS, liquid chromatography linked to tandem mass spectrometry
In the case of urine samples giving a negative result for benzodiazepines by immunoassay, 47 were confirmed positive for at least one benzodiazepine/metabolite and just over half of these (n = 24) were confirmed positive for two or more benzodiazepines/metabolites by LC/MS/MS, oxazepam and temazepam were the most common benzodiazepines/metabolites present (n = 24 and n = 37, respectively). The concentrations of all benzodiazepines identified and confirmed by LC/MS/MS in immunoassay-negative samples were below the immunoassay cut-off (200 μg/L). A large proportion (42%) of the urine samples that were confirmed to be false-negative results by LC/MS/MS were noted to have low creatinine concentrations (<4 mmol/L).
Discussion
Several methods have been developed to confirm the results of benzodiazepine screening by immunoassay, including HPLC with ultraviolet detection 15 and GC/MS. 5,16 However, these methods remain far from ideal, with complex sample preparation protocols, insufficient sensitivity and reports of temperature-induced degradation of certain benzodiazepines with GC/MS methods. 17 LC/MS/MS can be used to analyse almost all drugs of abuse in biological fluids, with equal or better specificity, usually better sensitivity and simpler sample preparation than GC/MS. 18 Recently, laboratories have invested increasingly in LC/MS/MS and it is set to become a core analytical tool. 19 This study demonstrates that LC/MS/MS can be used for the confirmation and quantitation of a range of benzodiazepines within a single chromatographic run of just six minutes.
The LC/MS/MS method developed and validated is highly sensitive method, with the LLOD below the recommended maximum detection limits for benzodiazepines in urine samples, set by the SOFT committee for drug-facilitated sexual assault. 20 The application of LC/MS/MS for the detection of benzodiazepines confers a number of advantages. The LLOD means that the window of detection for benzodiazepines in urine samples is longer and the identification of specific benzodiazepines will make more detailed clinical information available with the potential for detecting drug-facilitated sexual assault. However, the extensive metabolism of benzodiazepines prior to excretion may render identification of the parent compound difficult.
Previous studies have demonstrated that LC/MS/MS may lead to the reporting of false-positives due to the co-elution of isobaric compounds. 21–23 Confirmation of the identity of compounds of interest should follow specific accepted criteria to ensure that the analyte of interest is indistinguishable from that of the corresponding standard material (i.e. spiked samples in the same batch). 13,24 Some LC/MS/MS methods for the identification of benzodiazepines in urine have been without reference to international identification criteria. 8–12 Currently no specific criteria exist for the identification of illicit drugs by LC/MS/MS used in MRM mode. Two previous drugs of abuse confirmation studies 22,25 demonstrated that greater confidence in compound identification could be achieved by adopting the criteria defined by the EU. 13 Application of these criteria to the analyses of benzodiazepines and their metabolites by LC/MS/MS has provided confident identification following immunoassay screening.
It is widely accepted that immunoassay screening methods for the detection of benzodiazepines have significant limitations. Consequently, the results from immunoassay screening should be considered presumptive until confirmed by an alternative, more specific, accurate and sensitive technique. This study highlights the extent of the problems with one such current immunoassay screening method for benzodiazepines. A cut-off value of 200 μg/L for the immunoassay means a substantial number of samples submitted for routine drug screening are being reported as benzodiazepine negative when benzodiazepines are present; albeit at a concentration below the limit of detection. These false-negative samples represent a significant error rate in the current reporting practice for benzodiazepine results and may have considerable social, personal and economic impacts. Urine samples with a creatinine concentration below 4 mmol/L are indicative of possible adulteration or consumption of a large volume of fluid prior to sample collection and represent a significant subgroup within those received in the laboratory for routine drug screening. Dilute samples have an increased likelihood of containing benzodiazepines below the limit of detection of the immunoassay and being falsely reported as negative.
Previous reports have demonstrated that immunoassays for screening of benzodiazepines are subject to interference by a number of drugs and their metabolites. 26–29 In this study, 11% of samples with an immunoassay-positive result for benzodiazepines could not be confirmed by LC/MS/MS, highlighting the extent of possible drug interference in routine screening. Although the presence of an unknown benzodiazepine not included within the LC/MS/MS method needs to be considered, the manufacturer information provided for the EMIT immunoassay reports that, the presence of benzodiazepines could not be confirmed by GC/MS in 10 out of 158 immunoassay-positive urine samples but a number of other drugs were present in these samples. 30 Investigating possible drug interferences with immunoassays is challenging, because frequently drug metabolites, rather than the parent drug, may be responsible for the assay interference.
In summary, LC/MS/MS is effective in the confirmation and quantitation of benzodiazepines in urine samples submitted for routine drug screening by immunoassay. The application of internationally recognized validation criteria may help promote LC/MS/MS to the status of gold standard as an alternative to GC/MS.
DECLARATIONS
