Abstract
Background
Enzymatic assays using glutamate dehydrogenase (GLDH) to monitor the transformation of NAD(P)H to NAD(P)+ by a spectrophotometric technique are the most common methods to measure plasma ammonia (PA) in routine laboratories worldwide. However, these assays can potentially be subject to interference by substances in plasma able to oxidize NAD(P)H at a substantial rate, thereby providing falsely high results.
Methods
To study this potential interference, we spiked a plasma pool with a liver homogenate and measured the ammonia concentration using a dry chemistry system (Vitros 250, Ortho Clinical Diagnostic, Raritan, NJ, USA), an enzymatic assay without a sample blanking step (Infinity Ammonia Liquid Stable Reagent, Thermo Fisher Scientific, Waltham, USA) and an enzymatic assay that corrects for the non-specific oxidation of NADPH (Ammonia kit, RANDOX Laboratories Ltd, Crumlin, UK).
Results
This experiment shows that the Infinity™ ammonia reagent kit is subject to a clinically significant interference and explains the discrepancies previously reported between these methods in patients with acute liver failure (ALF).
Conclusion
When using enzymatic methods for the assessment of PA, we recommend including a sample blanking correction and this should be mandatory when monitoring patients with ALF.
Introduction
Interference produced by endogenous and exogenous substances in clinical chemistry assays is not an uncommon problem in laboratory medicine. 1
In a previous article, 2 we reported that the assessment of plasma ammonia (PA) in patients with acute liver failure (ALF) can be problematic when using enzymatic methods that do not include a sample blank correction step. We showed that the Infinity™ ammonia reagent kit was subject to interference by alanine amino transferase (ALT). However, we also concluded that this interference did not fully explain the discrepant results observed between this method and the dry chemistry system in patients with ALF. We speculated that the presence of dehydrogenases released into the systemic circulation during the acute episode may account for the bulk of the interference.
To study this potential interference, we spiked a plasma pool with a liver homogenate and measured the ammonia concentration (AC) using a dry chemistry system (Vitros 250) and two enzymatic assays (Infinity™ kit and RANDOX kit).
In the Vitros 250 method, the ammonia in the sample diffuses through a semipermeable membrane into a layer containing an indicator dye (bromphenol blue). The AC is measured by reflectance spectrophotometry at 600 nm.
The other two methods are enzymatic and use glutamate dehydrogenase (GLDH) to catalyse the condensation of NH4+ and 2-oxoglutarate to form glutamate in the presence of NADH (Infinity™ kit) or NADPH (RANDOX kit for automatic applications). The concentration of ammonia is determined by monitoring the transformation of NAD(P)H to NAD(P)+ by a spectrophotometric technique at 340 nm. The Infinity™ method is a one-step enzymatic assay and with this assay format it is not able to perform a sample blank. The RANDOX method is a two-step enzymatic assay and uses the change in absorbance, before GLDH is added to the sample mixture, to correct the final change in absorbance for the non-specific oxidation of NADPH.
Material and methods
120 mg of a spare donor human liver used as a normal control for our enzymatic assays were used to prepare a liver homogenate in 250 μL of Milli Q water. The liver was homogenized for two minutes using a battery-operated hand-held homogenizer and sonicated twice for 30 s using a Soniprep 150 (MSE, UK). After centrifugation the supernatant was used to spike aliquots of a plasma pool. No ethical approval was necessary to perform this experiment.
Ammonia was measured using three different methods: an enzymatic-UV kit from Thermo Fisher Scientific (Infinity™), two new formulations of this kit with an ALT inhibitor; an enzymatic-UV kit from RANDOX; and a dry chemistry system (Vitros 250). The regression curves and confidence intervals were calculated using Microsoft Excel 2003. ALT and aspartate aminotransferase (AST) were measured on Olympus AU640 analyser (Olympus UK Ltd, Hertfordshire, UK).
The correlation between the Infinity™ kit and the Vitros 250 was previously reported 2 (Infinity™ kit = 1.12 × Vitros 250 + 39, R 2 = 0.95, n = 105, range [5–800 μmol/L]) and between the RANDOX kit and the Vitros 250 was performed as part of the evaluation of the RANDOX kit (RANDOX kit = 1.13 × Vitros 250 + 100, R 2 = 0.90, n = 39, range [5–830 μmol/L]).
Results
Table 1 shows the concentrations of ammonia in the plasma pool spiked with the liver homogenate, assessed by the different methods. The AC in the plasma pool increased in all assays with increasing volumes of liver homogenate (VLH) due to the presence of ammonia in the liver homogenate; however, this increase is greater in the three formulations of the Infinity™ kit.
Summary of the ammonia concentration results
ALT, alanine amino transferase; AST, aspartate aminotransferase
In order to compare the ammonia results of these two methods with the Vitros 250 assay, the regression line of the AC against the VLH was calculated for the three methods (Figure 1). The slope and its 95% interval of confidence in brackets were used to compare the AC.
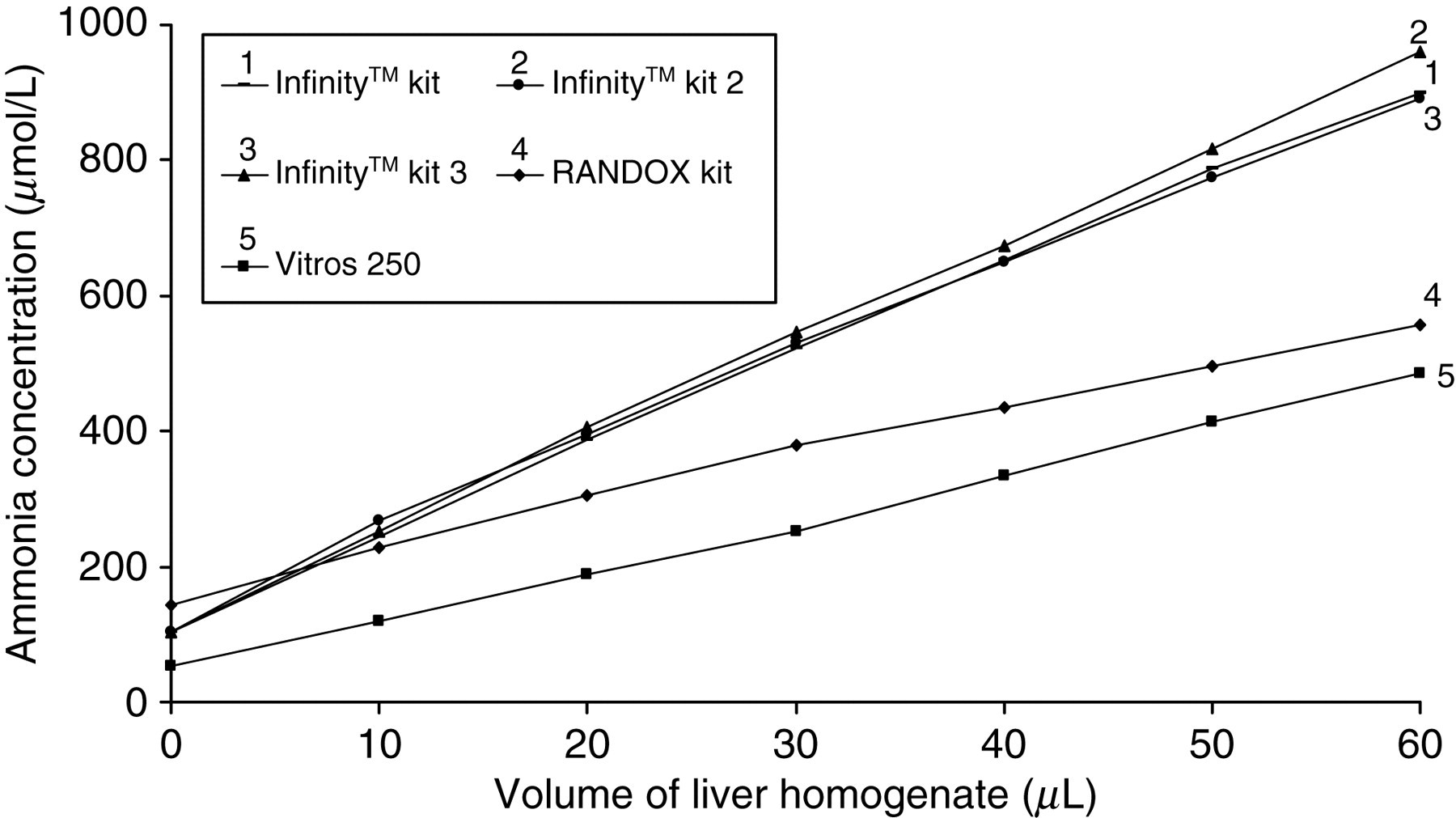
Ammonia concentration in the plasma pool spiked with liver homogenate measured with the three formulations of the enzymatic-UV (Infinity™) kit, the enzymatic-UV RANDOX kit and the dry chemistry Vitros 250 analyser. The regression curves are as follows: (AC [Vitros 250] = 47 + 7.26 [6.96–7.56] × VLH, R 2 = 0.999), AC [RANDOX] = 160 + 6.79 [6.20–7.38] × VLH, R 2 = 0.994), (AC [Infinity™] = 113 + 13.36 [12.87–13.86] × VLH, R 2 = 0.999) AC (Vitros 250) = ammonia concentration measured by Vitros 250; AC (RANDOX) = ammonia concentration measured by RANDOX kit; (AC (Infinity™) = ammonia concentration measured by Infinity™ kit; VLH, volume of liver homogenate
Discussion
We previously reported clinically significant differences in PA concentrations between the Infinity™ kit and Vitros 250 in patients with ALF. These differences were partly attributed due to interference of ALT in the enzymatic method but we speculated that dehydrogenases present in the sample may account for the majority of the interference. 2
As a result of this initial report, Thermo Electron Corporation formulated two new kits with an ALT inhibitor. These kits were provided to us for further evaluation in patients with ALF.
To investigate the potential effect of dehydrogenases and intermediate metabolites present in the plasma of patients with ALF, we used a liver homogenate to spike a plasma pool with the intention of mimicking the sort of sample we find in patients with this condition. As can be seen in Table 1, the elevation of AST and ALT, with AST > ALT, reflects that this procedure is effective in releasing the mitochondrial components. We acknowledge that the sample obtained through this procedure will not be identical to the samples in patients with ALF, but is still valid to assess its interference in the different ammonia methods.
Figure 1 shows an increase in the AC in the pool with increasing amounts of liver homogenate. The slope in the Infinity™ kit (13.36 [12.87–13.86]) is greater than in the RANDOX kit (6.79 [6.20–7.38]) and Vitros 250 (7.26 [6.96–7.56]). The three formulations of the Infinity™ kit behave similarly as the main difference between them is the presence of an ALT inhibitor and the interference due to ALT is not a main contributor at high concentrations of ammonia. The slopes of the RANDOX kit and Vitros 250 are not statistically different and these kits behave similarly with increased volumes of the liver homogenate.
The Infinity™ kit is a single-step enzymatic assay and because of its design is not able to correct for the non-specific oxidation of NADH to NAD+ and it is subject to interference by substances such as dehydrogenases. The RANDOX kit is a two-step enzymatic assay based on GLDH and NADPH and it does perform a sample blank correction step, explaining why the results obtained are comparable to the Vitros 250.
In summary, we have shown that the enzymatic -UV Infinity™ kit for ammonia is subject to interference. This is in agreement with the findings previously reported in patients with ALF. 2 The users of this enzymatic kit should be aware of the limitations of this kit to monitor PA in patients with metabolic disorders, such as urea cycle defects or organic acidurias, as some of them can deteriorate and develop organ failure including ALF. 3,4 Under these circumstances, the assessment of ammonia status can be difficult and result in inappropriate treatment.
If enzymatic methods are used to assess PA concentration, we recommend using an assay that performs a sample blank correction to account for the non-specific oxidation of NAD(P)H.
DECLARATIONS
